 Back
BackAcid–Base Equilibria: Concepts, Calculations, and Applications
Study Guide - Smart Notes

Acid–Base Equilibria
Introduction to Acid–Base Theories
Acid–base chemistry is fundamental to understanding chemical reactions in aqueous solutions. Several definitions exist to describe acids and bases, each broadening the scope of what substances can be considered acids or bases.
Arrhenius Definition: An acid increases the concentration of hydrogen ions (H+) in water, while a base increases the concentration of hydroxide ions (OH−).
Brønsted–Lowry Definition: An acid is a proton (H+) donor, and a base is a proton acceptor.
Lewis Definition: A Lewis acid is an electron pair acceptor, and a Lewis base is an electron pair donor. This definition encompasses a wider range of chemical species.
Amphiprotic and Amphoteric Substances
Some substances, such as water, can act as either acids or bases depending on the reaction. These are called amphiprotic (can donate or accept a proton) or amphoteric (can act as acid or base in general).
Examples: HCO3−, HSO4−, H2O
Conjugate Acid–Base Pairs
When an acid donates a proton, it forms its conjugate base; when a base accepts a proton, it forms its conjugate acid. These pairs differ by one proton.
Example: NH3 (base) ⇌ NH4+ (conjugate acid)
Example: HSO4− (acid) ⇌ SO42− (conjugate base)
Strengths of Acids and Bases
Strong and Weak Acids/Bases
The strength of an acid or base depends on its degree of ionization in water.
Strong acids and strong bases dissociate completely in water. Their conjugate bases/acids are extremely weak and do not react further in water.
Weak acids and weak bases only partially dissociate, establishing an equilibrium between the undissociated and dissociated forms.
In any acid–base reaction, equilibrium favors the formation of the weaker acid and base.
Autoionization of Water and the Ion Product Constant
Autoionization of Water
Water can act as both an acid and a base, leading to a small degree of self-ionization:
The equilibrium constant for this process is called the ion product constant for water ():
at 25°C
pH and pOH Scales
The pH scale is a logarithmic measure of hydrogen ion concentration:
Similarly, pOH is defined as:
At 25°C, .
Neutral solution: pH = 7.00
Acidic solution: pH < 7.00
Basic solution: pH > 7.00
![pH, pOH, [H+], and [OH-] relationships and examples](https://static.studychannel.pearsonprd.tech/study_guide_files/general-chemistry/sub_images/c113ae5b_image_2.png)
![Table: Relationships among [H+], [OH-], and pH at 25°C](https://static.studychannel.pearsonprd.tech/study_guide_files/general-chemistry/sub_images/c113ae5b_image_3.png)
Calculations Involving pH, pOH, and Ion Concentrations
Using to Find [H+] or [OH−]
Given one ion concentration, the other can be found using :
To find pH from [H+]:
To find [H+] from pH:
Strong and Weak Acids and Bases: Calculations
Strong Acids and Bases
Strong acids and bases dissociate completely, so their concentration equals the concentration of H+ or OH− produced.
Example: 0.10 M HCl yields [H+] = 0.10 M, pH = 1.00
Weak Acids and Bases
Weak acids and bases only partially dissociate. Their strength is described by equilibrium constants:
Acid-dissociation constant ():
Base-dissociation constant ():
The larger the or , the stronger the acid or base.
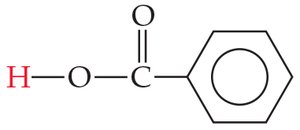
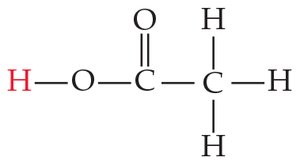
Percent Ionization
Percent ionization quantifies the fraction of acid molecules that ionize:
Polyprotic Acids
Definition and Properties
Polyprotic acids can donate more than one proton. Each successive ionization has a smaller value. If and differ by more than , the pH is determined mainly by the first ionization.
Weak Bases and Their Equilibria
Types of Weak Bases
Weak bases include neutral molecules with lone pairs (e.g., ammonia, amines) and anions of weak acids (conjugate bases).
Example: Ammonia (NH3) is a classic weak base.
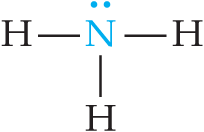

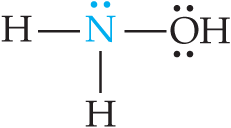
Relationship Between and
For a conjugate acid–base pair:
If you know one, you can calculate the other.
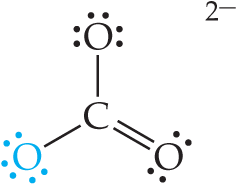
Acid–Base Properties of Salts
Hydrolysis of Salts
Many salts react with water to produce acidic or basic solutions. The pH depends on the nature of the cation and anion:
Anions of strong acids are neutral.
Anions of weak acids are basic (conjugate bases).
Cations of strong bases are neutral.
Cations of weak bases (polyatomic or transition metal cations) are acidic.
Factors Affecting Acid Strength
Binary Acids
For binary acids (H and one other element):
Down a group: bond strength is most important; weaker bonds = stronger acids.
Across a period: bond polarity is most important; greater polarity = stronger acid.
Oxyacids
For oxyacids (H, O, and another element):
Greater electronegativity of the central atom increases acidity.
More oxygen atoms increase acidity (higher oxidation state).
Carboxylic Acids
Carboxylic acids are organic acids with the –COOH group. Their acidity is enhanced by resonance stabilization of the conjugate base and electron-withdrawing effects of additional oxygen atoms.
Lewis Acid–Base Chemistry
Lewis Acids and Bases
Lewis acids accept electron pairs; Lewis bases donate electron pairs. This definition includes all Brønsted–Lowry acids and bases, as well as other species such as metal cations and electron-deficient molecules.
Additional info: The included images of molecular structures and tables are directly relevant to the explanation of acid/base strength, molecular structure, and the relationships among pH, pOH, [H+], and [OH−].
