 Back
BackAcid-Base Equilibria: Concepts, Calculations, and Applications
Study Guide - Smart Notes

Chapter 16: Acid-Base Equilibria
Classifications of Acids and Bases
Acids and bases can be classified according to several theories, each broadening the definition and scope of acid-base chemistry.
Arrhenius Definition: An acid increases the concentration of hydrogen ions (H+) in water, while a base increases the concentration of hydroxide ions (OH-).
Brønsted–Lowry Definition: An acid is a proton (H+) donor, and a base is a proton acceptor.
Lewis Definition: A Lewis acid is an electron pair acceptor, and a Lewis base is an electron pair donor.
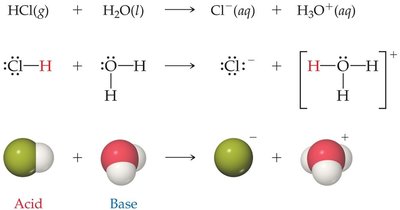
Example: HCl acts as an acid by donating a proton to H2O, which acts as a base.
Amphiprotic Substances
Some substances, such as water, can act as either an acid or a base depending on the reaction context. These are called amphiprotic substances.
Example: H2O can donate a proton (acting as an acid) or accept a proton (acting as a base).
Conjugate Acid-Base Pairs
When an acid donates a proton, it forms its conjugate base; when a base accepts a proton, it forms its conjugate acid. These pairs differ by one H+ ion.
Acid + Base → Conjugate Base + Conjugate Acid
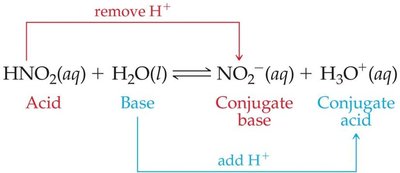
Example: In the reaction HNO2 + H2O ↔ NO2- + H3O+, HNO2 and NO2- are a conjugate acid-base pair.
Relative Strengths of Acids and Bases
Acids and bases vary in their ability to donate or accept protons. The stronger the acid, the weaker its conjugate base, and vice versa. Acid and base strengths can be arranged in order of reactivity.
Strong acids dissociate completely in water; weak acids only partially dissociate.
Strong bases dissociate completely in water; weak bases only partially accept protons.
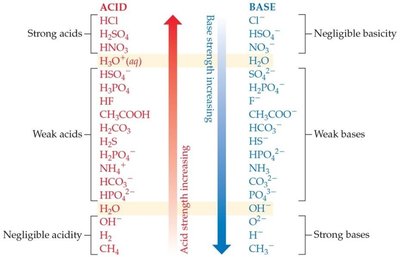
Example: HCl is a strong acid, while CH3COOH is a weak acid.
Autoionization of Water
Water can ionize to form hydronium and hydroxide ions, demonstrating its amphoteric nature. This process is called autoionization:
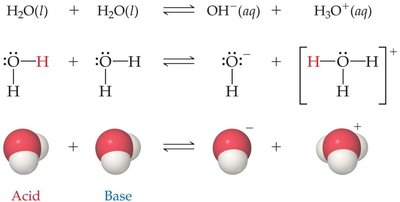
The equilibrium constant for this reaction is called the ion product constant for water, :
at 25°C
Aqueous Solutions: Acidic, Basic, or Neutral
The nature of an aqueous solution depends on the relative concentrations of H+ and OH- ions:
Acidic:
Neutral:
Basic:

The pH Scale
The pH scale is a logarithmic scale used to express the concentration of hydrogen ions in solution:
Neutral pH is 7.00; acidic pH is below 7.00; basic pH is above 7.00.
Only digits after the decimal point are significant in logarithms.
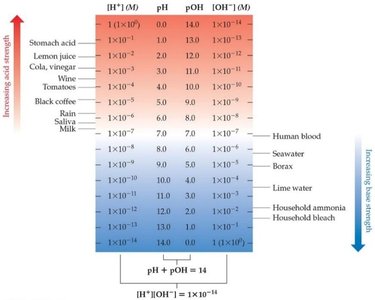
Example: If , then .
pOH and Other "p" Scales
The "p" notation indicates the negative logarithm of a quantity. For bases, the pOH is used:
at 25°C
Strong Acids and Bases
Strong acids completely dissociate in water and are strong electrolytes. The seven common strong acids are:
HCl, HBr, HI, HNO3, H2SO4, HClO3, HClO4
For monoprotic strong acids, acid concentration.
Strong bases are soluble hydroxides of alkali and heavier alkaline earth metals, which dissociate completely in water:
Weak Acids and Bases
Weak acids and weak bases only partially ionize in solution. Their strength is quantified by the acid dissociation constant () or base dissociation constant ().
Example: Phenol, ; Acetic acid,
To calculate from pH:
Find from pH, set up an ICE table, and solve for .
Percent Ionization:
Polyprotic Acids
Polyprotic acids can donate more than one proton. The first proton is always lost more easily than subsequent protons.
Example: For H2SO3, ,
Weak Bases
Weak bases, such as NH2OH (), only partially accept protons in solution. Their strength is measured by and .
Example: Codeine, a weak organic base, with for a M solution.
Relationship Between and
For a conjugate acid-base pair, the product of and equals :
at 25°C
Example: For NH3 () and NH4+, can be calculated using this relationship.
Lewis Acids and Bases
All Brønsted–Lowry acids and bases are also Lewis acids and bases. A Lewis base must have an unshared pair of electrons to bind a proton or other electron-deficient species.
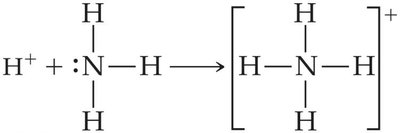
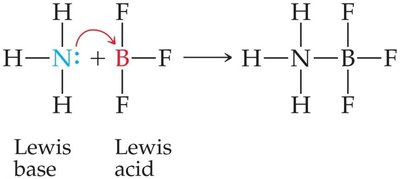
Example: NH3 acts as a Lewis base by donating an electron pair to H+ or BF3 (a Lewis acid).
Additional info: For all calculations, use the appropriate equilibrium expressions and ICE tables to solve for unknowns. Practice problems provided in the notes reinforce these concepts and calculation methods.
