 Back
BackThe Macromolecules of the Cell: Carbohydrates and Bioenergetics
Study Guide - Smart Notes

The Macromolecules of the Cell
Monosaccharides: Structure and Classification
Monosaccharides are the simplest form of carbohydrates, consisting of a single sugar unit. They serve as fundamental building blocks for more complex carbohydrates and play crucial roles in cellular metabolism.
Definition: Monosaccharides are single carbohydrate molecules with the general formula C(H2O)n.
Classification: Two broad classes exist: aldosugars (with an aldehyde group at carbon 1) and ketosugars (with a ketone group at carbon 2).
Example: Glucose is an aldosugar, while fructose is a ketosugar.
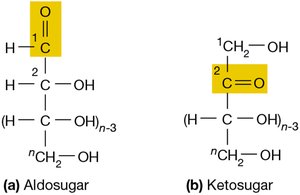
Additional info: The functional group determines the chemical reactivity and biological function of the sugar.
Monosaccharide Ring Formation and Isomerism
Monosaccharides can exist in both linear and ring forms, with the ring structure predominating in aqueous solutions. The ring formation involves a reaction between a carbonyl group and a hydroxyl group within the molecule.
Pyranose Ring: In glucose, the oxygen on carbon 5 forms a bond with carbon 1, creating a six-membered ring (pyranose).
Equilibrium: Sugars exist in equilibrium between linear and ring forms.
Projections: The Fischer projection shows the linear form, while the Haworth projection depicts the ring form.
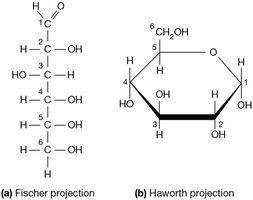
Additional info: The ring form is more stable and biologically relevant in most cellular contexts.
Alpha and Beta Anomers of Glucose
When glucose cyclizes, it forms two distinct anomers: alpha (α) and beta (β), depending on the orientation of the hydroxyl group at carbon 1.
Alpha (α) form: The hydroxyl group on carbon 1 points downward.
Beta (β) form: The hydroxyl group on carbon 1 points upward.
Biological significance: α-D-glucose is the repeating unit of starch and glycogen, while β-D-glucose is the repeating unit of cellulose.
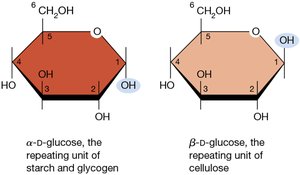
Additional info: The difference in anomeric configuration leads to distinct structural and functional properties in polysaccharides.
Disaccharides: Formation and Types
Disaccharides are formed by condensation reactions between two monosaccharides, resulting in a glycosidic bond. They serve as important energy sources and structural components.
Common disaccharides: Maltose (glucose + glucose), Lactose (glucose + galactose), Sucrose (glucose + fructose).
Glycosidic bond: The bond formed between the anomeric carbon of one sugar and a hydroxyl group of another.
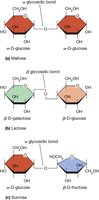
Additional info: The type of glycosidic bond (α or β) affects digestibility and function.
Polysaccharides: Structure and Function
Polysaccharides are long chains of monosaccharide units linked by glycosidic bonds. They serve both structural and storage roles in cells.
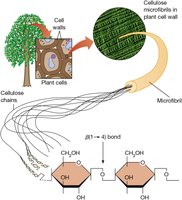
Structural polysaccharides: Cellulose is composed of β-D-glucose units joined by β(1→4) linkages, forming rigid microfibrils in plant cell walls.
Storage polysaccharides: Starch (plants) and glycogen (animals, bacteria) are polymers of α-D-glucose, primarily linked by α(1→4) bonds with occasional α(1→6) branches.
Additional info: The branching in glycogen allows rapid mobilization of glucose during metabolic demand.
Bioenergetics: The Flow of Energy in the Cell
Redox State of Carbon and Energy Availability
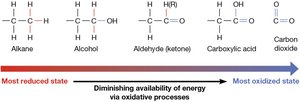
The energy content of carbon compounds is determined by their redox state. More reduced carbon atoms possess higher energy, which is released during oxidation.
Oxidation of glucose: Eukaryotic cells oxidize glucose to carbon dioxide, releasing energy.
Reduction in photosynthesis: Plants use sunlight to reduce carbon dioxide to glucose.
Equation for glucose oxidation:
Equation for photosynthesis:
Additional info: The flow of energy in cells is tightly linked to the oxidation and reduction of carbon compounds.
ATP: The Universal Energy Coupler
ATP (adenosine triphosphate) is the primary energy currency of the cell. Its hydrolysis releases free energy, which is used to drive cellular reactions.
Phosphoanhydride bonds: These are high-energy bonds; hydrolysis releases significant free energy.
Hydrolysis reaction:
Pi: Inorganic phosphate ()
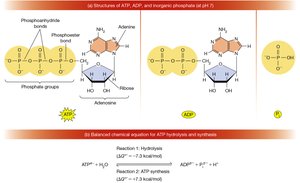
Additional info: ATP hydrolysis is highly exergonic due to charge repulsion, resonance stabilization, and increased entropy.
Why ATP Hydrolysis is Exergonic
The hydrolysis of ATP to ADP and Pi is exergonic for three main reasons:
Charge repulsion: Adjacent negatively charged phosphate groups repel each other.
Resonance stabilization: Both ADP and Pi have greater electron delocalization than ATP.
Increased entropy and solubility: The products are more disordered and soluble.
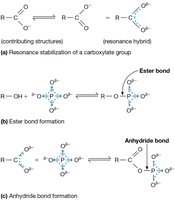
Additional info: Resonance stabilization lowers the energy of the products, making the reaction favorable.
ATP as an Intermediate in Cellular Energy Transfer
ATP occupies an intermediate position among phosphorylated compounds, allowing it to act as both a phosphate donor and acceptor in metabolic reactions.
Energy hierarchy: Compounds exist both above and below ATP/ADP in terms of phosphate transfer potential.
Biological significance: This enables ATP to efficiently couple catabolic and anabolic processes.
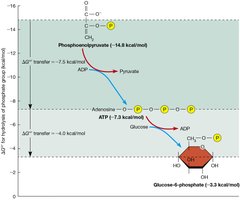
Additional info: The transfer of phosphate groups is central to cellular energy management.
ATP Synthesis and Utilization in Cells
The energy released from nutrient oxidation is used to synthesize ATP, which is then utilized to perform various types of cellular work.
Catabolism: Oxidation of glucose and other nutrients releases energy for ATP synthesis.
Cellular work: ATP is used for synthetic, concentration, electrical, mechanical, and bioluminescent work.
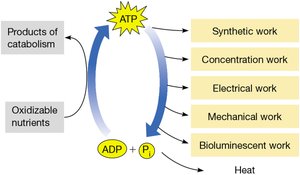
Additional info: The ATP cycle is fundamental to all living cells, linking energy release to energy utilization.
