 Back
BackAcid-Base Equilibrium: Foundations, Theories, and Calculations
Study Guide - Smart Notes

Acid-Base Equilibrium
Acids and Bases – A Review
Acids and bases are fundamental chemical species with distinct properties and definitions. Their behavior in aqueous solutions is central to many chemical processes, including biological and industrial systems.
Acids: Substances that can donate protons (H+) or accept electron pairs.
Bases: Substances that can accept protons or donate electron pairs.
Water is amphoteric, meaning it can act as both an acid and a base depending on the reaction context.

Historical Development of Acid-Base Theories
The understanding of acids and bases has evolved through several key theories:
Boyle's Observations: Acids have a sour taste, are corrosive, and turn litmus red; bases (alkalies) are slippery and turn litmus blue.
Lemery's Model: Proposed acids have sharp, prickly particles, while bases have soft, round particles.


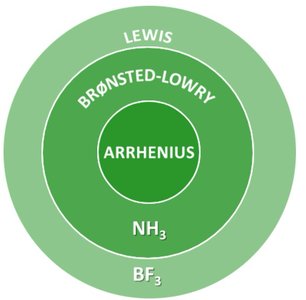
Modern Acid-Base Theories
Arrhenius Theory: Acids produce H+ in water; bases produce OH-.
Brønsted-Lowry Theory: Acids are proton donors; bases are proton acceptors. This theory expands the definition to include more substances and reactions.
Lewis Theory: Acids are electron pair acceptors; bases are electron pair donors. This is the broadest definition and includes non-protic systems.
Conjugate Acid-Base Pairs
Every acid-base reaction involves the transfer of a proton from an acid to a base, forming conjugate acid-base pairs:
Conjugate Acid: The species formed when a base gains a proton.
Conjugate Base: The species formed when an acid loses a proton.
Example: NH3 + H2O ⇌ NH4+ + OH-

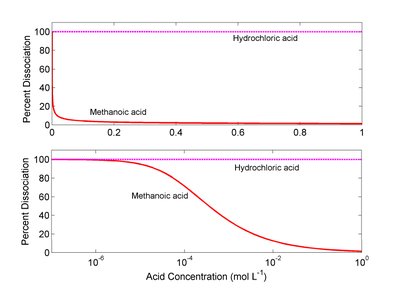
Strength of Acids and Bases
The strength of an acid or base is determined by its degree of ionization in water:
Strong acids/bases: Completely ionize in water (e.g., HCl, NaOH).
Weak acids/bases: Partially ionize in water (e.g., CH3COOH, NH3).
The conjugate of a strong acid/base is extremely weak and does not react with water.

Acid and Base Equilibrium Constants
The extent of ionization for weak acids and bases is quantified by equilibrium constants:
Acid dissociation constant (Ka):
Base hydrolysis constant (Kb):
Relationship: at 25°C
pKa: ; pKb:
The pH and pOH Scales
The pH scale is a logarithmic measure of hydronium ion concentration:
(at 25°C)

Calculating pH of Strong and Weak Acids/Bases
Strong acids/bases: [H3O+] or [OH-] equals the initial concentration.
Weak acids/bases: Use an ICE table and the equilibrium constant to solve for [H3O+] or [OH-].
Percent ionization:
Small x approximation: Valid if (or 5%).


pH of Salt Solutions
The pH of a salt solution depends on the acid-base properties of the ions formed upon dissolution:
Salts of strong acid and strong base (e.g., NaCl): Neutral solution (pH = 7).
Salts of weak acid and strong base (e.g., NaF): Basic solution (pH > 7).
Salts of strong acid and weak base (e.g., NH4Cl): Acidic solution (pH < 7).
Structural Factors Affecting Acid and Base Strength
Binary acids: Acid strength increases with greater electronegativity (across a period) and larger atomic radius (down a group).
Oxyacids: Acid strength increases with more oxygen atoms (resonance stabilization) and higher electronegativity of the central atom (inductive effect).
Resonance: More resonance forms in the conjugate base stabilize the negative charge, increasing acid strength.
Polyprotic Acids and Bases
Polyprotic acids and bases can donate or accept more than one proton. Each ionization step has its own Ka or Kb, with the first ionization being the strongest.
Example: Carbonic acid (H2CO3) is diprotic, with two dissociation steps.
Summary Table: Strong and Weak Acids and Bases
Strong Acids | Weak Acids | Strong Bases | Weak Bases |
|---|---|---|---|
HCl, HBr, HI, HNO3, HClO4, HClO3, H2SO4 | HF, H2S, H2CO3, CH3COOH, HNO2, H3PO4 | NaOH, KOH, Ca(OH)2, Sr(OH)2, Ba(OH)2, NaNH2 | NH3, CH3NH2, HCO3-, F- |
Key Equations
Additional info: This guide covers the foundational concepts, calculations, and structural factors for acid-base equilibrium, suitable for first-year college chemistry students.
