 Back
BackThe Macromolecules of the Cell: Proteins, Nucleic Acids, Polysaccharides, and Lipids
Study Guide - Smart Notes

The Macromolecules of the Cell
Introduction
Cells are composed of four major classes of biological macromolecules: proteins, nucleic acids, polysaccharides, and lipids. These macromolecules are essential for structure, function, and information storage in all living organisms. Most are polymers, assembled from small monomeric subunits through condensation (dehydration) reactions.
Proteins
Overview and Functions
Proteins are the most versatile macromolecules in the cell, performing a wide range of functions:
Enzymes: Catalyze biochemical reactions.
Structural proteins: Provide support and shape.
Motility proteins: Enable movement and contraction.
Regulatory proteins: Control cellular processes.
Transport proteins: Move substances across membranes.
Signaling proteins: Mediate communication between cells.
Receptor proteins: Detect and respond to external signals.
Defensive proteins: Protect against disease.
Storage proteins: Store amino acids for later use.
Amino Acids: The Monomers of Proteins
Proteins are polymers of amino acids (AAs). There are 20 standard amino acids, each with a central (α) carbon, an amino group, a carboxyl group, a hydrogen atom, and a unique side chain (R group).
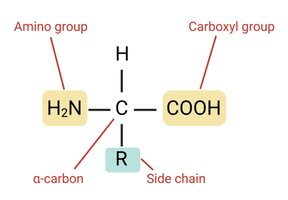
Chirality: All amino acids except glycine are chiral, existing as L- and D- enantiomers. Only L-amino acids are incorporated into proteins in nature.
R Groups: Determine the chemical properties of each amino acid (nonpolar, polar, acidic, or basic).
Chirality and Biological Significance
Chirality is a fundamental property of amino acids, affecting protein structure and function. The existence of only L-amino acids in proteins is considered a 'frozen accident' of evolution.
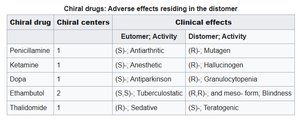
Example: Many drugs are chiral, and their biological activity can differ dramatically between enantiomers.
Peptide Bond Formation
Amino acids are linked by peptide bonds formed via condensation reactions, resulting in the release of water. This process occurs on ribosomes during translation.
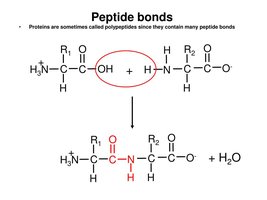
Directionality: Polypeptides have an N-terminus (amino end) and a C-terminus (carboxyl end).
Protein Structure: Four Levels of Organization
Protein structure is organized into four hierarchical levels:
Primary structure: Linear sequence of amino acids.
Secondary structure: Local folding patterns (α-helix, β-sheet) stabilized by hydrogen bonds.
Tertiary structure: Overall 3D shape formed by interactions among R groups.
Quaternary structure: Association of multiple polypeptide subunits.
Primary Structure
The unique sequence of amino acids in a polypeptide determines its final structure and function. Even a single amino acid change can have dramatic effects (e.g., sickle cell anemia).
Secondary Structure
Stabilized by hydrogen bonds between backbone atoms, secondary structures include:
α-Helix: Right-handed coil with 3.6 residues per turn.
β-Sheet: Extended strands connected by hydrogen bonds, can be parallel or antiparallel.
Tertiary Structure
Determined by interactions among R groups, including hydrophobic interactions, hydrogen bonds, ionic bonds, van der Waals forces, and disulfide bridges.
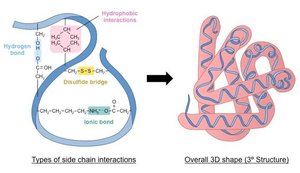
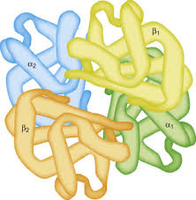
Quaternary Structure
Describes the assembly of multiple polypeptide chains into a functional protein complex (e.g., hemoglobin is a heterotetramer).
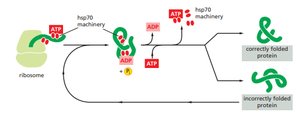
Protein Folding and Chaperones
Protein folding is guided by the amino acid sequence but often requires assistance from molecular chaperones to prevent misfolding and aggregation.
Misfolded proteins are targeted for degradation by the proteasome.
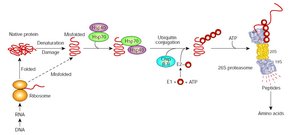
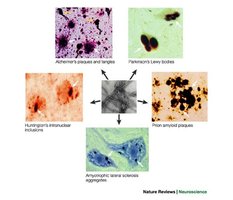
Failure to manage misfolded proteins can lead to diseases such as Alzheimer's, Parkinson's, and prion diseases.
Nucleic Acids
Overview and Functions
Nucleic acids (DNA and RNA) store, transmit, and express genetic information. They are linear polymers of nucleotides.
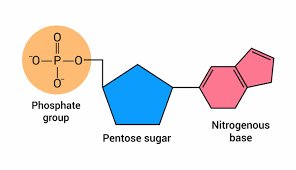
Nucleotide Structure
Each nucleotide consists of a phosphate group, a five-carbon sugar (ribose or deoxyribose), and a nitrogenous base (purine or pyrimidine).
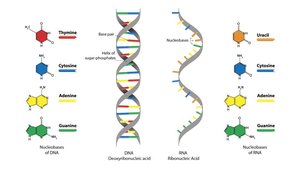
Purines: Adenine (A), Guanine (G)
Pyrimidines: Cytosine (C), Thymine (T, in DNA), Uracil (U, in RNA)
DNA vs RNA
DNA: Double-stranded, deoxyribose sugar, bases A, T, G, C; stores genetic information.
RNA: Single-stranded, ribose sugar, bases A, U, G, C; involved in protein synthesis and regulation.
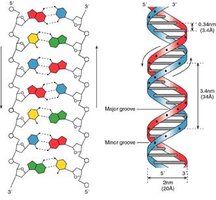
Base Pairing and Double Helix
DNA strands are antiparallel and held together by complementary base pairing (A-T, G-C) via hydrogen bonds, forming a double helix.
Types of RNA
mRNA: Messenger RNA, template for protein synthesis.
tRNA: Transfer RNA, brings amino acids to ribosome.
rRNA: Ribosomal RNA, structural and catalytic component of ribosomes.
Other RNAs: Regulatory and catalytic roles.
Polysaccharides
Overview and Functions
Polysaccharides are long chains of monosaccharides, serving as energy storage (starch, glycogen) or structural components (cellulose).
Monosaccharides and Disaccharides
Monosaccharides are simple sugars (e.g., glucose), which can form disaccharides (e.g., maltose, lactose, sucrose) via glycosidic bonds.
Storage Polysaccharides
Starch: Plant storage, composed of amylose and amylopectin (α-glucose units).
Glycogen: Animal storage, highly branched (α-glucose units).
Structural Polysaccharides
Cellulose: Plant cell walls, β-glucose units, forms straight fibers.
Alpha vs Beta Linkages
The type of glycosidic linkage (α or β) determines the structure and digestibility of polysaccharides.

Lipids
Overview and Functions
Lipids are hydrophobic macromolecules important for energy storage, membrane structure, and signaling. Unlike other macromolecules, they are not true polymers.
Types and Functions
Triglycerides: Energy storage molecules composed of glycerol and three fatty acids.
Phospholipids: Major component of plasma membranes, amphipathic (hydrophilic head, hydrophobic tails).
Steroids: Cholesterol and steroid hormones (e.g., testosterone, estrogen).
Fatty Acids
Fatty acids vary in length and degree of saturation:
Saturated fatty acids: No double bonds, straight chains, solid at room temperature.
Unsaturated fatty acids: One or more double bonds, bent chains, liquid at room temperature.
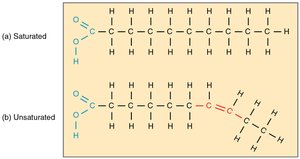
Phospholipids and Membranes
Phospholipids spontaneously form bilayers in aqueous environments, creating the fundamental structure of cell membranes.
Lipoproteins: LDL vs HDL
Lipoproteins transport lipids in the bloodstream:
LDL (Low-Density Lipoprotein): Delivers cholesterol to tissues; high levels increase atherosclerosis risk.
HDL (High-Density Lipoprotein): Removes excess cholesterol from tissues; high levels are protective.
Summary Table: Macromolecules of the Cell
Macromolecule | Monomer | Bond Type | Main Functions |
|---|---|---|---|
Proteins | Amino acids | Peptide bond | Catalysis, structure, transport, signaling |
Nucleic Acids | Nucleotides | Phosphodiester bond | Information storage, transmission |
Polysaccharides | Monosaccharides | Glycosidic bond | Energy storage, structure |
Lipids | Fatty acids, glycerol | Ester bond | Energy storage, membranes, signaling |
Key Equations
Peptide bond formation:
Phosphodiester bond in nucleic acids:
Glycosidic bond in polysaccharides:
Practice Questions
What type of bond links amino acids in a protein? Peptide bond
Which level of protein structure is characterized by the sequence of amino acids? Primary
What is the monomer unit of polysaccharides? Monosaccharide
Which base is found in DNA but not in RNA? Thymine
What is the primary role of LDL in the body? Deliver cholesterol from the liver to peripheral tissues
