 Back
BackChemotropic Energy Metabolism: Glycolysis and ATP in Cellular Bioenergetics
Study Guide - Smart Notes

Metabolism and ATP
Overview of Metabolism
Metabolism encompasses all chemical reactions occurring within a cell, divided into two main categories: catabolism (breakdown of molecules to release energy) and anabolism (synthesis of complex molecules from simpler ones, requiring energy input). These processes are fundamental to cellular function and energy management.
Catabolism: Increases entropy (S), is exergonic (releases energy), and occurs spontaneously.
Anabolism: Decreases entropy (S), is endergonic (requires energy), and is nonspontaneous.
ATP (Adenosine Triphosphate): Serves as the primary energy currency, linking catabolic and anabolic reactions.
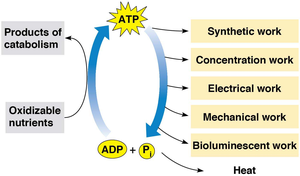
Additional info: ATP hydrolysis provides energy for various cellular processes, including biosynthesis, transport, and mechanical work.
ATP Structure and Energetics
ATP Hydrolysis and Bond Types
ATP contains high-energy phosphoanhydride bonds between its phosphate groups. Hydrolysis of the terminal phosphate group is highly exergonic, with a standard free energy change ( kcal/mol). This reaction is central to energy transfer in cells.
Phosphoanhydride bonds: Found between phosphate groups; hydrolysis yields two resonance-stabilized, negatively charged products.
Phosphoester bonds: Found between phosphate and sugar; hydrolysis yields only one resonance-stabilized product.
ATP hydrolysis: kcal/mol
AMP hydrolysis: kcal/mol
Why is ATP hydrolysis exergonic?
Charge repulsion: Three negatively charged phosphate groups repel each other.
Resonance stabilization: Hydrolysis products are more stabilized by resonance than ATP itself.
Increased entropy: Hydrolysis increases the number of molecules, raising entropy.
Additional info: The energy released from ATP hydrolysis is used to drive endergonic reactions in the cell.
Redox Reactions and NAD+
Oxidation-Reduction in Metabolism
Redox reactions are essential for energy transfer in cells. Oxidation is the loss of electrons (and usually protons), while reduction is the gain of electrons. Enzymes called dehydrogenases facilitate these reactions, often transferring hydrogen atoms.
NAD+ (Nicotinamide Adenine Dinucleotide): Functions as an electron carrier, becoming reduced to NADH during catabolic reactions.
Glucose oxidation: Glucose loses electrons (is oxidized), while oxygen gains electrons (is reduced to water).
Key equation for aerobic respiration:
Additional info: NADH produced in glycolysis and other pathways is later used to generate more ATP via oxidative phosphorylation.
Classification of Organisms by Oxygen Requirement
Oxygen Utilization Strategies
Organisms are classified based on their oxygen requirements for metabolism:
Obligate aerobes: Require oxygen for survival (e.g., most animals).
Obligate anaerobes: Oxygen is toxic; survive only in its absence (e.g., some bacteria and archaea).
Facultative anaerobes: Can use oxygen if present but can also survive anaerobically (e.g., some bacteria and fungi).
Glycolysis: The Central Pathway of Glucose Catabolism
Overview and Location
Glycolysis is a universal metabolic pathway that occurs in the cytosol of nearly all organisms. It does not require oxygen and converts one molecule of glucose (6 carbons) into two molecules of pyruvate (3 carbons each), generating ATP and NADH in the process.
Location: Cytosol (or glycosomes in some organisms)
Oxygen requirement: Anaerobic (does not require oxygen)
Products: 2 pyruvate, 2 NADH, 2 net ATP per glucose
Phases of Glycolysis
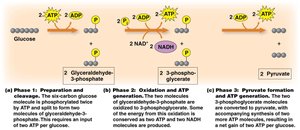
Glycolysis consists of three main phases:
Phase 1: Preparation and Cleavage (Energy Investment)
Phase 2: Oxidation and ATP Generation (First Energy Payoff)
Phase 3: Pyruvate Formation and ATP Generation (Second Energy Payoff)
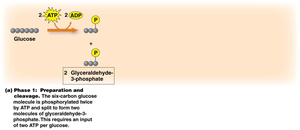
Phase 1: Preparation and Cleavage
In this phase, glucose is phosphorylated and split into two molecules of glyceraldehyde-3-phosphate (G3P). This step requires the input of 2 ATP per glucose molecule.
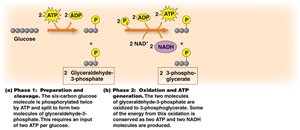
Phase 2: Oxidation and ATP Generation
Each G3P is oxidized, reducing NAD+ to NADH and generating ATP via substrate-level phosphorylation. This phase produces 2 NADH and 2 ATP per glucose.
Phase 3: Pyruvate Formation and ATP Generation
Further rearrangement and phosphorylation steps lead to the formation of pyruvate and additional ATP production. The net result is 2 pyruvate, 2 NADH, and 2 ATP per glucose molecule.
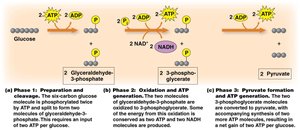
Summary of Glycolysis
1 Glucose molecule is oxidized to 2 pyruvate.
2 NADH are generated (used later for further ATP production).
2 ATP are used, 4 ATP are produced, resulting in a net gain of 2 ATP.
Overall free energy change: kcal/mol.
Comparison of Phosphoanhydride and Phosphoester Bonds
Bond Type | Location | Hydrolysis Products | Resonance Stabilization | Standard Free Energy Change () |
|---|---|---|---|---|
Phosphoanhydride | Between phosphate groups in ATP/ADP | 2 charged, resonance-stabilized products | Both products stabilized | -7.3 kcal/mol |
Phosphoester | Between phosphate and sugar in AMP | 1 charged, resonance-stabilized product | Only one product stabilized | -3.6 kcal/mol |
Key Enzymes in Glycolysis
Hexokinase: Catalyzes the phosphorylation of glucose to glucose-6-phosphate (G6P); highly exergonic and essentially irreversible.
Phosphofructokinase-1 (PFK-1): Catalyzes the phosphorylation of fructose-6-phosphate to fructose-1,6-bisphosphate; a key regulatory step.
Dehydrogenases: Catalyze oxidation-reduction reactions, transferring electrons to NAD+.
Summary
Metabolism is the sum of catabolic and anabolic reactions in the cell.
ATP acts as an intermediate-energy compound, linking energy-releasing and energy-consuming processes.
NAD+ is a key electron carrier, reduced to NADH during glycolysis and other pathways.
Glycolysis is a central pathway for glucose catabolism, producing ATP and NADH without the need for oxygen.
