Back
BackAcids and Bases: Definitions, Strength, Equilibria, and Applications
Study Guide - Smart Notes

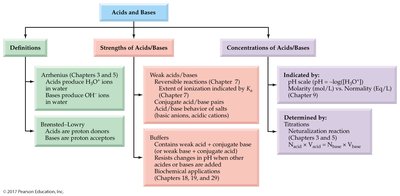
Acids and Bases
Definitions of Acids and Bases
Acids and bases are fundamental concepts in chemistry, with several definitions used to describe their behavior in solution. The Arrhenius and Brønsted-Lowry definitions are most commonly used in general chemistry.
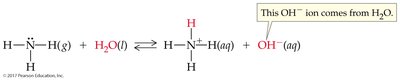
Arrhenius Acid: Produces hydronium ion (H3O+) when dissolved in water.
Arrhenius Base: Produces hydroxide ion (OH–) when dissolved in water.
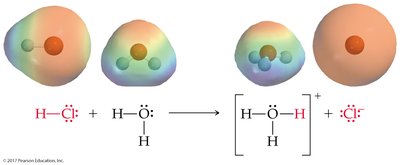
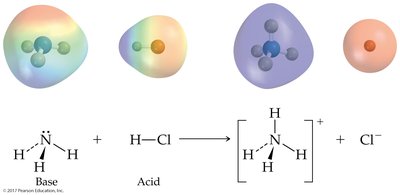
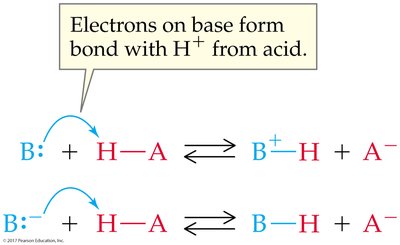
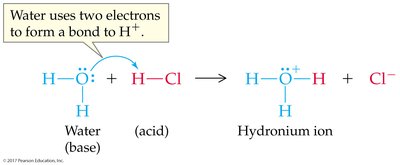
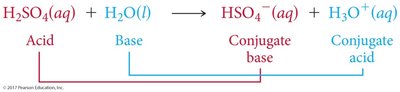
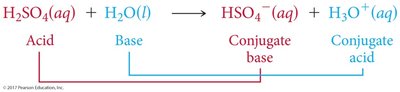
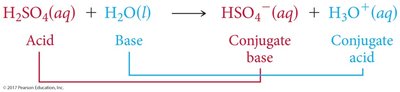
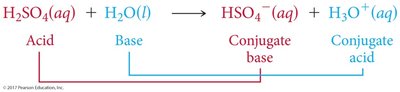
Brønsted-Lowry Acid: Proton donor; can donate H+ to another molecule or ion.
Brønsted-Lowry Base: Proton acceptor; must have a lone pair of electrons to accept H+.
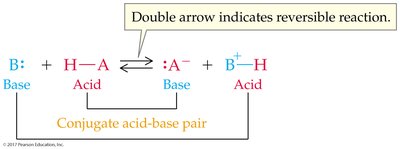
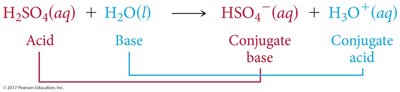
Conjugate acid-base pairs are species that differ by one proton (H+), found on opposite sides of a reaction.
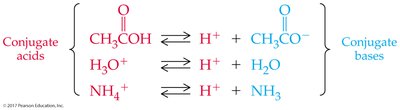
Acid and Base Strength
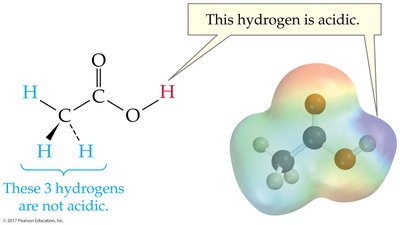
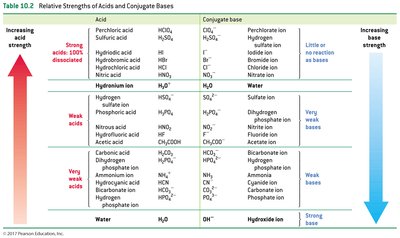
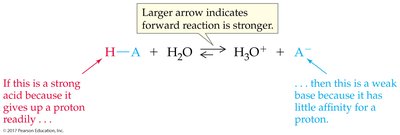
The strength of an acid or base is determined by its ability to donate or accept protons. Strong acids and bases dissociate completely in water, while weak acids and bases only partially dissociate.
Strong acids: Nearly 100% dissociated in water.
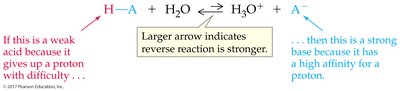
Weak acids: Less than 100% dissociated.
Strong bases: High affinity for protons.
Weak bases: Low affinity for protons.
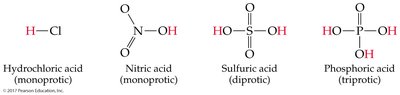
Polyprotic acids: Undergo stepwise dissociation; each successive dissociation is less favored.
Inverse relationship: The stronger the acid, the weaker its conjugate base, and vice versa.
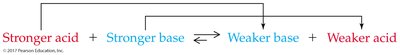
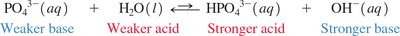
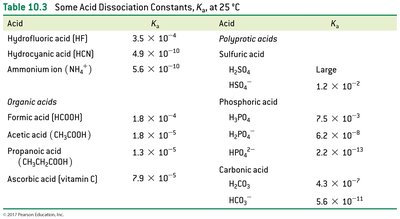
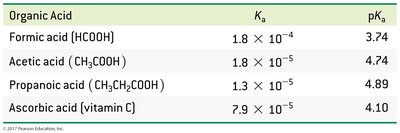
Acid Dissociation Constants (Ka)
The acid dissociation constant (Ka) quantifies the extent of dissociation for a weak acid in water. It is derived from the equilibrium constant for the reaction:
Ka expression:
Strong acids: Ka >> 1 (dissociation favored)
Weak acids: Ka << 1 (dissociation not favored)
Polyprotic acids: Successive Ka values decrease
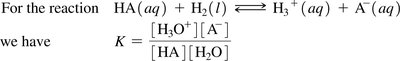
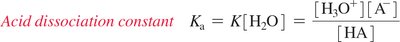
Water as Both an Acid and a Base (Amphoteric)
Water can act as both an acid and a base, a property called amphoterism. It can donate or accept protons depending on the reacting species.
Ion-product constant for water (Kw):
Acidic solution: [H3O+] > 10–7 M
Neutral solution: [H3O+] = 10–7 M
Basic solution: [H3O+] < 10–7 M

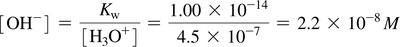
Measuring Acidity: The pH Scale
The pH scale is a logarithmic measure of the hydronium ion concentration in solution, ranging typically from 0 to 14.
pH formula:
Reverse formula:
Acidic: pH < 7
Neutral: pH = 7
Basic: pH > 7
pH + pOH = 14.00
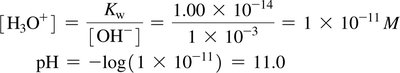
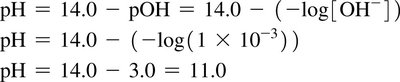

Working with pH
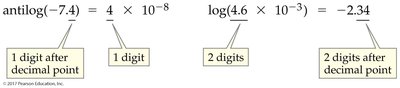
Conversions between pH and [H3O+] involve logarithms and antilogarithms. The number of significant figures in pH and [H3O+] is important for accurate calculations.
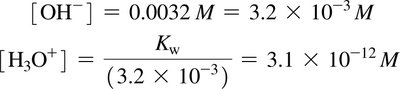
Acid and Base Equivalents
Acid and base equivalents are used to quantify the neutralization capacity of a solution. Normality (N) is the number of equivalents per liter.
Equivalent of acid: Amount containing 1 mole of H+
Equivalent of base: Amount containing 1 mole of OH–
Normality:
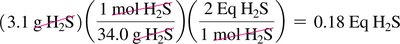
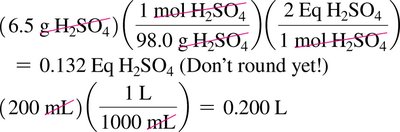
Common Acid-Base Reactions
Acids and bases participate in several important reactions, including neutralization, reactions with carbonates, and reactions with ammonia or amines.
Neutralization: Acid reacts with base to form water and salt.
Acids with carbonates: Produce CO2 gas and water.
Acids with ammonia/amines: Produce ammonium salts.
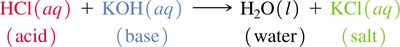
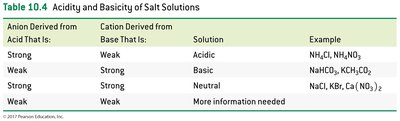
Acidity and Basicity of Salt Solutions
Salt solutions can be acidic, basic, or neutral depending on the ions present and their origins from strong or weak acids and bases.
Salt of strong acid + weak base: Acidic solution
Salt of weak acid + strong base: Basic solution
Salt of strong acid + strong base: Neutral solution
Salt of weak acid + weak base: pH depends on which ion reacts more with water
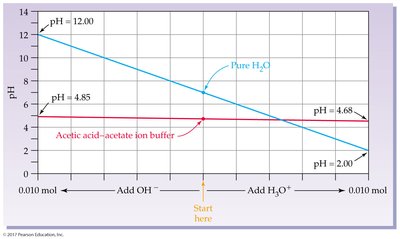
Buffer Solutions
Buffers are solutions that resist drastic changes in pH when small amounts of acid or base are added. They typically consist of a weak acid and its conjugate base.
Henderson-Hasselbalch equation:
Effective buffer: pKa close to desired pH, [HA]/[A–] ratio near 1, and buffer components much greater than added acid/base.
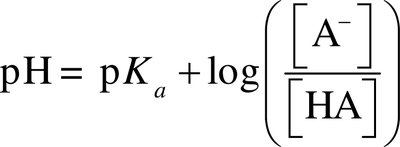

Titration
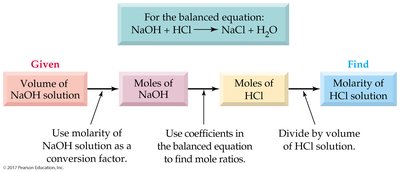
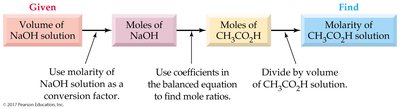
Titration is a technique used to determine the total acid or base concentration in a solution by reacting it with a solution of known concentration.
Neutralization reaction: One mole of acid reacts with one mole of base.
Molarity calculation:
Normality calculation:
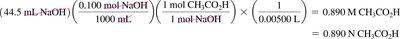
Concept Map
The concept map summarizes the relationships between acid-base definitions, strengths, and concentrations, as well as their determination and applications.