 Back
BackThe Cytoskeleton: Structure, Function, and Dynamics of Microtubules, Microfilaments, and Intermediate Filaments
Study Guide - Smart Notes

Cytoskeletal Systems
Overview of the Cytoskeleton
The cytoskeleton is a dynamic network of interconnected protein filaments and tubules that extends throughout the cytosol, providing structural support, facilitating intracellular transport, and enabling cellular movement. It is composed of three main types of protein filaments: microtubules, microfilaments (actin filaments), and intermediate filaments.
Structure and Support: Maintains cell shape and provides mechanical resistance to deformation.
Intracellular Transport: Serves as tracks for the movement of organelles and vesicles.
Contractility and Motility: Enables cell movement and division.
Spatial Organization: Positions organelles and organizes the cytoplasm.
Microtubules
Structure and Properties of Microtubules
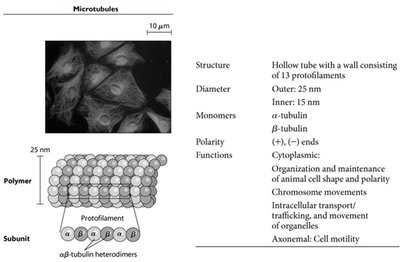
Microtubules are hollow tubes composed of 13 protofilaments made from α- and β-tubulin heterodimers. They have a fixed outer diameter of 25 nm and an inner diameter of 15 nm. Microtubules are polar structures with distinct plus (+) and minus (−) ends, which is critical for their dynamic behavior and cellular functions.
Monomers: α-tubulin and β-tubulin
Polarity: (+) and (−) ends
Functions: Maintenance of cell shape, chromosome movement during mitosis, intracellular transport, and cell motility (e.g., cilia and flagella)

Tubulin Heterodimers and Nucleotide Binding
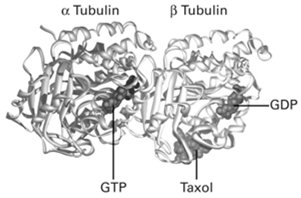
Each tubulin heterodimer consists of one α-tubulin and one β-tubulin subunit. The α-subunit binds GTP permanently, while the β-subunit has GTPase activity and hydrolyzes GTP to GDP after incorporation into the microtubule lattice. This nucleotide state regulates microtubule dynamics and stability.
Microtubule Assembly and Dynamics
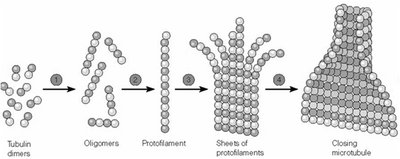
Microtubule assembly occurs in two main phases: nucleation (formation of short oligomers and protofilaments) and elongation (addition of tubulin dimers to both ends). Assembly is reversible and highly dependent on temperature and tubulin concentration. The process is characterized by a critical concentration (Cc) of tubulin dimers, above which polymerization occurs and below which depolymerization dominates.
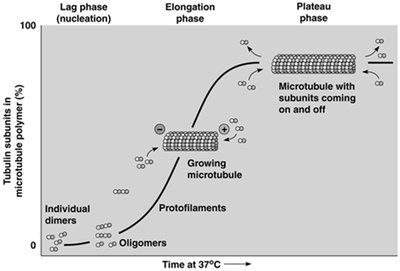
Treadmilling and Dynamic Instability
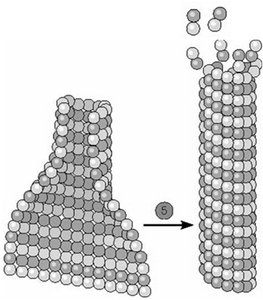
Microtubules exhibit treadmilling when the critical concentration at the plus end is lower than at the minus end, resulting in net addition at the plus end and net loss at the minus end. Dynamic instability refers to the stochastic switching between phases of growth and rapid shrinkage (catastrophe), regulated by the presence of a GTP cap at the plus end. High tubulin-GTP concentration stabilizes the microtubule, while loss of the GTP cap leads to rapid depolymerization.
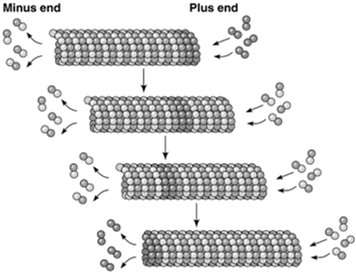
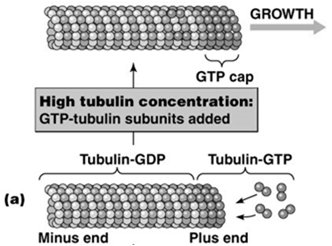
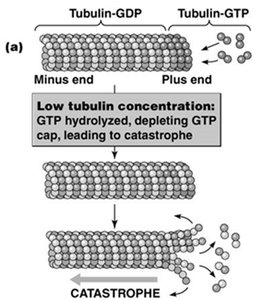

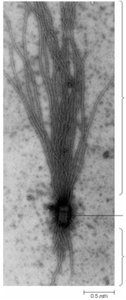
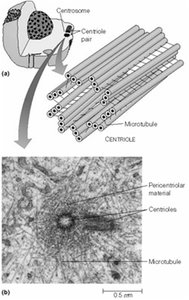
Microtubule-Organizing Centers (MTOCs)
Microtubules originate from specialized structures called microtubule-organizing centers (MTOCs), such as centrosomes and basal bodies. MTOCs serve as nucleation and anchoring sites, with microtubules growing faster from the plus end. Centrosomes contain two centrioles oriented perpendicularly and surrounded by pericentriolar material.
Basal bodies: Found at the base of cilia and flagella
Centrosomes: Main MTOC in animal cells, important for spindle formation during mitosis
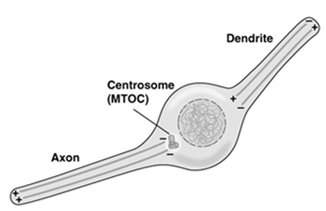
Microtubule-Associated Proteins (MAPs)
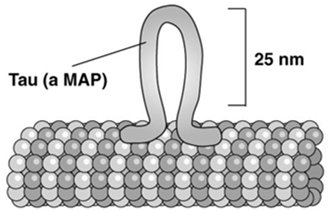
MAPs regulate microtubule stability, organization, and interactions with other cellular structures. Tau is a MAP that promotes tight bundling of microtubules in axons, while MAP2 forms looser bundles in dendrites. Abnormalities in MAPs, such as Tau aggregation, are linked to neurodegenerative diseases (tauopathies).
Microfilaments (Actin Filaments)
Structure and Properties of Microfilaments
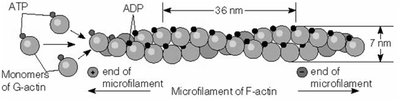
Microfilaments are composed of two intertwined chains of F-actin (filamentous actin) polymers, each 7 nm in diameter. The basic unit is G-actin (globular actin), which binds ATP and assembles into polarized filaments with distinct plus (barbed) and minus (pointed) ends. Microfilaments are essential for muscle contraction, cell locomotion, cytokinesis, and maintenance of cell shape.
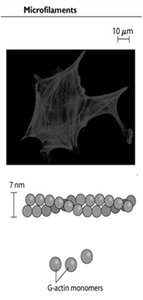

Microfilament Assembly and Polarity
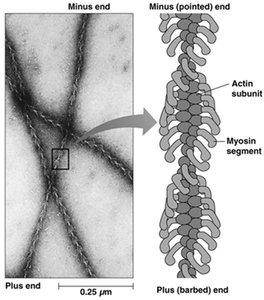
Microfilament assembly is reversible and characterized by a critical concentration of ATP-bound G-actin. The process includes a lag (nucleation) phase, a rapid elongation phase, and a plateau phase. Myosin S1 fragments bind to actin filaments, revealing their polarity: the plus end is the barbed end, and the minus end is the pointed end.
Regulation of Actin Polymerization
Actin polymerization is regulated by several actin-binding proteins:
Arp2/3 complex: Initiates actin branching
Formin: Promotes elongation of straight filaments
Cofilin: Promotes disassembly of actin filaments
Profilin: Acts as a nucleotide-exchange factor, promoting assembly
Capping proteins (e.g., CapZ): Bind filament ends to prevent further addition or loss of subunits
Specialized Actin Structures
Microvilli are actin-based, non-motile projections found on intestinal mucosal cells, increasing surface area for absorption. Other actin-based structures include stress fibers (in adherent cells), the cell cortex (in motile cells), and lamellipodia/filopodia (in crawling cells).
Intermediate Filaments
Structure and Properties of Intermediate Filaments
Intermediate filaments (IFs) are 10–12 nm in diameter, less organized but more stable than microtubules or microfilaments. They are composed of fibrous protein subunits arranged in antiparallel tetramers, resulting in non-polar filaments. IFs are tissue-specific and primarily provide structural support.
No polarity: Antiparallel arrangement of subunits
Tissue specificity: Protein sequence varies by cell type, useful in medical diagnostics
No motor proteins: Mainly structural role
Nuclear Lamins
Nuclear lamins are a class of intermediate filaments that form a structural network underlying the nuclear envelope. Phosphorylation of lamins leads to nuclear envelope breakdown during mitosis.
Summary Table: Comparison of Cytoskeletal Filaments
Property | Microtubules | Microfilaments | Intermediate Filaments |
|---|---|---|---|
Diameter | 25 nm | 7 nm | 10–12 nm |
Monomers | α/β-tubulin | G-actin | Tissue-specific proteins |
Polarity | Yes | Yes | No |
Functions | Cell shape, transport, division, motility | Contraction, movement, shape, cytokinesis | Structural support, nuclear envelope |
