 Back
BackThe Endomembrane System: Protein Targeting, ER, and Golgi Function
Study Guide - Smart Notes

The Endomembrane System and Protein Targeting
Overview of the Endomembrane System
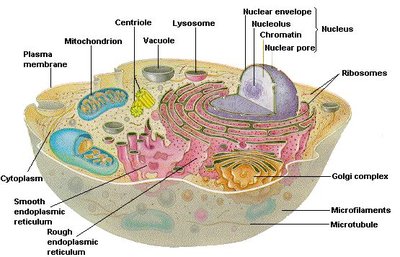
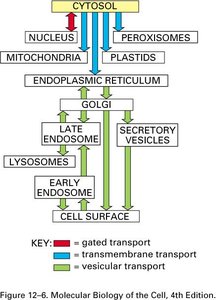
The endomembrane system is a network of organelles and membranes within eukaryotic cells that work together to modify, package, and transport lipids and proteins. Key components include the endoplasmic reticulum (ER), Golgi complex, lysosomes, endosomes, and vesicles. This system is essential for maintaining cellular organization and directing the flow of materials within the cell.
Endoplasmic Reticulum (ER): Site of protein and lipid synthesis; divided into rough (RER) and smooth (SER) regions.
Golgi Complex: Modifies, sorts, and packages proteins and lipids for delivery to various destinations.
Lysosomes and Endosomes: Involved in degradation and recycling of cellular materials.
Vesicles: Transport materials between organelles and to/from the plasma membrane.
Intracellular Protein Targeting and Sorting
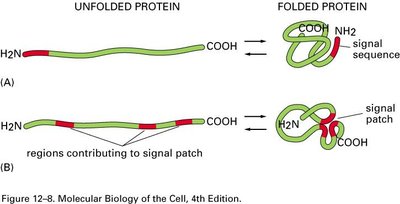
Proteins are directed to specific cellular compartments by targeting sequences or "zip codes" within their amino acid sequence. These signals ensure proteins reach their correct destinations, such as the ER, nucleus, mitochondria, or peroxisomes.
Signal Sequence: Directs import into the ER.
Nuclear Localization Signal (NLS): Directs import into the nucleus.
Nuclear Export Signal (NES): Directs export from the nucleus.
Mitochondrial Targeting Sequence (MTS): Directs import into mitochondria.
Peroxisomal Targeting Signal (PTS): Directs import into peroxisomes.
Endoplasmic Reticulum (ER): Structure and Function
Types and Functions of the ER
The ER is a continuous membrane system with two main types: rough ER (RER), studded with ribosomes, and smooth ER (SER), which lacks ribosomes. The RER is primarily involved in protein synthesis and processing, while the SER is associated with lipid synthesis and detoxification.
Rough ER: Synthesis of secretory, lysosomal, and membrane proteins.
Smooth ER: Synthesis of lipids, metabolism of carbohydrates, and detoxification of drugs.
Specialized Cells: Leydig cells have extensive SER for steroid hormone synthesis.

Membrane Synthesis and Lipid Asymmetry
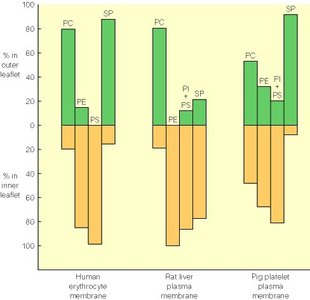
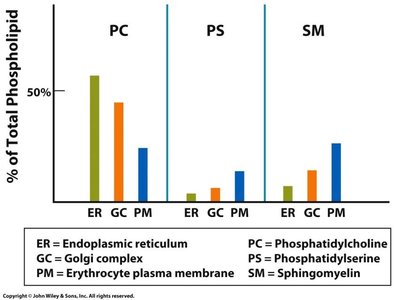
Membrane lipids are synthesized in the ER and distributed to other organelles. The composition and asymmetry of phospholipids in membranes are tightly regulated, affecting membrane function and cell signaling.
Phospholipid Distribution: Different membranes have distinct phospholipid compositions.
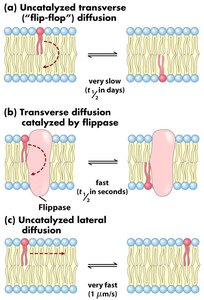
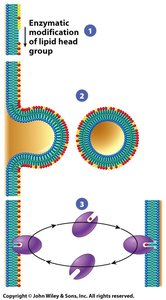
Asymmetry: The inner and outer leaflets of the bilayer have different lipid compositions, maintained by enzymes such as flippases.
Protein Synthesis and Translocation into the ER
Co-Translational Translocation
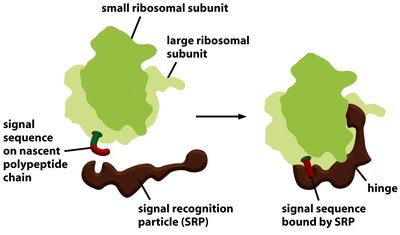
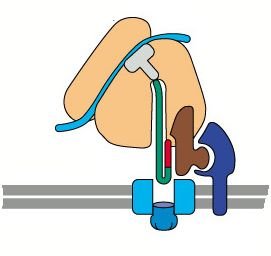
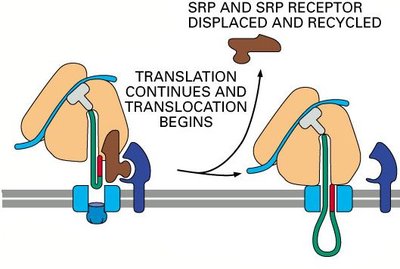
Proteins destined for secretion or for organelles of the endomembrane system are synthesized by ribosomes bound to the ER membrane. The process of moving these proteins into the ER is called co-translational translocation.
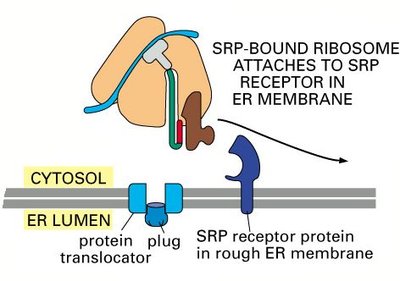
Signal Recognition Particle (SRP): Binds to the signal sequence on the nascent polypeptide and pauses translation.
SRP Receptor: Located on the ER membrane, it binds the SRP-ribosome complex.
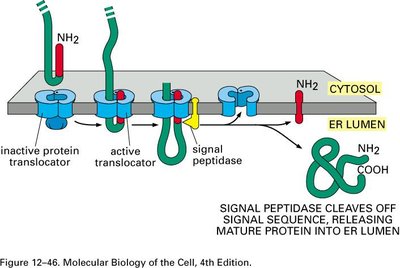
Translocon (Sec61 complex): Channel through which the polypeptide enters the ER lumen.
Signal Peptidase: Cleaves the signal sequence, releasing the mature protein into the ER lumen.
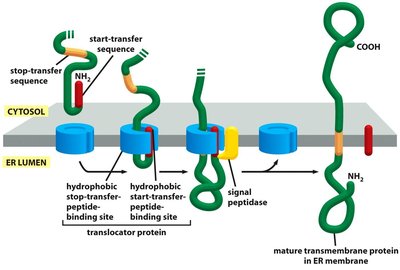
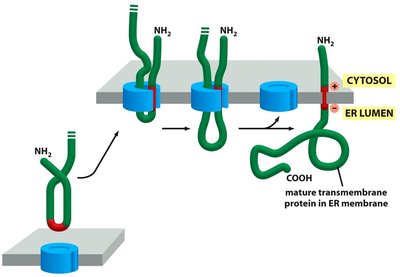
Insertion of Membrane Proteins
Integral membrane proteins are inserted into the ER membrane during translation. Start-transfer and stop-transfer sequences determine the orientation and number of membrane-spanning domains.
Start-Transfer Sequence: Initiates translocation into the membrane.
Stop-Transfer Sequence: Halts translocation, anchoring the protein in the membrane.
Multiple Membrane-Spanning Domains: Achieved by alternating start- and stop-transfer sequences.
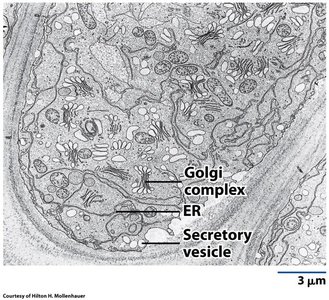
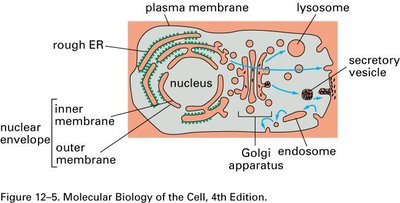
Golgi Complex: Structure and Function
Organization and Regional Specialization
The Golgi complex is a stack of flattened cisternae with distinct functional regions: the cis face (entry, facing the ER), medial cisternae, and trans face (exit, facing the plasma membrane). Each region contains specific enzymes and performs unique processing steps.
Cis Golgi Network (CGN): Sorts proteins for return to the ER or forward transport.
Trans Golgi Network (TGN): Sorts proteins for delivery to the plasma membrane, lysosomes, or secretory vesicles.
Regional Differences: Enzyme composition and membrane properties vary across the stack.
Glycosylation and Protein Modification
Proteins and lipids are modified in the Golgi, most notably by glycosylation—the addition and processing of carbohydrate groups. Glycosyltransferases in each cisterna add specific sugars in a defined sequence, resulting in complex oligosaccharide structures.
N-linked Glycosylation: Addition of oligosaccharides to asparagine residues, initiated in the ER and completed in the Golgi.
O-linked Glycosylation: Addition of sugars to serine or threonine residues, primarily in the Golgi.
Models of Transport Through the Golgi
Two main models describe how cargo moves through the Golgi:
Vesicular Transport Model: Cargo is shuttled between static cisternae via vesicles.
Cisternal Maturation Model: Cisternae themselves mature and move from cis to trans, carrying cargo with them.
Current View: A combination of cisternal maturation with vesicle-mediated retrograde transport for resident enzymes.
Vesicular Transport and Membrane Trafficking
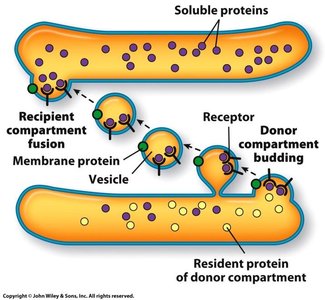
Vesicle Formation and Fusion
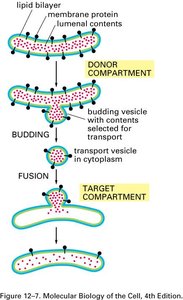
Vesicles transport proteins and lipids between organelles. The process involves budding from a donor compartment, transport, and fusion with a target compartment. Protein coats (COPI, COPII, clathrin) help shape vesicles and select cargo.
COPII-coated Vesicles: Mediate anterograde transport from ER to Golgi.
COPI-coated Vesicles: Mediate retrograde transport from Golgi to ER and within Golgi.
Clathrin-coated Vesicles: Mediate transport from TGN to endosomes/lysosomes and in endocytosis.

Targeting and Fusion of Vesicles
Vesicle targeting and fusion are highly specific, involving Rab proteins (tethering), SNARE proteins (docking and fusion), and recognition of membrane markers. This ensures cargo is delivered to the correct compartment.
Rab Proteins: Guide vesicles to the correct membrane by recruiting tethering factors.
SNARE Proteins: v-SNAREs on vesicles interact with t-SNAREs on target membranes to mediate fusion.
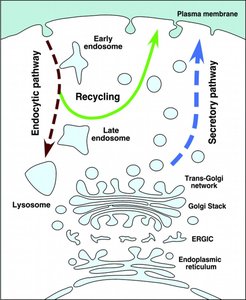
Endocytosis and Exocytosis
Endocytic and Secretory Pathways
Endocytosis brings materials into the cell via vesicles, while exocytosis releases materials to the extracellular space. These pathways are interconnected and essential for nutrient uptake, signaling, and membrane turnover.
Early Endosomes: Sort internalized material and recycle receptors to the plasma membrane.
Late Endosomes: Mature into lysosomes for degradation of cargo.
Secretory Vesicles: Store and release proteins and other molecules via exocytosis.
Protein Import into Mitochondria and Other Organelles
Protein Translocation into Mitochondria
Proteins destined for mitochondria are synthesized in the cytosol and imported post-translationally. They must remain unfolded during import and are recognized by specific targeting sequences.
TOM Complex: Translocase of the Outer Membrane, mediates initial import into mitochondria.
TIM Complex: Translocase of the Inner Membrane, mediates import into the mitochondrial matrix or insertion into the inner membrane.
Chaperones: Assist in maintaining proteins in an unfolded state for import.
Summary Table: Major Protein Targeting Signals
Destination | Targeting Signal | Function |
|---|---|---|
ER | Signal sequence | Import into ER |
Nucleus | NLS (Nuclear Localization Signal) | Import into nucleus |
Nucleus (export) | NES (Nuclear Export Signal) | Export from nucleus |
Mitochondria | MTS (Mitochondrial Targeting Sequence) | Import into mitochondria |
Peroxisome | PTS (Peroxisomal Targeting Signal) | Import into peroxisome |
