Back
BackAcid–Base Equilibria: Concepts, Calculations, and Chemical Structure
Study Guide - Smart Notes

Chapter 16: Acid–Base Equilibria
16.1 Classifications of Acids and Bases
Acids and bases are fundamental chemical species with several definitions, each broadening the scope of acid–base chemistry:
Arrhenius Definition: An acid increases the concentration of hydrogen ions (H+) in water, while a base increases the concentration of hydroxide ions (OH−).
Brønsted–Lowry Definition: An acid is a proton (H+) donor; a base is a proton acceptor. A Brønsted–Lowry acid must have at least one removable (acidic) proton, and a base must have a nonbonding pair of electrons to accept a proton.
Lewis Definition: A Lewis acid is an electron pair acceptor, and a Lewis base is an electron pair donor. All Brønsted–Lowry acids and bases are also Lewis acids and bases.
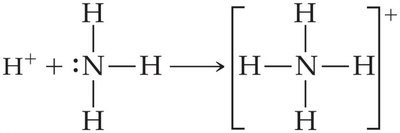
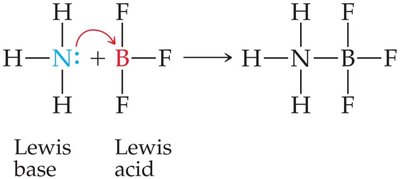
Amphiprotic Substances
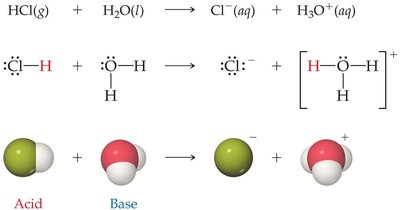
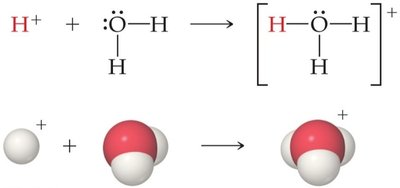
Some substances, such as water, can act as both acids and bases (amphiprotic). Water can accept a proton (acting as a base) or donate a proton (acting as an acid) depending on the reaction partner.
16.2 Conjugate Acid–Base Pairs
In acid–base reactions, the acid and base differ by one proton (H+), forming a conjugate acid–base pair. The acid yields its conjugate base after donating a proton, and the base yields its conjugate acid after accepting a proton.
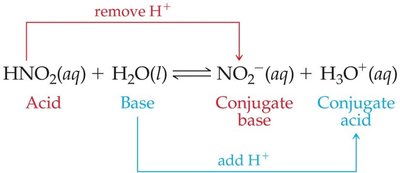
Relative Strengths of Acids and Bases
Acids and bases vary in their ability to donate or accept protons. The stronger the acid, the weaker its conjugate base, and vice versa. In aqueous solution, H3O+ and OH− are the strongest acid and base, respectively, that can exist.
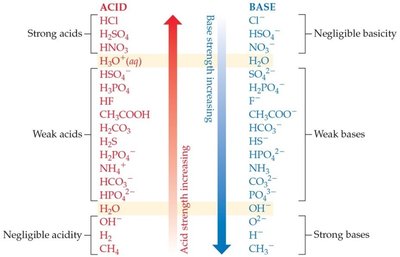
Acid–Base Equilibrium
Equilibrium in acid–base reactions favors the transfer of a proton from the stronger acid to the stronger base, forming the weaker acid and base. For example, in the reaction of HCl with water, the equilibrium lies far to the right because H2O is a much stronger base than Cl−.
16.3 Autoionization of Water
Water is amphoteric and can undergo autoionization, producing hydronium and hydroxide ions:
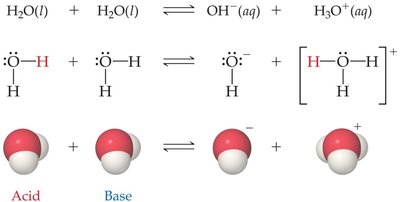
The equilibrium constant for this process is the ion product of water, :
at 25°C
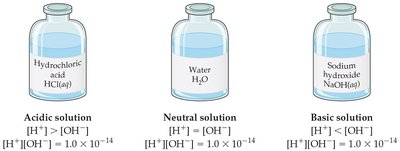
Solutions can be classified as:
Neutral:
Acidic:
Basic:
16.4 The pH Scale
The pH scale quantifies the concentration of hydrogen ions in solution:
Neutral pH is 7.00; acidic solutions have pH < 7.00; basic solutions have pH > 7.00.
![pH, pOH, [H+], and [OH-] scale](https://static.studychannel.pearsonprd.tech/study_guide_files/general-chemistry/sub_images/94fa4df1_image_11.png)
The pOH scale is similarly defined:
at 25°C
![pH, pOH, [H+], and [OH-] scale (duplicate for clarity)](https://static.studychannel.pearsonprd.tech/study_guide_files/general-chemistry/sub_images/94fa4df1_image_12.png)
Measuring pH
pH meters provide accurate measurements using electrodes to detect voltage changes.
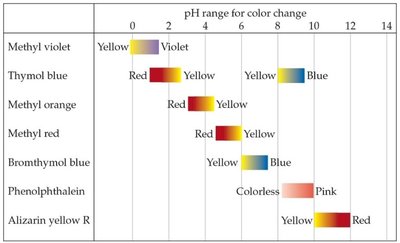
Indicators are compounds that change color depending on the pH, useful for quick, less precise measurements.


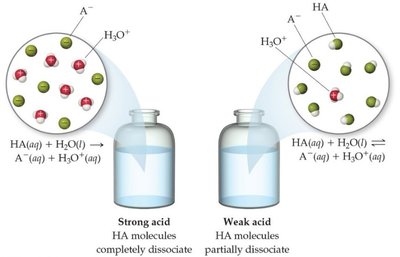
16.5 Strong Acids and Bases
Strong acids and bases dissociate completely in aqueous solution, acting as strong electrolytes. The seven strong acids are HCl, HBr, HI, HNO3, H2SO4, HClO3, and HClO4. Strong bases are soluble hydroxides of alkali and heavier alkaline earth metals.
For monoprotic strong acids:
16.6 Weak Acids
Weak acids only partially dissociate in water, establishing an equilibrium. The acid-dissociation constant () quantifies their strength:
The larger the , the stronger the acid.
Strong acids completely dissociate, while weak acids only partially dissociate.
Calculating from pH
Write the equilibrium equation and expression for .
Determine equilibrium concentrations using initial values and changes (often with an ICE table).
Calculate using measured pH to find .
Percent ionization is calculated as:
Calculating pH Using
Write the ionization equilibrium and expression.
Set up an ICE table to solve for .
Calculate pH from .
Polyprotic Acids
Polyprotic acids can donate more than one proton. Each dissociation step has its own value, with the first dissociation being the largest. If and differ by more than 1000, the pH depends mainly on the first dissociation.
16.7 Weak Bases
Weak bases, such as ammonia (NH3), partially accept protons in water. The base-dissociation constant () quantifies their strength:
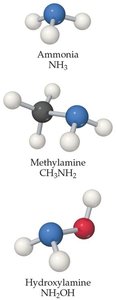
Types of weak bases include neutral molecules with lone pairs (e.g., amines) and anions of weak acids (conjugate bases).
Calculating pH Using
Write the equilibrium equation and expression.
Set up an ICE table to solve for .
Calculate pOH, then pH using .
16.8 Relationship Between and
For a conjugate acid–base pair, and are related by the ion-product constant for water:
If you know one, you can calculate the other.
16.9 Acid–Base Properties of Salt Solutions
Many salts react with water (hydrolysis) to produce acidic or basic solutions. The acid–base nature of a salt depends on the properties of its cation and anion:
Anions of strong acids are neutral; anions of weak acids are basic.
Cations of group I/II metals are neutral; polyatomic cations (e.g., NH4+) are acidic; transition metal cations are acidic due to their Lewis acidity.

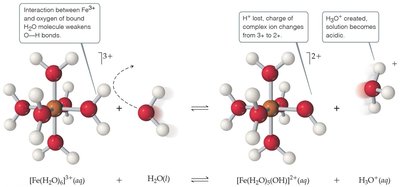
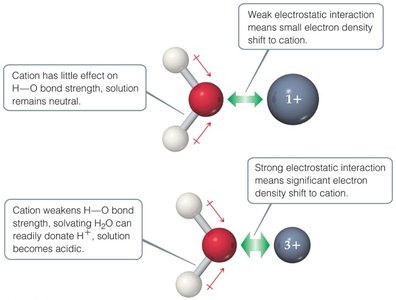
16.10 Acid–Base Behavior and Chemical Structure
The strength of an acid depends on:
Bond polarity: The H–A bond must be polarized with δ+ on H and δ− on A.
Bond strength: Weaker H–A bonds lead to stronger acids.
Stability of the conjugate base: Greater stability (e.g., via resonance) increases acid strength.
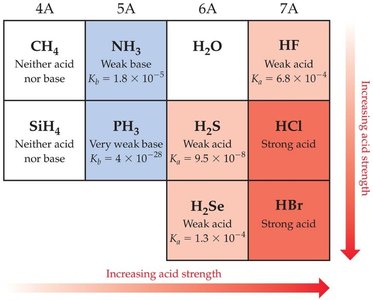
Binary Acids
Binary acids consist of hydrogen and one other element. Down a group, bond strength is most important; across a period, bond polarity dominates.
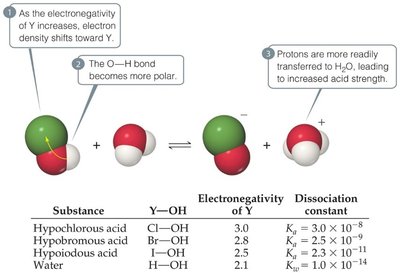
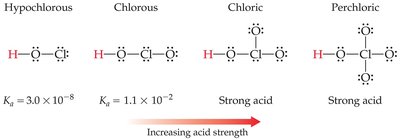
Oxyacids
Oxyacids contain H, O, and a nonmetal. Acidity increases with the electronegativity of the nonmetal and with the number of oxygen atoms (higher oxidation state).
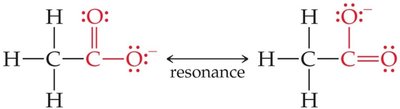
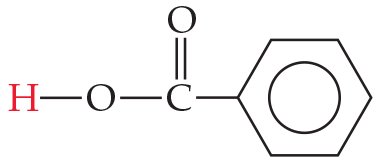
Carboxylic Acids
Carboxylic acids (–COOH group) are stabilized by resonance and the electron-withdrawing effect of the second oxygen, making them relatively strong organic acids.