 Back
BackChemotrophic Energy Metabolism: Glycolysis and Fermentation – Study Notes
Study Guide - Smart Notes

Introduction to Metabolism
Definition and Importance
Metabolism is the sum of all chemical reactions within a cell or organism. These reactions are essential for life, enabling growth, repair, and energy production. Metabolism is divided into two major categories: anabolic and catabolic pathways.
Anabolic pathways: Build up molecules (e.g., protein synthesis, DNA replication). These processes require energy input.
Catabolic pathways: Break down molecules to release energy (e.g., digestion, cellular respiration).
Metabolism drives essential biological processes such as growth, repair, and energy production.
ATP as an Energy Intermediate
Structure and Function of ATP
ATP (Adenosine Triphosphate) is the primary energy currency of the cell. It consists of adenine, ribose, and three phosphate groups. The bonds between the phosphate groups, known as phosphoanhydride bonds, store significant potential energy.
Energy Release: Hydrolysis of ATP (breaking the bond between the last phosphate) releases energy used for cellular processes.
ATP Cycle: ATP is regenerated from ADP and inorganic phosphate (Pi) through catabolic reactions such as cellular respiration.
Cellular Work: Energy from ATP hydrolysis powers muscle contraction, active transport, and enzyme-catalyzed reactions.
ATP is continuously recycled in cells, with humans turning over an amount of ATP roughly equivalent to their body weight each day.
Chemical Forces Behind ATP's High Energy
Phosphoanhydride bond strain and electrostatic repulsion between phosphate groups contribute to ATP's high energy.
Resonance stabilization upon hydrolysis further lowers the energy of the products, making the reaction highly exergonic.
Other High-Energy Molecules
Other molecules, such as GTP and creatine phosphate, also store chemical energy that can be converted to ATP. Reduced coenzymes like NADH are important energy carriers in metabolism.
Chemotrophic Energy Metabolism
Definition and Key Pathways
Chemotrophic energy metabolism refers to the process by which organisms obtain energy by oxidizing organic molecules. This process relies on redox reactions, where electrons are transferred from molecules such as glucose to electron carriers like NAD+ and FAD. The energy released is used to synthesize ATP.
Key Pathways: Glycolysis, Krebs Cycle, Oxidative Phosphorylation (Electron Transport Chain)
Energy Carriers: NADH and FADH2 shuttle energy to the electron transport chain.
Stages of Cellular Catabolism
Cellular catabolism occurs in three main stages:
Breakdown of macromolecules into simple subunits
Oxidation of subunits to acetyl-CoA (with production of some ATP and NADH)
Complete oxidation of acetyl-CoA to CO2 and H2O in the mitochondria, generating large amounts of ATP
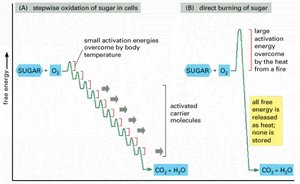
Stepwise Metabolism – Controlled Energy Harvesting
Cells harvest energy from nutrients in a stepwise manner, using a series of enzyme-catalyzed reactions. This allows energy to be captured efficiently as ATP, rather than being lost as heat.
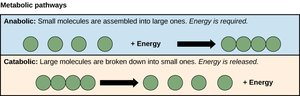
Metabolic Pathways
Anabolic vs. Catabolic Pathways
Metabolic pathways are sequences of enzyme-catalyzed reactions. Anabolic pathways synthesize cellular components and are endergonic (energy-requiring), while catabolic pathways break down molecules and are exergonic (energy-releasing).
Anabolic: Synthesize polymers like starch and glycogen; increase order, decrease entropy.
Catabolic: Degrade molecules like glucose; decrease order, increase entropy; release free energy.
Catabolic Pathways: Aerobic and Anaerobic
Catabolism can occur in the presence (aerobic) or absence (anaerobic) of oxygen. The reactions are not simply the reverse of anabolic pathways; different enzymes and intermediates are involved.
ATP: The Primary Energy Molecule in Cells
Structure and Hydrolysis of ATP
ATP contains adenine, ribose, and three phosphate groups. The phosphate groups are linked by phosphoanhydride bonds, which are "energy-rich." Hydrolysis of ATP to ADP and Pi is highly exergonic:
Charge repulsion: Negative charges on phosphate groups repel each other.
Resonance stabilization: Hydrolysis products (ADP and Pi) are more resonance-stabilized than ATP.
Increased entropy: Hydrolysis increases disorder and solubility of products.
ATP and Cellular Energy Metabolism
ATP occupies an intermediate position among energy-rich phosphorylated compounds. It can donate or accept phosphate groups, acting as the cell's "energy currency." The ATP/ADP system is a reversible means of conserving, transferring, and releasing energy.
Biological Oxidations and Electron Carriers
Oxidation and Reduction in Cells
Biological oxidations usually involve the removal of both electrons and protons (as hydrogen atoms) and are highly exergonic. Oxidation and reduction always occur together (redox reactions).
Coenzymes: Molecules like NAD+ serve as electron acceptors, becoming reduced to NADH.
Coenzymes are present in low concentrations and are recycled in the cell.
Glucose as an Energy Source
Glucose is the main energy source for most cells. Its oxidation is highly exergonic:
Complete oxidation of glucose in the presence of oxygen is called aerobic respiration. In the absence of oxygen, cells can extract limited energy from glucose via glycolysis and fermentation.
Aerobic and Anaerobic Respiration
Definitions and Comparisons
Aerobic respiration: Uses oxygen as the final electron acceptor; produces much more ATP (up to 38 ATP per glucose).
Anaerobic respiration: Uses alternative electron acceptors (e.g., sulfate, nitrate); produces less ATP.
Fermentation: A common form of anaerobic metabolism in eukaryotes; regenerates NAD+ by transferring electrons to organic molecules (e.g., lactate in humans, ethanol in yeast).
Types of Fermentation
Lactate fermentation: End product is lactate (in animals and some bacteria).
Alcoholic fermentation: End product is ethanol (in yeast and some plants).
Organismal Oxygen Requirements
Obligate aerobes: Require oxygen.
Obligate anaerobes: Oxygen is toxic; cannot use it as an electron acceptor.
Facultative organisms: Can function with or without oxygen.
ATP and Epigenetic Regulation
ATP in Epigenetic Modifications
ATP is not only an energy source but also plays a key role in epigenetic regulation, including histone modification (acetylation, methylation, phosphorylation) and ubiquitination. These modifications affect gene expression and cellular function.
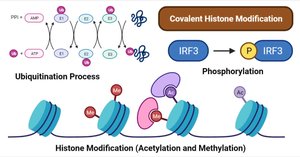
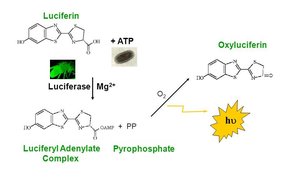
ATP Assay Example
Bioluminescence and ATP Measurement
ATP can be measured using bioluminescent assays, such as the luciferase reaction. In this reaction, luciferin is oxidized in the presence of ATP and luciferase, producing light. This is used in research and diagnostics to quantify ATP levels.
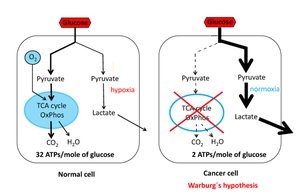
Aerobic Glycolysis in Cancer Cells
Warburg Effect
Cancer cells often rely on aerobic glycolysis (the Warburg effect), converting glucose to lactate even in the presence of oxygen. This process is less energy efficient (2 ATP per glucose) but is much faster than oxidative phosphorylation, supporting rapid cell proliferation.
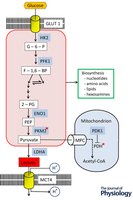
Enhanced Glycolysis and Fermentation in Cancer
Cancer cells upregulate glycolytic enzymes and lactate production, supporting biosynthesis and adaptation to hypoxic environments.
Sample Questions and Answers
Question | Answer |
|---|---|
Metabolism refers to: | All chemical reactions in a cell (C) |
Which is an anabolic process? | Protein synthesis (B) |
Catabolic pathways are responsible for: | Releasing energy by breaking down molecules (B) |
Main reason ATP is the energy currency of the cell: | It can release large amounts of energy when the bond between its last phosphate group is broken (B) |
Type of bond storing energy in ATP: | Phosphoanhydride bond (C) |
ATP hydrolysis produces: | ADP and Pi, releasing energy (B) |
During anaerobic respiration in humans, pyruvate is converted into: | Lactate (D) |
Anaerobic respiration generates less ATP because: | Oxygen is not used as the terminal electron acceptor (C) |
