 Back
BackAcids and Bases: Concepts, Calculations, and Applications
Study Guide - Smart Notes

Acids and Bases
Definitions of Acids and Bases
Acids and bases are fundamental concepts in chemistry, with several definitions that describe their behavior in aqueous solutions.
Arrhenius Definition: An acid increases the concentration of hydrogen ions (H+) in water, while a base increases the concentration of hydroxide ions (OH-).
Brønsted-Lowry Definition: An acid is a proton (H+) donor, and a base is a proton acceptor. Substances that can act as either are called amphiprotic.
Lewis Definition: A Lewis acid is an electron-pair acceptor, and a Lewis base is an electron-pair donor.
Example: Water (H2O) is amphiprotic because it can act as both an acid and a base.
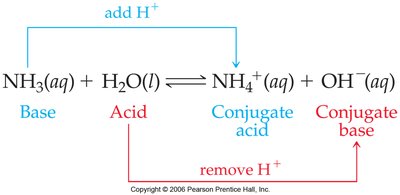
Proton Transfer and Conjugate Acid-Base Pairs
When an acid dissolves in water, a proton is transferred from the acid to the base (often water), forming a conjugate acid and a conjugate base.
Conjugate Acid: The species formed when a base gains a proton.
Conjugate Base: The species formed when an acid loses a proton.
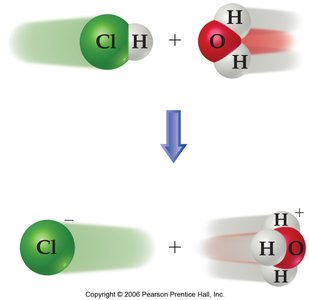
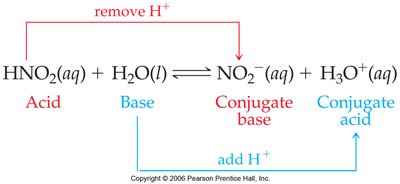
Example: In the reaction HNO2 + H2O ↔ NO2- + H3O+, HNO2 is the acid, H2O is the base, NO2- is the conjugate base, and H3O+ is the conjugate acid.
Acid and Base Strength
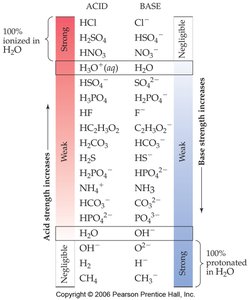
The strength of an acid or base depends on its degree of ionization in water.
Strong acids and strong bases dissociate completely in water.
Weak acids and weak bases only partially dissociate.
Substances with negligible acidity or basicity do not ionize in water.
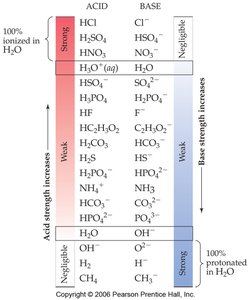
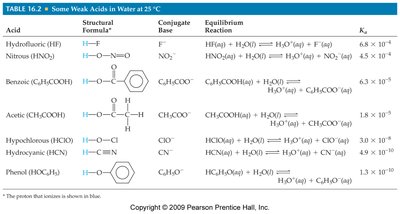
Example: HCl is a strong acid, while CH3COOH (acetic acid) is a weak acid.
Predicting the Position of Acid-Base Equilibria
In any acid-base reaction, the equilibrium favors the transfer of the proton to the stronger base. The equilibrium constant (K) indicates the extent of the reaction:
If K > 1, products are favored (reaction goes to the right).
If K < 1, reactants are favored (reaction goes to the left).
pH, pOH, and the Ion-Product Constant
Autoionization of Water and the Ion-Product Constant (Kw)
Water undergoes autoionization, producing hydronium and hydroxide ions:
The equilibrium constant for this process is:
At 25°C, .
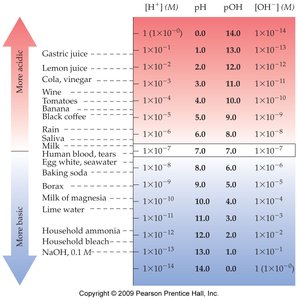
pH and pOH Scales
The pH scale quantifies the acidity or basicity of a solution:
At 25°C,

Example: Pure water has M, so pH = 7.00.
Relationships Among [H+], [OH-], and pH
Solution Type | [H+] (M) | [OH-] (M) | pH Value |
|---|---|---|---|
Acidic | >1.0 × 10-7 | <1.0 × 10-7 | <7.00 |
Neutral | =1.0 × 10-7 | =1.0 × 10-7 | =7.00 |
Basic | <1.0 × 10-7 | >1.0 × 10-7 | >7.00 |
![Table of relationships among [H+], [OH-], and pH](https://static.studychannel.pearsonprd.tech/study_guide_files/general-chemistry/sub_images/db0f3de0_image_7.png)
Calculating pH and Concentrations
Given [H+], calculate pH:
Given pH, calculate [H+]:
Given [OH-], use to find [H+]:
Example: If [OH-] = 0.010 M, then [H+] = M, so pH = 12.
Acid and Base Dissociation Constants
Acid Dissociation Constant (Ka)
The acid dissociation constant quantifies the strength of a weak acid:
The larger the Ka, the stronger the acid.
Base Dissociation Constant (Kb)
For weak bases, the base dissociation constant is:
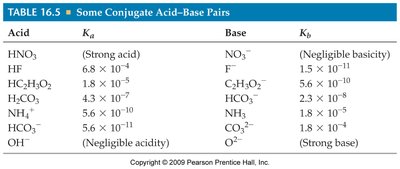
Relationship Between Ka and Kb
For a conjugate acid-base pair:
Polyprotic Acids
Definition and Properties
Polyprotic acids can donate more than one proton. Each dissociation step has its own Ka value, with the first dissociation usually being the largest.
Factors Affecting Acid Strength
Bond Polarity and Strength
The more polar and weaker the H-X bond, the stronger the acid. Acidity increases across a period and down a group in the periodic table.
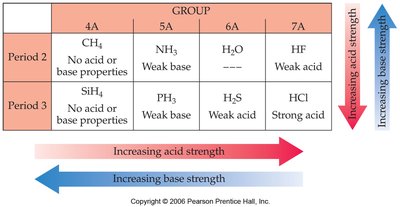
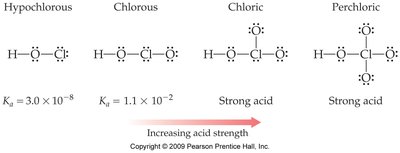
Oxyacids and Electronegativity
For oxyacids (acids with the structure H-O-Y), acidity increases with the electronegativity of Y and with the number of oxygen atoms attached to Y.
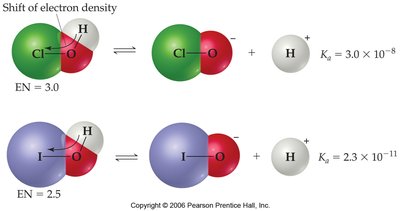
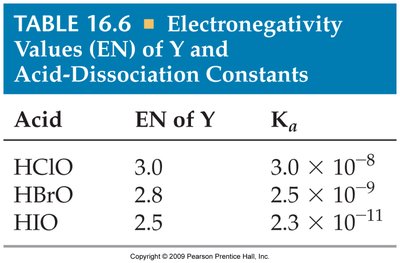
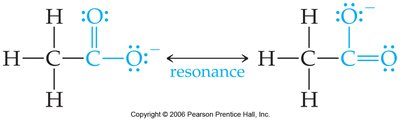
Resonance Stabilization
Resonance in the conjugate base stabilizes the base, making the acid stronger.
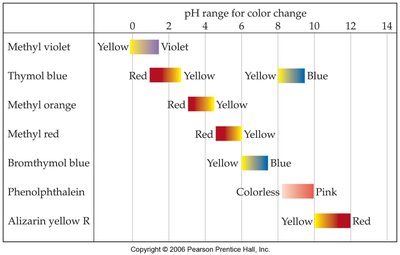
Measuring pH
Indicators and pH Meters
pH can be measured using indicators (which change color at specific pH ranges) or with electronic pH meters for more precise measurements.
Summary Table: Common Weak Acids and Bases
Acid | Ka | Base | Kb |
|---|---|---|---|
HF | 6.8 × 10-4 | F- | 1.5 × 10-11 |
CH3COOH | 1.8 × 10-5 | CH3COO- | 5.6 × 10-10 |
NH4+ | 5.6 × 10-10 | NH3 | 1.8 × 10-5 |
Approximation in Equilibrium Calculations
5% Rule for Approximations
When the equilibrium constant is very small and the initial concentration of reactant is large, the change in concentration (x) can often be neglected. The approximation is valid if:

