 Back
BackAcid-Base Equilibria and pH Calculations: Study Notes
Study Guide - Smart Notes

Acid-Base Equilibria and pH Calculations
Introduction to pH, pOH, and Acid-Base Concepts
The concept of pH and pOH is fundamental in understanding acid-base equilibria in aqueous solutions. pH measures the concentration of hydrogen ions ([H+]), while pOH measures hydroxide ions ([OH-]). The relationship between acids, bases, and their conjugates is essential for predicting solution behavior.
pH: Defined as
pOH: Defined as
Relationship: at 25°C
Acids: Increase [H+] in solution
Bases: Increase [OH-] in solution

Calculating pH and pOH in Aqueous Solutions
To determine the pH or pOH of a solution, use the concentration of the relevant ion and apply logarithmic calculations. For mixtures, consider the contributions from both acids and bases.
Strong Acids: Fully dissociate; [H+] equals acid concentration.
Strong Bases: Fully dissociate; [OH-] equals base concentration.
Example: For 0.1 M HCl,
Example: For 0.1 M NaOH, ,
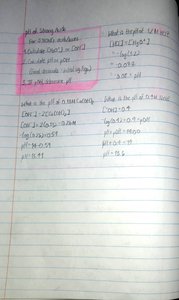
Acid-Base Equilibria: Weak Acids and Bases
Weak acids and bases do not fully dissociate in water. Their equilibrium concentrations are determined using the acid dissociation constant (Ka) or base dissociation constant (Kb), often with ICE tables (Initial, Change, Equilibrium).
ICE Table: Used to track changes in concentration during dissociation.
Equilibrium Expression:
Calculation Steps:
Write the dissociation equation.
Set up the ICE table.
Write the equilibrium expression.
Solve for x (change in concentration).
Calculate pH from [H+].
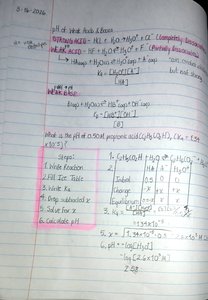
Conjugate Acids and Bases
Every acid has a conjugate base, and every base has a conjugate acid. The strength of an acid or base is inversely related to the strength of its conjugate.
Conjugate Acid: Formed when a base gains a proton.
Conjugate Base: Formed when an acid loses a proton.
Strong acids: Have weak conjugate bases.
Strong bases: Have weak conjugate acids.

Classification of Acids and Bases
Acids and bases can be classified as strong or weak based on their degree of dissociation in water. Common strong acids and bases are listed for reference.
Strong Acids: HCl, HBr, HI, HNO3, HClO4, H2SO4
Strong Bases: NaOH, KOH, LiOH, Ca(OH)2, Ba(OH)2
Weak Acids: Acetic acid, propanoic acid, etc.
Weak Bases: Ammonia, etc.

Sample Calculations: pH of Mixtures and Polyprotic Acids
Calculating the pH of mixtures or polyprotic acids involves considering all sources of H+ or OH- and using equilibrium concepts. For polyprotic acids, each dissociation step is treated separately.
Example: Mixing HCl and KOH, calculate final pH using stoichiometry and equilibrium.
Polyprotic Acids: Acids that can donate more than one proton, e.g., H2SO4.

Practice Problems and ICE Table Applications
ICE tables are used extensively in acid-base equilibrium calculations. Practice problems reinforce the method and help students master pH calculations for various scenarios.
Example: Calculate pH for 0.1 M NH3 using ICE table and Kb.
Example: Calculate pH for 0.5 M propanoic acid using ICE table and Ka.

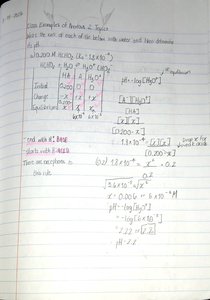
Summary Table: Strong and Weak Acids and Bases
The following table summarizes the classification of common acids and bases:
Type | Examples |
|---|---|
Strong Acids | HCl, HBr, HI, HNO3, HClO4, H2SO4 |
Weak Acids | CH3COOH, HCOOH, C3H7COOH |
Strong Bases | NaOH, KOH, LiOH, Ca(OH)2, Ba(OH)2 |
Weak Bases | NH3, CH3NH2 |
Additional info: These notes cover the essential concepts and calculations for acid-base equilibria, pH, pOH, and the use of ICE tables, which are directly relevant to Ch.14 and Ch.15 of a general chemistry course.
