Consider the elements C, Se, B, Sn, and Cl. Identify which of these elements:
e. Forms a hydride with the empirical formula

 Verified step by step guidance
Verified step by step guidance


Consider the elements C, Se, B, Sn, and Cl. Identify which of these elements:
e. Forms a hydride with the empirical formula
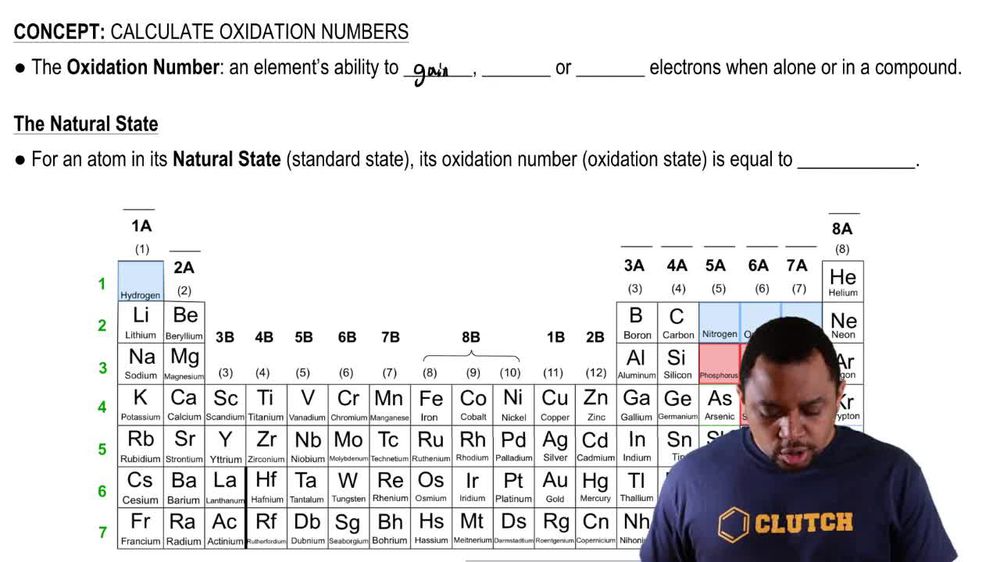
Locate each of the following elements on the periodic table.
(f) Group 5A element that forms the strongest p bonds
The following pictures represent structures of the hydrides of four second-row elements:
(1)
(2)
(3)
(4)
(c) Which compounds yield H2 gas when they are mixed together?
Look at the location of elements A, B, C, and D in the following periodic table:
(a) Write the formula of the oxide that has each of these elements in its highest oxidation state.
The following models represent the structures of binary
oxides of second- and third-row elements in their highest
oxidation states:
(a) Identify the non-oxygen atom in each case, and write the molecular formula for each oxide.