 Back
BackCytoskeletal Systems: Structure, Regulation, and Function
Study Guide - Smart Notes

Cytoskeletal Structure
Overview of Cytoskeletal Elements
The cytoskeleton is a dynamic network of protein filaments that provides structural support, facilitates cellular movement, and organizes the cell's contents. Eukaryotic cells possess three main types of cytoskeletal filaments: microtubules, microfilaments (actin filaments), and intermediate filaments. Bacteria also have homologues for these proteins, including microtubule-like, actin-like, and intermediate filament-like structures.
Microtubules
Structure and Types of Microtubules
Microtubules are the largest cytoskeletal components, composed of tubulin subunits. They exist in two main forms:
Cytoplasmic microtubules: Pervade the cytosol and are involved in maintaining axons, forming mitotic and meiotic spindles, maintaining or altering cell shape, and facilitating vesicle movement.
Axonemal microtubules: Organized and stable, found in cilia, flagella, and basal bodies. The axoneme is the central shaft of a cilium or flagellum, consisting of a highly ordered bundle of microtubules.
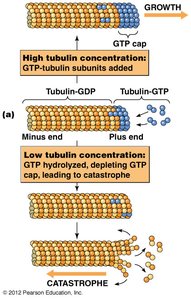
Microtubule Dynamics: Growth and Catastrophe
Microtubules exhibit dynamic instability, alternating between phases of growth and shrinkage. The presence of a GTP cap at the plus end stabilizes the microtubule, while loss of the cap leads to rapid depolymerization (catastrophe).
Microtubule-Organizing Centers (MTOCs)
MTOCs are cellular structures that nucleate and anchor microtubules, playing a critical role in organizing the cytoskeleton during cell division and interphase.
Microtubule-Binding Proteins
Microtubule stability and function are regulated by various microtubule-binding proteins:
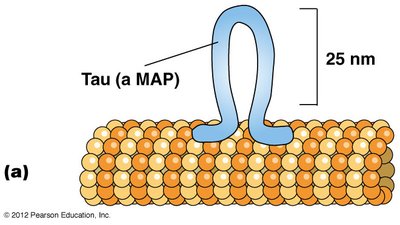
Microtubule-associated proteins (MAPs): Bind at regular intervals along microtubules, promoting bundling and interaction with other cellular structures. Tau forms tight bundles in axons, while MAP2 forms looser bundles in dendrites.
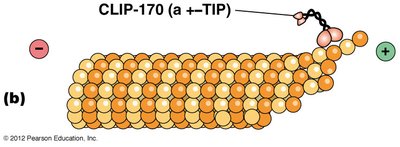
+–TIP proteins: Stabilize microtubules by capturing and protecting the growing plus ends, decreasing the likelihood of catastrophic loss.
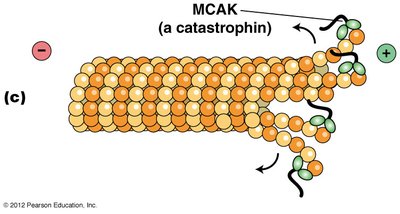
Microtubule-destabilizing/severing proteins: Promote depolymerization or severing of microtubules. Stathmin/Op18 binds tubulin heterodimers, preventing polymerization; catastrophins promote subunit loss; katanins sever microtubules.
The Mesh Organelle and Microtubule Stabilization
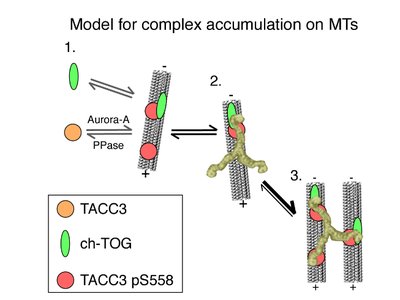
The Mesh is a recently discovered organelle composed of microtubule connectors that stabilize kinetochore fibers of the mitotic spindle. Clathrin, in its trimeric state, is a key component, holding two microtubules together. The Mesh is essential for chromosome transport during mitosis and meiosis, and mutations may contribute to cancer.

Clathrin complexes with TACC3 and ch-TOG proteins to stabilize kinetochore fibers. TACC3 recruitment is regulated by Aurora-A kinase phosphorylation, and clathrin bridges adjacent microtubules, locking the complex.
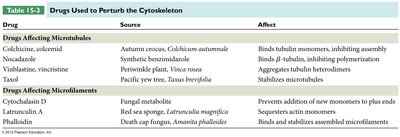
Drugs Affecting Microtubules
Several drugs can perturb microtubule dynamics, affecting cell division and structure.
Drug | Source | Affect |
|---|---|---|
Colchicine, colcemid | Autumn crocus, Colchicum autumnale | Binds tubulin monomers, inhibiting assembly |
Nocodazole | Synthetic benzimidazole | Binds β-tubulin, inhibiting polymerization |
Vinblastine, vincristine | Periwinkle plant, Vinca rosea | Aggregates tubulin heterodimers |
Taxol | Pacific yew tree, Taxus brevifolia | Stabilizes microtubules |
Microfilaments (Actin Filaments)
Structure and Function of Microfilaments
Microfilaments are the smallest cytoskeletal filaments, primarily composed of actin. They are crucial for muscle contraction, cell migration, amoeboid movement, and cytoplasmic streaming. Microfilaments also contribute to cell shape and form the structural core of microvilli.
Actin Polymerization and Microfilament Polarity
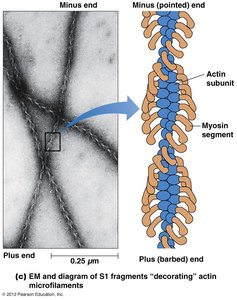
Actin exists as globular (G-actin) monomers that polymerize to form filamentous (F-actin) microfilaments. Microfilaments are polarized, with a barbed (plus) end and a pointed (minus) end, as demonstrated by the binding of myosin S1 fragments.
Drugs Affecting Microfilaments
Specific drugs can alter microfilament dynamics:
Drug | Source | Affect |
|---|---|---|
Cytochalasin D | Fungal metabolite | Prevents addition of new monomers to plus ends |
Latrunculin A | Red sea sponge, Latrunculia magnifica | Sequesters actin monomers |
Phalloidin | Death cap fungus, Amanita phalloides | Binds and stabilizes assembled microfilaments |

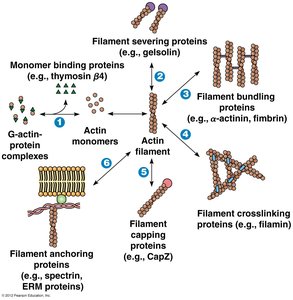
Actin-Binding Proteins
Actin-binding proteins regulate the polymerization, length, and organization of actin filaments. They control nucleation, elongation, severing, and the association of microfilaments into networks.
Intermediate Filaments
Structure and Function of Intermediate Filaments
Intermediate filaments (IFs) are the most stable and least soluble cytoskeletal components. They are not polarized and confer mechanical strength to tissues. Keratin is a prominent IF protein in animals, supporting structures that grow from skin.
Classes of Intermediate Filament Proteins
IF proteins are tissue-specific and grouped into six classes:
Class I: Acidic keratins
Class II: Basic or neutral keratins (tonofilaments in epithelial surfaces)
Class III: Vimentin (connective tissue), desmin (muscle cells), glial fibrillary acidic protein (glial cells)
Class IV: Neurofilament proteins (nerve cells)
Class V: Nuclear lamins A, B, and C (network along inner nuclear membrane)
Class VI: Nestin (nerve cells of embryos)
Animal cells can be distinguished based on the types of IF proteins they contain, a process known as intermediate filament typing.
Mechanical Integration of the Cytoskeleton
Cellular architecture depends on the unique properties of cytoskeletal elements:
Microtubules: Resist bending when a cell is compressed
Microfilaments: Serve as contractile elements generating tension
Intermediate filaments: Elastic, withstand tensile forces, and bear tension
IFs are dynamically transported and remodeled, especially during mitosis when the nuclear lamina disassembles and reassembles.
Integration of Cytoskeletal Elements
Linker proteins called plakins connect intermediate filaments, microfilaments, and microtubules. Plectin is a plakin found at sites where intermediate filaments connect to microfilaments and microtubules, contributing to the mechanical integration of the cytoskeleton.
Summary Table: Drugs Used to Perturb the Cytoskeleton
The following table summarizes drugs that affect microtubules and microfilaments:
Drug | Source | Affect |
|---|---|---|
Colchicine, colcemid | Autumn crocus, Colchicum autumnale | Binds tubulin monomers, inhibiting assembly |
Nocodazole | Synthetic benzimidazole | Binds β-tubulin, inhibiting polymerization |
Vinblastine, vincristine | Periwinkle plant, Vinca rosea | Aggregates tubulin heterodimers |
Taxol | Pacific yew tree, Taxus brevifolia | Stabilizes microtubules |
Cytochalasin D | Fungal metabolite | Prevents addition of new monomers to plus ends |
Latrunculin A | Red sea sponge, Latrunculia magnifica | Sequesters actin monomers |
Phalloidin | Death cap fungus, Amanita phalloides | Binds and stabilizes assembled microfilaments |

Additional info: The cytoskeleton is a mechanically integrated structure, with each filament type contributing unique properties to cellular architecture and function. The regulation of cytoskeletal dynamics is essential for processes such as cell division, migration, and intracellular transport.
