 Back
BackEnzyme Kinetics and Regulation: Mechanisms, Inhibition, and Biological Relevance
Study Guide - Smart Notes

Enzyme Kinetics and Regulation
Introduction to Enzyme Kinetics
Enzyme kinetics is the study of the rates at which enzymatic reactions proceed and the factors that affect these rates. Enzymes are biological catalysts that accelerate chemical reactions by lowering the activation energy required for the reaction to occur. Understanding enzyme kinetics is essential for elucidating how enzymes function in cellular metabolism and how their activity is regulated.
Enzyme Inhibition
Enzyme activity can be regulated by molecules known as inhibitors, which decrease or eliminate enzyme activity by interfering with substrate binding or enzyme function. There are two main types of reversible inhibition: competitive and noncompetitive inhibition.
Competitive Inhibition
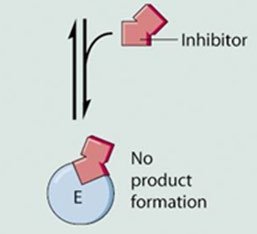
Definition: A competitive inhibitor resembles the substrate and competes for binding at the enzyme's active site.
Mechanism: When the inhibitor occupies the active site, the substrate cannot bind, and no product is formed.
Effect on Kinetics: Vmax remains unchanged, but a higher substrate concentration is required to reach Vmax, resulting in an increased Km.
Example: Many drugs act as competitive inhibitors, such as sulfa drugs inhibiting folic acid synthesis in bacteria.
Noncompetitive Inhibition
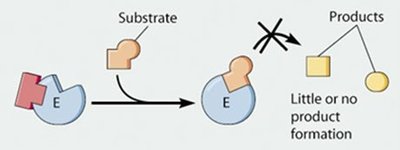
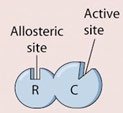
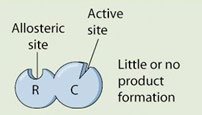
Definition: A noncompetitive inhibitor binds to an allosteric site (not the active site), causing a conformational change in the enzyme.
Mechanism: The substrate may still bind to the active site, but the enzyme is rendered inactive or less active, so little or no product is formed.
Effect on Kinetics: Vmax is reduced, but Km remains unchanged because substrate binding is not directly affected.
Example: Heavy metals like lead or mercury often act as noncompetitive inhibitors.
Allosteric Regulation
Allosteric regulation involves the binding of regulatory molecules (activators or inhibitors) at sites other than the active site, known as allosteric sites. This binding induces conformational changes that alter enzyme activity.
Negative Allosteric Regulation: Inhibitor binding decreases enzyme activity by reducing substrate affinity.
Positive Allosteric Regulation: Activator binding increases enzyme activity by enhancing substrate affinity.
Feedback Regulation in Metabolic Pathways
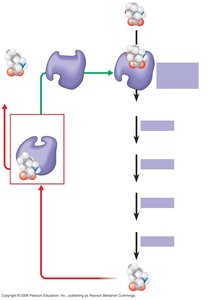
Metabolic pathways consist of a series of enzyme-catalyzed reactions. The activity of enzymes in these pathways can be regulated by the concentration of substrates or products, often through feedback mechanisms.
Feedback Inhibition: The end product of a pathway inhibits an enzyme involved earlier in the pathway, preventing overproduction of the product.
Example: Isoleucine biosynthesis from threonine is regulated by feedback inhibition, where isoleucine binds to an allosteric site on threonine deaminase, inhibiting its activity.
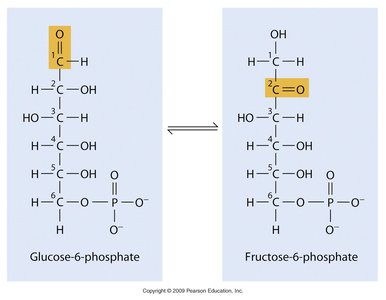
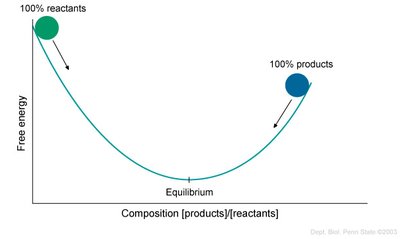
Reversible Reactions and Chemical Equilibrium
Many enzyme-catalyzed reactions are reversible, and the direction of the reaction depends on the relative concentrations of substrates and products. The equilibrium constant (Keq) is defined as:
Directionality: The reaction proceeds in the direction that moves the system toward equilibrium, where the free energy is minimized.
Genetic and Post-Translational Regulation of Enzymes
Genetic Regulation: The synthesis of enzymes can be controlled at the level of gene expression, affecting which enzymes are present in the cell.
Post-Translational Modification: Enzyme activity can be rapidly altered by covalent modifications such as phosphorylation or dephosphorylation.
Enzyme Localization
Enzymes are often compartmentalized within specific organelles in eukaryotic cells, which helps organize metabolic pathways and increases efficiency.
Example: Enzymes for cellular respiration are located in mitochondria.
Cofactors and Coenzymes
Cofactors: Non-protein components required for enzyme activity. They can be inorganic (e.g., metal ions like Fe2+, Ca2+) or organic (coenzymes, often derived from vitamins).
Examples: NAD+/NADH, FAD/FADH2
Enzyme Kinetics: Quantitative Analysis
Enzyme kinetics quantitatively describes how reaction velocity (v) depends on substrate concentration ([S]). The Michaelis-Menten equation is fundamental:
Vmax: Maximum reaction velocity when the enzyme is saturated with substrate.
Km: Substrate concentration at which the reaction velocity is half of Vmax; indicates enzyme affinity for substrate (lower Km = higher affinity).

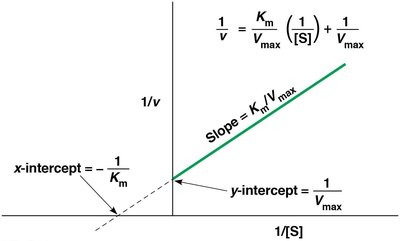
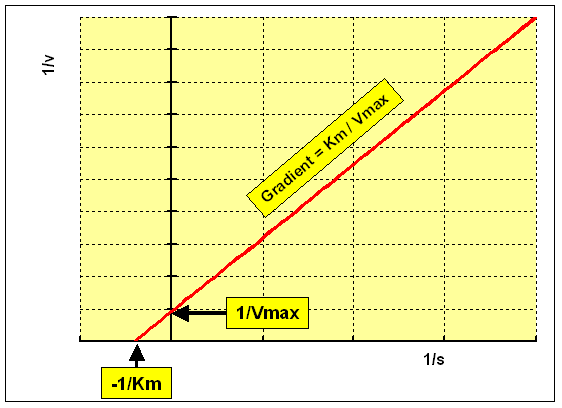
Lineweaver-Burk Plot
The Lineweaver-Burk plot is a double-reciprocal transformation of the Michaelis-Menten equation, yielding a straight line:
y-intercept:
x-intercept:
Slope:
Effects of Inhibition on Enzyme Kinetics
Competitive and noncompetitive inhibitors have distinct effects on kinetic parameters:
Type of Inhibition | Effect on Vmax | Effect on Km |
|---|---|---|
Competitive | No change | Increases |
Noncompetitive | Decreases | No change |
Summary Table: Key Terms and Concepts
Term | Definition |
|---|---|
Enzyme | Biological catalyst that accelerates chemical reactions |
Active Site | Region of enzyme where substrate binds |
Allosteric Site | Regulatory site distinct from the active site |
Competitive Inhibitor | Binds active site, blocks substrate |
Noncompetitive Inhibitor | Binds allosteric site, alters enzyme conformation |
Vmax | Maximum reaction velocity |
Km | Substrate concentration at 1/2 Vmax |
Cofactor | Non-protein component required for enzyme activity |
Coenzyme | Organic cofactor, often vitamin-derived |
Additional info: This guide integrates foundational concepts from cell biology and biochemistry, including enzyme structure, function, and regulation, as well as practical applications in medicine and biotechnology.
