 Back
BackThe Electron Transport Chain and ATP Synthesis in Aerobic Respiration
Study Guide - Smart Notes

Electron Transport Chain (ETC) and ATP Production
Overview of the Electron Transport Chain
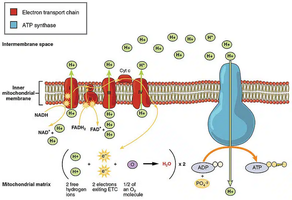
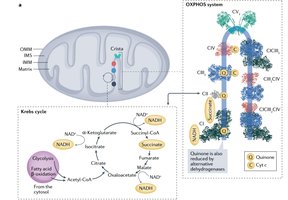
The Electron Transport Chain (ETC) is a series of protein complexes and small molecules embedded in the inner mitochondrial membrane. It is the final stage of aerobic respiration, where most of the energy from glucose catabolism is harvested. The ETC transfers electrons from reduced coenzymes (NADH and FADH2) to molecular oxygen, forming water and generating a proton gradient that drives ATP synthesis.
Location: Inner mitochondrial membrane (in eukaryotes); plasma membrane (in prokaryotes).
Key Components: Four main respiratory complexes (I-IV), coenzyme Q (ubiquinone), and cytochrome c.
Ultimate Electron Acceptor: Oxygen (O2), which is reduced to water (H2O).
Energy Yield: Most ATP from glucose is produced during this stage.
Electron Flow and Respiratory Complexes
Electrons from NADH and FADH2 are transferred through a series of carriers with increasing reduction potentials, making the process spontaneous and highly exergonic. The main complexes are:
Complex I (NADH dehydrogenase): Accepts electrons from NADH.
Complex II (Succinate dehydrogenase): Accepts electrons from FADH2.
Complex III (Cytochrome bc1): Transfers electrons from coenzyme Q to cytochrome c.
Complex IV (Cytochrome c oxidase): Transfers electrons to O2, forming H2O.
Coenzyme Q and cytochrome c act as mobile electron carriers between complexes. The complexes are organized into respirasomes (supercomplexes) to enhance efficiency.
Proton Pumping and the Electrochemical Gradient
As electrons flow through complexes I, III, and IV, protons (H+) are pumped from the mitochondrial matrix into the intermembrane space. This creates an electrochemical proton gradient (proton motive force, pmf) across the inner membrane, which stores potential energy.
Proton Motive Force (pmf): The combined effect of a proton concentration gradient and an electrical potential across the membrane.
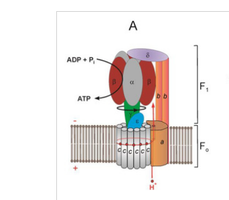
Energy Coupling: The pmf is used to drive ATP synthesis as protons flow back into the matrix through ATP synthase.
ATP Synthesis: The Role of ATP Synthase
ATP synthase is a large enzyme complex composed of two main parts: F0 (membrane-embedded proton channel) and F1 (catalytic ATP synthesis domain). The flow of protons through F0 drives conformational changes in F1, catalyzing the formation of ATP from ADP and inorganic phosphate (Pi).
Oxidative Phosphorylation (OXPHOS): The process of ATP synthesis powered by the transfer of electrons to oxygen and the resulting proton gradient.
ATP Yield: Each NADH yields about 3 ATP; each FADH2 yields about 2 ATP (theoretical maximum).
Summary Equation for Aerobic Respiration
The complete oxidation of one glucose molecule in aerobic respiration can be summarized as:
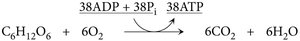
Note: The actual ATP yield is often lower (about 30–32 ATP) due to losses from proton leakage, use of the gradient for other transport processes, and variability in shuttle systems.
Regulation and Inhibition of the ETC
The rate of electron transport and ATP synthesis is tightly regulated by the availability of ADP (respiratory control), substrate supply, and the assembly of ETC complexes. Inhibitors and uncouplers can disrupt these processes:
Inhibitors: Block electron flow (e.g., cyanide, carbon monoxide, rotenone, antimycin C, oligomycin), reducing ATP and O2 consumption.
Uncouplers: Dissipate the proton gradient (e.g., 2,4-dinitrophenol, FCCP), increasing O2 consumption but decreasing ATP production and generating heat.
Physiological Roles and Regulation of the ETC
The ETC is regulated by factors such as nutrient availability, NAD+ import, oxygen levels, and the ADP/ATP ratio. It plays roles beyond ATP synthesis, including cell death, reactive oxygen species (ROS) production, adaptation to hypoxia, tumor growth, exercise performance, fertility, inflammation, and thermogenesis.

Practice and Application
Understand how inhibitors and uncouplers affect the ETC and ATP synthesis.
Be able to calculate theoretical and actual ATP yields from glucose oxidation.
Explain the chemiosmotic model and the importance of the proton gradient in energy coupling.
Additional info:
The analogy of the ETC as waterwheels along a river helps visualize how electron flow drives proton pumping and ATP synthesis.
Proton mass scaling is a thought experiment to illustrate the density of subatomic particles, not directly relevant to ETC function.
