 Back
BackAcids and Bases: Definitions, Properties, and Calculations
Study Guide - Smart Notes

Acids and Bases
Definitions of Acids and Bases
Acids and bases are fundamental concepts in chemistry, with several definitions that help explain their behavior in different contexts. The three main definitions are Arrhenius, Brønsted-Lowry, and Lewis.
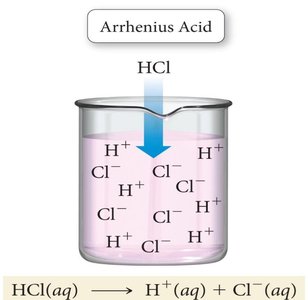
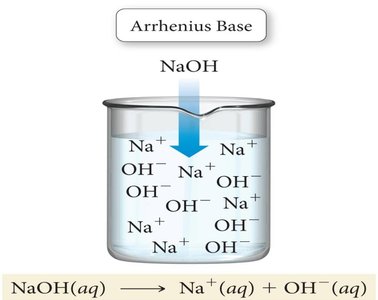
Arrhenius Acids and Bases: An Arrhenius acid is a substance that produces H+ ions when dissolved in water, while an Arrhenius base produces OH− ions in water.
Example: Hydrochloric acid (HCl) dissociates in water to produce H+ and Cl− ions.
Example: Sodium hydroxide (NaOH) dissociates in water to produce Na+ and OH− ions.
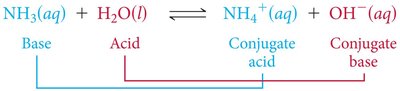
Brønsted-Lowry Acids and Bases: A Brønsted-Lowry acid is a proton (H+) donor, and a Brønsted-Lowry base is a proton acceptor. Acid-base reactions always involve the transfer of a proton.
Example: Ammonia (NH3) acts as a base, accepting a proton from water, which acts as an acid. $\mathrm{NH_3(aq) + H_2O(l) \rightleftharpoons NH_4^+(aq) + OH^-(aq)}$
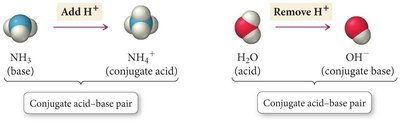
Conjugate Acid-Base Pairs: When an acid donates a proton, it forms its conjugate base; when a base accepts a proton, it forms its conjugate acid.
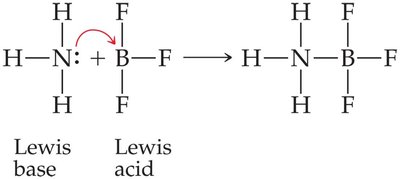
Lewis Acids and Bases: A Lewis acid is an electron pair acceptor, and a Lewis base is an electron pair donor. This definition is broader and includes reactions not involving protons.
Example: Ammonia (Lewis base) donates an electron pair to boron trifluoride (Lewis acid) to form an adduct.
Water as a Solvent
Water is a unique solvent because it can act as both an acid and a base (amphoteric). This property leads to the auto-ionization of water, which is fundamental to understanding pH and acid-base equilibria.
Auto-Ionization of Water: Water molecules can transfer protons between each other, resulting in the formation of hydronium (H3O+) and hydroxide (OH−) ions. $\mathrm{H_2O(l) + H_2O(l) \rightleftharpoons H_3O^+(aq) + OH^-(aq)}$
Ion-Product Constant (Kw): The equilibrium constant for water's auto-ionization is $K_w = [H_3O^+][OH^-] = 1.0 \times 10^{-14}$ at 25°C.
pH, pOH, and pKw: These are logarithmic measures of acidity and basicity. $\mathrm{pH = -log[H_3O^+]}$ $\mathrm{pOH = -log[OH^-]}$ $\mathrm{pK_w = -log K_w = 14.00}$ $\mathrm{pH + pOH = 14.00}$ at 25°C
pH Scale and Measurements
The pH scale is a logarithmic scale used to quantify the acidity or basicity of a solution. It ranges from 0 (very acidic) to 14 (very basic), but values outside this range are possible in highly concentrated solutions.
pH Scale: Each unit change in pH corresponds to a tenfold change in [H+].

pOH Scale: Similar to pH, but for hydroxide ion concentration.

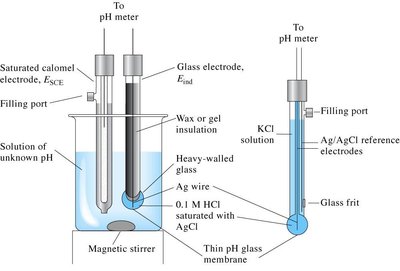
pH Measurement: pH is measured using a pH meter, which uses a glass electrode sensitive to [H+].

Example Calculation: If pH = 4.70, then $[H_3O^+] = 10^{-4.70} = 2.0 \times 10^{-5}$ M (acidic solution).
Example Calculation: If $[OH^-] = 2.9 \times 10^{-4}$ M, then $\mathrm{pOH = -log(2.9 \times 10^{-4}) = 3.54}$, $\mathrm{pH = 14.00 - 3.54 = 10.46}$ (basic solution).
Weak Acids: Acid Dissociation Constant (Ka), % Ionization, pH Calculations
Weak acids only partially ionize in water. Their strength is quantified by the acid dissociation constant (Ka), and their degree of ionization can be calculated.
Strong Acids: Ionize completely in water. Examples include HCl, HBr, HI, HNO3, HClO4, HClO3, and H2SO4 (first proton only).
Weak Acids: Ionize partially. The equilibrium reaction for a generic monoprotic acid (HA) is: $\mathrm{HA(aq) + H_2O(l) \rightleftharpoons H_3O^+(aq) + A^-(aq)}$ $\mathrm{K_a = \frac{[H_3O^+][A^-]}{[HA]}}$
pKa: $\mathrm{pK_a = -log K_a}$; smaller pKa means stronger acid.
% Ionization: The percent ionization of a weak acid is calculated as: $\mathrm{\%\,ionization = \frac{[H_3O^+]_{eq}}{[HA]_i} \times 100}$
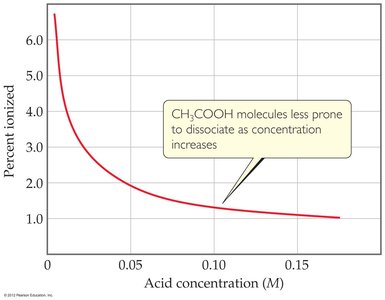
Effect of Dilution: Diluting a weak acid increases its percent ionization.

Example Calculation: For 0.15 M acetic acid ($K_a = 1.8 \times 10^{-5}$): $\mathrm{K_a = \frac{x^2}{0.15} = 1.8 \times 10^{-5}}$ $x = \sqrt{1.8 \times 10^{-5} \times 0.15} = 1.6 \times 10^{-3}$ $\mathrm{pH = -log(1.6 \times 10^{-3}) = 2.80}$ $\mathrm{\%\,ionization = \frac{1.6 \times 10^{-3}}{0.15} \times 100 = 1.1\%}$
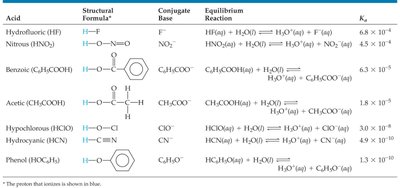
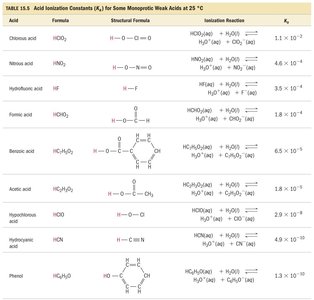
Acid Dissociation Constants for Selected Weak Acids
Tables of Ka values help compare the strengths of various weak acids. The higher the Ka, the stronger the acid.
Acid | Formula | Conjugate Base | Equilibrium Reaction | Ka |
|---|---|---|---|---|
Hydrofluoric acid | HF | F− | HF(aq) + H2O(l) ⇌ H3O+(aq) + F−(aq) | 6.8 × 10−4 |
Benzoic acid | C6H5COOH | C6H5COO− | C6H5COOH(aq) + H2O(l) ⇌ H3O+(aq) + C6H5COO−(aq) | 6.3 × 10−5 |
Acetic acid | CH3COOH | CH3COO− | CH3COOH(aq) + H2O(l) ⇌ H3O+(aq) + CH3COO−(aq) | 1.8 × 10−5 |
Phenol | C6H5OH | C6H5O− | C6H5OH(aq) + H2O(l) ⇌ H3O+(aq) + C6H5O−(aq) | 1.3 × 10−10 |




Additional info: The tables above are reconstructed from the original materials and may include inferred content for completeness.
