 Back
BackThe Macromolecules of the Cell: Structure, Function, and Organization
Study Guide - Smart Notes

The Macromolecules of the Cell
Overview of Cellular Macromolecules
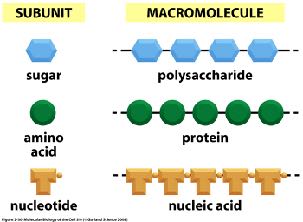
Cells are composed of four major families of macromolecules: proteins, nucleic acids, polysaccharides, and lipids. These macromolecules are essential for cellular structure, function, and metabolism. Each is built from smaller subunits, and their assembly and properties are fundamental to cell biology.
Proteins: Polymers of amino acids, responsible for catalysis, structure, regulation, transport, signaling, defense, storage, and movement.
Nucleic acids: Polymers of nucleotides, store and transmit genetic information (DNA and RNA).
Polysaccharides: Polymers of sugars, serve structural and energy storage roles.
Lipids: Built from fatty acids, important for membranes, energy storage, and signaling.
Proteins
Protein Structure and Function
Proteins are the most abundant macromolecules in cells, accounting for over 50% of dry mass. They are involved in nearly every cellular process, including photosynthesis, communication, and gene regulation. Proteins are classified into nine major functional classes:
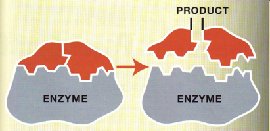
Enzymes: Catalysts for biochemical reactions.
Structural proteins: Provide support and shape (e.g., keratin, collagen).
Motility proteins: Enable movement and contraction (e.g., actin, myosin).
Regulatory proteins: Control cellular functions (e.g., transcription factors).
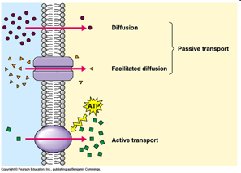
Transport proteins: Move substances across membranes (e.g., Na+/K+ ATPase).
Signaling proteins: Mediate cell communication (e.g., insulin).
Receptor proteins: Respond to environmental stimuli (e.g., growth factor receptors).
Defensive proteins: Protect against disease (e.g., antibodies).
Storage proteins: Store amino acids and ions (e.g., ovalbumin in eggs).
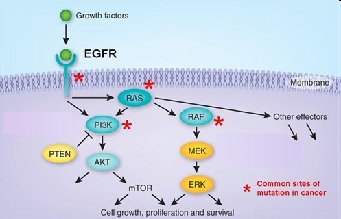
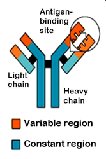


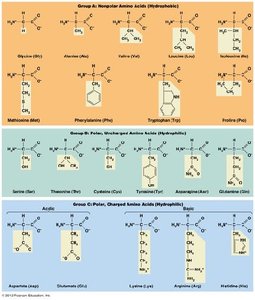
Amino Acids: The Monomers of Proteins
Proteins are linear polymers of amino acids. Only 20 amino acids are used in protein synthesis, each with a central α-carbon, an amino group, a carboxyl group, a hydrogen atom, and a unique side chain (R group). The properties of amino acids depend on their R groups, which can be nonpolar (hydrophobic), polar, acidic (negatively charged), or basic (positively charged).
Structure: Amino acids are chiral, existing as L- and D- forms; only L-amino acids are found in proteins.
Classification: Nine amino acids are hydrophobic; eleven are hydrophilic.
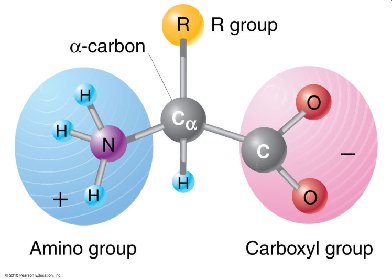
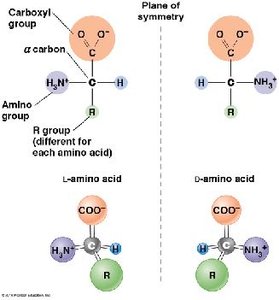
Polypeptide Formation and Protein Structure
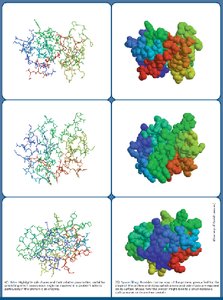
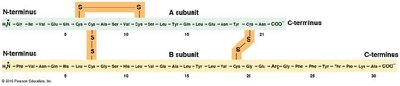
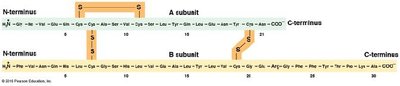
Amino acids are linked by peptide bonds formed via condensation reactions. Polypeptides have directionality, with an N-terminus (amino end) and a C-terminus (carboxyl end). A polypeptide becomes a protein when it folds into a stable, biologically active three-dimensional shape (conformation).
Monomeric proteins: Single polypeptide chain.
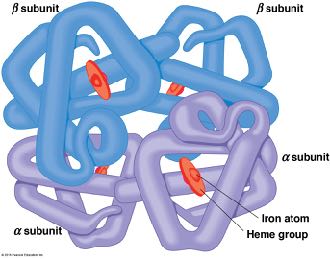
Multimeric proteins: Two or more polypeptide chains (e.g., hemoglobin is a tetramer).

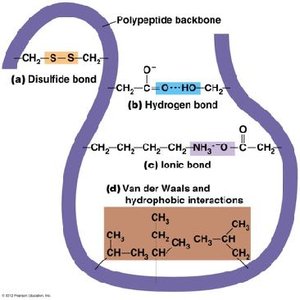
Bonds and Interactions in Protein Folding
Protein folding and stability depend on both covalent and noncovalent interactions:
Disulfide bonds: Covalent bonds between cysteine residues, stabilize protein structure.
Hydrogen bonds: Form between backbone and side chains, contribute to secondary and tertiary structure.
Ionic bonds: Electrostatic interactions between charged R groups.
Van der Waals forces: Weak interactions between nonpolar regions.
Hydrophobic interactions: Nonpolar side chains cluster away from water.
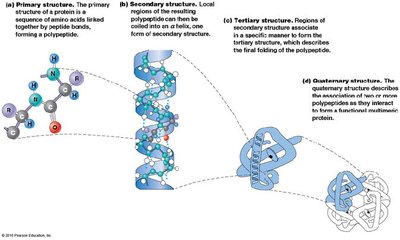
Levels of Protein Structure
Protein structure is described in four hierarchical levels:
Primary structure: Linear sequence of amino acids.
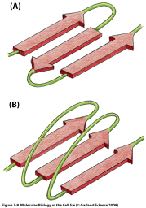
Secondary structure: Local folding into α helices and β sheets, stabilized by hydrogen bonds.
Tertiary structure: Overall three-dimensional conformation, determined by interactions among R groups.
Quaternary structure: Association of multiple polypeptides into a functional unit.
Nucleic Acids
Structure and Function of Nucleic Acids
Nucleic acids are informational macromolecules that store, transmit, and express genetic information. The two main types are DNA (deoxyribonucleic acid) and RNA (ribonucleic acid). DNA serves as the genetic repository, while RNA is involved in gene expression.
Monomers: Nucleotides, each consisting of a five-carbon sugar, a phosphate group, and a nitrogenous base (purine or pyrimidine).
Purines: Adenine (A), Guanine (G).
Pyrimidines: Thymine (T), Cytosine (C), Uracil (U in RNA).
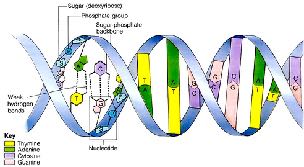
Nucleic Acid Polymerization and Structure
Nucleic acids are linear polymers of nucleotides linked by 3ʹ,5ʹ phosphodiester bonds. DNA is double-stranded, forming a right-handed helix with antiparallel strands, while RNA is usually single-stranded but can form local base-paired regions.
Complementary base pairing: A-T (DNA), A-U (RNA), G-C.
Directionality: 5ʹ to 3ʹ.
Polysaccharides
Structure and Function of Polysaccharides
Polysaccharides are long-chain polymers of sugars, serving structural and energy storage roles. The repeating units are monosaccharides, which can be classified by the number of carbon atoms (triose, tetrose, pentose, hexose, heptose).
Glucose: Most common monosaccharide, exists in linear and ring forms (α and β).
Disaccharides: Two monosaccharides linked by glycosidic bonds (e.g., maltose, lactose, sucrose).
Storage polysaccharides: Starch (plants), glycogen (animals, bacteria).
Structural polysaccharides: Cellulose (plants), chitin (fungi, insects), bacterial cell wall polysaccharides.


Lipids
Structure and Function of Lipids
Lipids are hydrophobic macromolecules important for membrane structure, energy storage, and signaling. Unlike other macromolecules, lipids are not formed by linear polymerization but by condensation reactions.
Fatty acids: Long hydrocarbon chains with a carboxyl group; can be saturated or unsaturated.
Triacylglycerols: Glycerol with three fatty acids, main storage form of energy.
Phospholipids: Amphipathic molecules, key components of cell membranes.
Glycolipids: Lipids with carbohydrate groups, important for cell recognition.
Steroids: Four-ringed structures, include cholesterol and hormones.
Terpenes: Isoprene-based compounds, include vitamins and pigments.

Comparison of Macromolecules
Summary Table: Macromolecule Subunits and Functions
Macromolecule | Subunit | Function |
|---|---|---|
Protein | Amino acid | Catalysis, structure, regulation, transport, defense, storage, movement |
Nucleic acid | Nucleotide | Genetic information storage and expression |
Polysaccharide | Sugar (monosaccharide) | Structure, energy storage, recognition |
Lipid | Fatty acid (not always polymeric) | Membrane structure, energy storage, signaling |

Concept Checks
Macromolecule Similarities and Differences
All macromolecules are essential for cell structure and function.
Proteins, nucleic acids, and polysaccharides are linear polymers; lipids are not.
Each macromolecule has unique monomers and assembly mechanisms.
Functions range from catalysis and information storage to energy storage and membrane structure.
Additional info:
Protein folding is assisted by molecular chaperones for some complex proteins.
Essential fatty acids must be obtained from the diet.
Cellulose and chitin are not digestible by mammals without microbial assistance.
