 Back
BackCH 8 - Transport Across Membranes: Overcoming the Permeability Barrier
Study Guide - Smart Notes

Transport Across Membranes: Overcoming the Permeability Barrier
Overview of Membrane Transport
Cell membranes are selectively permeable, allowing certain molecules and ions to pass while restricting others. The proper functioning of cells depends on overcoming the permeability barrier to maintain homeostasis and regulate the internal environment.
Selective permeability: Membranes permit the passage of specific substances while blocking others.
Homeostasis: Cells actively regulate the movement of solutes to maintain concentrations different from their surroundings.
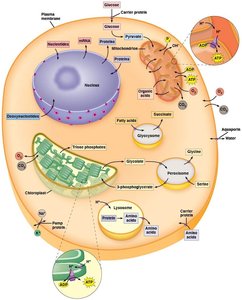
Mechanisms of Solute Transport Across Membranes
Solutes cross membranes via three primary mechanisms, each with distinct properties and energy requirements.
Simple diffusion: Unassisted movement of solutes down their concentration gradient.
Facilitated diffusion: Protein-mediated movement down the gradient, requiring no energy.
Active transport: Protein-mediated movement against the gradient, requiring energy (usually ATP).
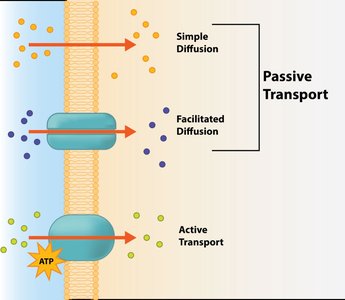
Transport Proteins
Integral membrane proteins play a crucial role in solute transport, recognizing and assisting specific molecules across the membrane.
Facilitated diffusion: Transport proteins move solutes down their concentration gradient without energy input.
Active transport: Transport proteins move solutes against their gradient, requiring energy from ATP hydrolysis (primary) or coupled transport (secondary).
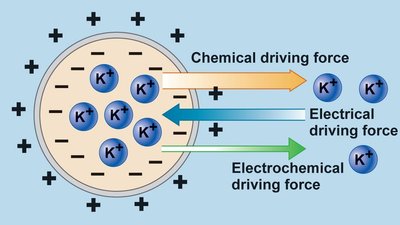
Thermodynamics of Solute Movement
The movement of solutes is governed by their concentration gradients and, for ions, by electrochemical gradients.
Exergonic movement: Down the gradient (negative ΔG), as in simple and facilitated diffusion.
Endergonic movement: Up the gradient (positive ΔG), as in active transport.
Electrochemical potential: The combined effect of concentration and charge gradients determines ion movement.
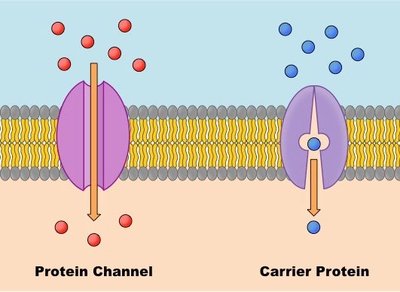
Types of Transport Proteins
Transport proteins are classified as channel proteins and carrier proteins, each facilitating solute movement in distinct ways.
Channel proteins: Form hydrophilic channels for rapid passage of solutes.
Carrier proteins: Bind solutes and undergo conformational changes to transport them across the membrane.
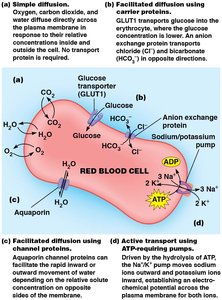
Simple Diffusion
Simple diffusion is the unassisted movement of solutes from high to low concentration, typically limited to small, nonpolar molecules.
Examples: CO2, O2, lipid-soluble substances, water, glycerol, ethanol.
Thermodynamic equilibrium: Achieved when no net movement occurs and free energy is minimized.
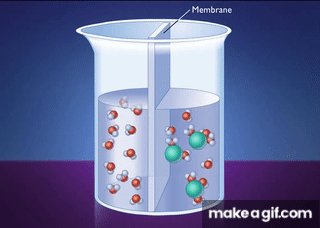
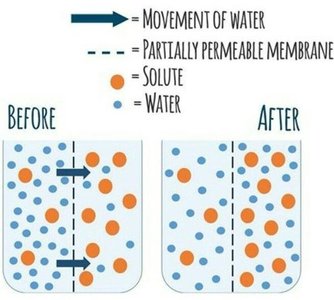
Osmosis: Diffusion of Water
Osmosis is the diffusion of water across a selectively permeable membrane, driven by differences in solute concentration.
Water moves toward higher solute concentration.
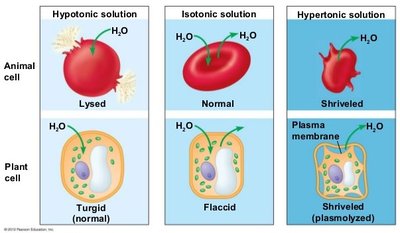
Osmolarity: Total solute concentration (Osm/L).
Tonicity: Effect of extracellular solution on cell water movement (hypotonic, isotonic, hypertonic).
Factors Affecting Diffusion
The rate of diffusion depends on solute size, polarity, and concentration gradient.
Size: Small molecules diffuse faster; large molecules require transport proteins.
Polarity: Nonpolar molecules diffuse faster; polar molecules are slowed by hydration shells.
Concentration gradient: Greater gradient increases diffusion rate.
Facilitated Diffusion
Facilitated diffusion enables large or polar substances to cross membranes with the help of transport proteins, moving down their concentration gradient.
Channel proteins: Provide hydrophilic passageways for solutes.
Carrier proteins: Bind solutes and undergo conformational changes.
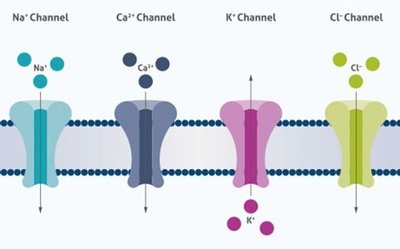
Channel Proteins
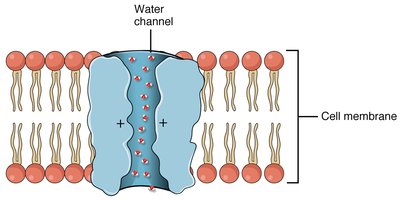
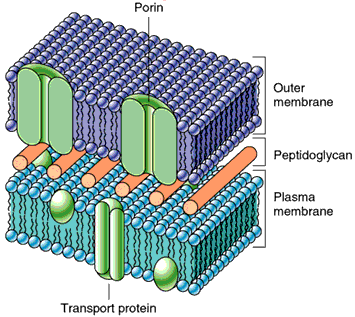
Channel proteins form hydrophilic transmembrane channels for specific solutes, including ions, porins, and aquaporins.
Ion channels: Highly selective for specific ions (Na+, K+, Ca2+, Cl-).
Porins: Larger, less specific channels found in bacteria, mitochondria, and chloroplasts.
Aquaporins: Channels for rapid water transport.
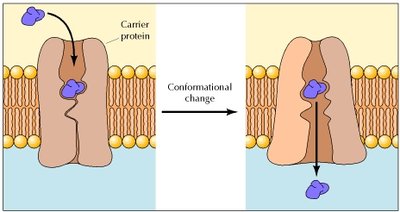
Carrier Proteins
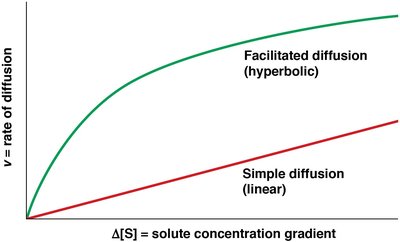
Carrier proteins are allosteric proteins that bind solutes and transport them via conformational changes. They exhibit saturation kinetics and can be subject to competitive inhibition.
Saturation kinetics: Transport rate reaches a maximum (Vmax) as solute concentration increases.
Competitive inhibition: Structurally similar molecules can inhibit transport.
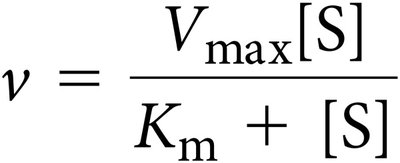
Types of Carrier Protein Transport
Carrier proteins can transport one or two solutes, classified as uniport, symport, or antiport.
Uniport: Single solute transported (e.g., GLUT1 for glucose).
Symport: Two solutes moved in the same direction.
Antiport: Two solutes moved in opposite directions (e.g., anion exchange protein for Cl- and HCO3-).
Passive vs Active Transport
Passive transport includes simple diffusion, facilitated diffusion, and osmosis, moving solutes down their gradient. Active transport moves solutes up their gradient, requiring energy.
Passive transport: No energy required; moves toward equilibrium.
Active transport: Requires energy; maintains nonequilibrium concentrations.
Comparison of Transport Mechanisms
The following table summarizes the properties of simple diffusion, facilitated diffusion, and active transport:
Properties | Simple Diffusion | Facilitated Diffusion | Active Transport |
|---|---|---|---|
Solutes transported | Small polar (H2O, glycerol), small nonpolar (O2, CO2), large nonpolar (oils, steroids) | Small polar (H2O, glycerol), large polar (glucose), ions (Na+, K+, Ca2+) | Large polar (glucose), ions (Na+, K+, Ca2+) |
Direction relative to gradient | Down | Down | Up |
Metabolic energy required | No | No | Yes |
Intrinsic directionality | No | No | Yes |
Membrane protein required | No | Yes | Yes |
Saturates kinetics | No | Yes | Yes |
Competitive inhibition | No | Yes | Yes |
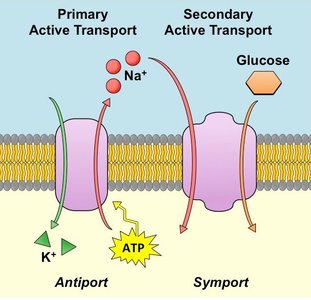
Active Transport: Direct and Indirect Mechanisms
Active transport is coupled to energy sources, either directly (primary) or indirectly (secondary).
Primary active transport: Directly powered by ATP hydrolysis (e.g., Na+/K+ ATPase).
Secondary active transport: Driven by the movement of one solute down its gradient, coupled to the movement of another solute up its gradient (e.g., sodium-glucose symporter).
Types of Transport ATPases
Transport ATPases are classified into four types based on structure and function:
P-Type ATPases: Reversibly phosphorylated; maintain ion gradients (e.g., Na+/K+ pump).
V-Type ATPases: Pump protons into organelles; establish ion gradients in vacuoles, lysosomes, etc.
F-Type ATPases: Found in mitochondria and chloroplasts; transport protons and synthesize ATP.
ABC-Type ATPases: Large family; importers and exporters, including multidrug resistance proteins.
Biological Relevance and Examples
Active transport is essential for nutrient uptake, waste removal, and maintenance of ion gradients. Examples include:
Na+/K+ ATPase: Maintains electrochemical gradients in animal cells.
Sodium-glucose symporter: Uptake of glucose in intestinal cells.
Bacteriorhodopsin proton pump: Uses light energy to create proton gradients in archaea.
Summary
Transport across membranes is vital for cellular function, involving a variety of mechanisms and proteins to overcome the permeability barrier. Understanding these processes is fundamental to cell biology.
