 Back
BackMicrofilament-Based Motility and Muscle Contraction
Study Guide - Smart Notes

Microfilament-Based Motility
Overview of Microfilament-Based Motility
Microfilament-based motility is a fundamental process in eukaryotic cells, primarily driven by the interaction of actin filaments with myosin motor proteins. This system underlies a variety of cellular movements, including muscle contraction, cell migration, and intracellular transport.
Microfilaments are composed of actin and serve as tracks for myosin motors.
Myosins are ATP-dependent motor proteins that move along actin filaments, generally toward the plus (+) end.
Key cellular events involving myosins include muscle contraction, cell movement (e.g., lamellipodia, filopodia), phagocytosis, rearrangement of the cell cortex, and vesicle transport.
Myosin Motor Proteins
Structure and Function of Myosins
Myosins are a large superfamily of motor proteins that convert chemical energy from ATP hydrolysis into mechanical work, enabling movement along actin filaments.
All myosins have at least one heavy chain with a globular head (actin-binding and ATPase activity) and a tail of variable length.
Light chains are associated with the heavy chains and regulate ATPase activity.
Type II myosins, the best understood, have two heavy chains, each with a globular head, a hinge region, and a rodlike tail. Each heavy chain is associated with one essential and one regulatory light chain.
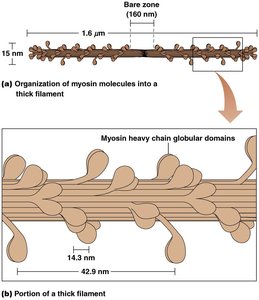
Type II myosins assemble into thick filaments, which pull arrays of actin filaments together, resulting in contraction.
Common feature of all motor proteins: They convert chemical energy (from ATP) into motion.
Muscle Contraction
Structure of Skeletal Muscle Cells
Skeletal muscle contraction is a classic example of microfilament-based motility. The contractile unit of muscle is the sarcomere, composed of interdigitating thick (myosin) and thin (actin) filaments.
Each muscle fiber contains many myofibrils, which are divided into repeating units called sarcomeres.
Sarcomeres contain bundles of thin filaments (actin, troponin, tropomyosin) and thick filaments (myosin).
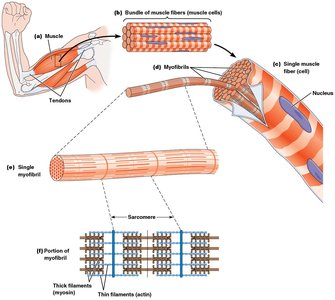
Organization of Sarcomeres
The arrangement of thick and thin filaments within sarcomeres gives skeletal muscle its striated appearance.
Dark bands (A bands) and light bands (I bands) are visible under the microscope.
The Z line marks the boundary of each sarcomere; actin filaments are anchored at the Z line with their plus ends.
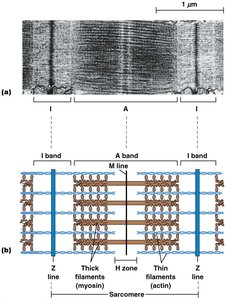
Thick and Thin Filaments
Thick filaments are composed of myosin molecules arranged in a staggered fashion, while thin filaments are composed of F-actin intertwined with tropomyosin and troponin.
Myosin heads protrude from thick filaments and form cross-bridges with actin in thin filaments.
Thin filaments interdigitate with thick filaments, enabling contraction.

The Sliding-Filament Model
Muscle contraction occurs as thin filaments slide past thick filaments, shortening the sarcomere without changing the length of either filament type.
Myosin II heads move toward the plus ends of actin filaments, pulling thick filaments toward the Z lines.
The force generated depends on the number of myosin-actin cross-bridges formed.
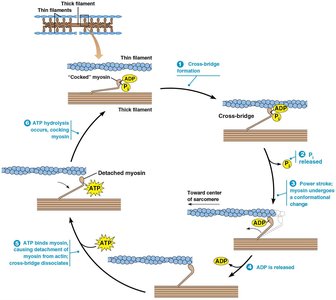
The Contraction Cycle
The contraction cycle is powered by ATP and involves repeated formation and dissociation of cross-bridges between myosin heads and actin filaments.
ATP binds to myosin, causing it to release from actin.
ATP hydrolysis "cocks" the myosin head.
Myosin binds to a new position on actin, releasing Pi.
The power stroke occurs as ADP is released, moving the actin filament.
Cycle repeats as long as ATP and Ca2+ are present.
Regulation of Muscle Contraction: The Role of Calcium
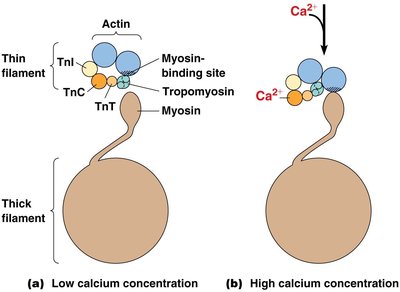
Muscle contraction is regulated by the availability of myosin-binding sites on actin, controlled by the proteins tropomyosin and troponin in a calcium-dependent manner.
At low Ca2+ concentration, tropomyosin blocks myosin-binding sites on actin.
When Ca2+ concentration increases, Ca2+ binds to troponin C (TnC), causing tropomyosin to shift and expose binding sites for myosin.
When Ca2+ levels fall, tropomyosin returns to its blocking position, and the muscle relaxes.
Regulation of Calcium Levels in Skeletal Muscle
Calcium levels in muscle cells are tightly regulated by nerve impulses and specialized ion channels and pumps.
Acetylcholine from motor neurons opens Na+ channels, depolarizing the muscle cell membrane.
Depolarization opens voltage-gated Ca2+ channels, leading to Ca2+ influx and release from the sarcoplasmic reticulum.
Muscle relaxation occurs as Ca2+ is pumped back into the sarcoplasmic reticulum by ATP-dependent pumps.
Smooth Muscle Contraction
Structure and Function of Smooth Muscle
Smooth muscle is responsible for involuntary contractions in various tissues and is structurally distinct from skeletal muscle.
Smooth muscle cells are long, thin, and lack striations; they have dense bodies instead of Z lines.
Bundles of actin and myosin are anchored at both ends to dense bodies.
Contractions are slower and of greater duration than those in skeletal muscle.
Regulation of Contraction in Smooth Muscle
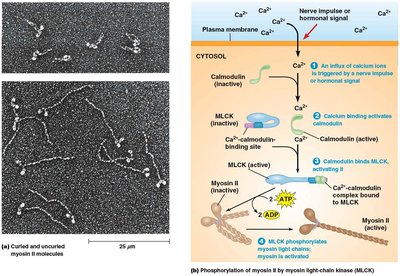
Contraction in smooth muscle is regulated by a cascade involving Ca2+, calmodulin, and myosin light-chain kinase (MLCK).
Increased Ca2+ binds to calmodulin, activating it.
Activated calmodulin activates MLCK, which phosphorylates the regulatory light chain of myosin.
Phosphorylation causes a conformational change in myosin, promoting filament assembly and activation for cross-bridge cycling with actin.
Relaxation occurs when Ca2+ levels fall, MLCK is inactivated, and myosin light-chain phosphatase removes the phosphate group.
Comparison: Calcium Regulation in Skeletal vs. Smooth Muscle
Feature | Skeletal Muscle | Smooth Muscle |
|---|---|---|
Ca2+ binding partner | Troponin C (TnC) | Calmodulin |
Regulatory mechanism | Moves tropomyosin to expose myosin-binding sites on actin | Activates MLCK, which phosphorylates myosin light chain |
Contraction speed | Fast | Slow, sustained |
Additional info: In both muscle types, Ca2+ is essential for contraction, but the molecular targets and regulatory pathways differ.
