Back
BackAcids and Bases: Definitions, Strength, Equilibria, and Applications
Study Guide - Smart Notes

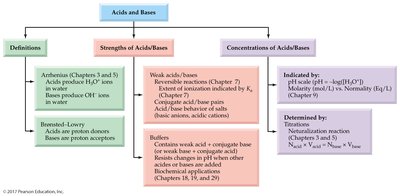
Acids and Bases: Definitions
Arrhenius and Brønsted-Lowry Definitions
Acids and bases are fundamental concepts in chemistry, defined by their behavior in aqueous solutions and their ability to donate or accept protons.
Arrhenius Acid: Produces hydronium ion (H3O+) when dissolved in water.
Arrhenius Base: Produces hydroxide ion (OH–) when dissolved in water.
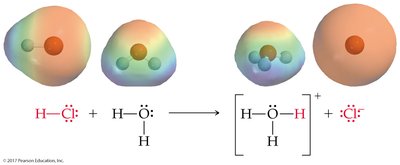
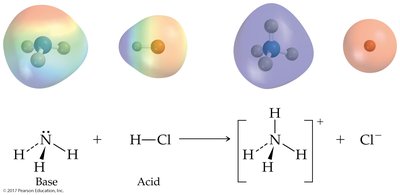
Brønsted-Lowry Acid: Proton donor; can donate H+ to another molecule or ion.
Brønsted-Lowry Base: Proton acceptor; must have a lone pair of electrons to accept H+.
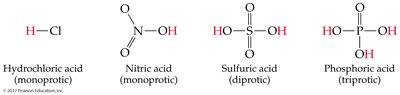
Polyprotic Acids
Acids can supply different numbers of protons:
Monoprotic: One proton (e.g., HCl)
Diprotic: Two protons (e.g., H2SO4)
Triprotic: Three protons (e.g., H3PO4)
Acidic Hydrogen Atoms
Acidic hydrogens are bonded to electronegative atoms, making them more likely to be donated as protons.
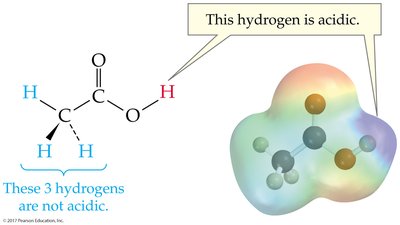
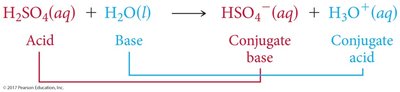
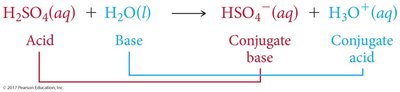
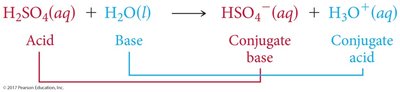
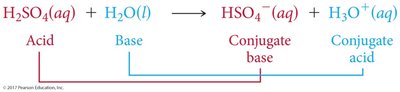
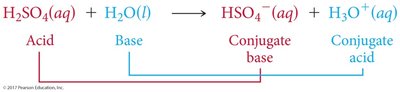
Acid-Base Reactions and Conjugate Pairs
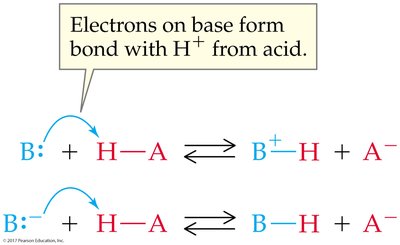
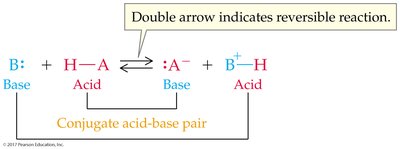
Acid-base reactions involve the transfer of a proton. The products can also behave as acids or bases, forming conjugate acid-base pairs.
Conjugate Acid: Formed when a base gains a proton.
Conjugate Base: Formed when an acid loses a proton.
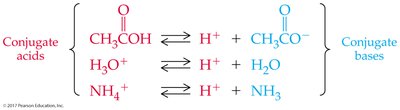
Acid and Base Strength
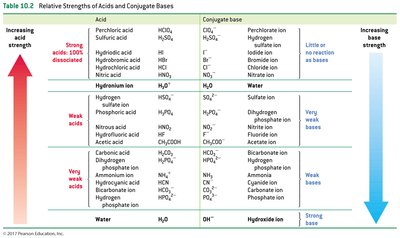
Strong vs. Weak Acids and Bases
The strength of acids and bases is determined by their ability to donate or accept protons.
Strong Acids: Completely dissociate in water; give up protons easily.
Weak Acids: Partially dissociate; give up protons with difficulty.
Strong Bases: Readily accept protons.
Weak Bases: Have little affinity for protons.
Relationship Between Acid and Base Strength
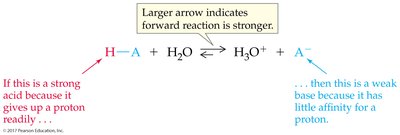
The stronger the acid, the weaker its conjugate base.
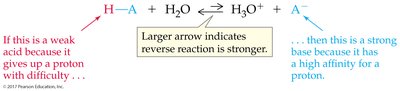
The weaker the acid, the stronger its conjugate base.
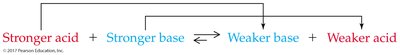
Predicting Proton-Transfer Equilibria
Equilibrium favors the reaction of the stronger acid and base, forming the weaker acid and base.
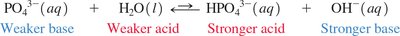
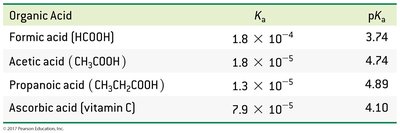
Acid Dissociation Constants (Ka)
Equilibrium Expression for Weak Acids
The dissociation of a weak acid in water is described by an equilibrium constant.
Acid Dissociation Constant (Ka): Predicts acid strength; higher Ka means stronger acid.
Equilibrium expression:
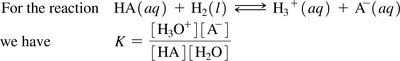
Acid dissociation constant:
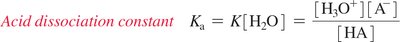
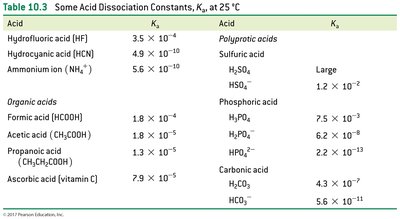
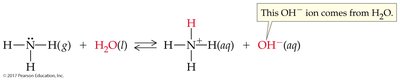
Water as Both an Acid and a Base
Amphoteric Nature of Water
Water can act as both an acid and a base, depending on the reaction partner.
Amphoteric: Substance that can react as either an acid or a base.

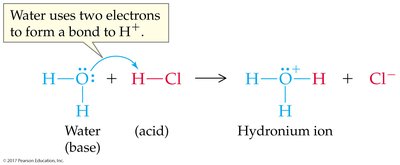
Ion-Product Constant for Water (Kw)
Water can dissociate to form hydronium and hydroxide ions. The ion-product constant is:
Acidic, Neutral, and Basic Solutions
Acidic: [H3O+] > 10–7 M, [OH–] < 10–7 M
Neutral: [H3O+] = [OH–] = 10–7 M
Basic: [H3O+] < 10–7 M, [OH–] > 10–7 M
Measuring Acidity: The pH Scale
pH and pOH Calculations
The pH scale is a logarithmic measure of acidity:

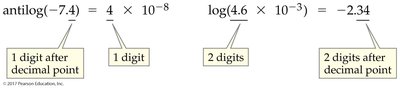
Significant Figures in pH Calculations
Logarithms and antilogarithms in pH calculations require careful attention to significant figures.
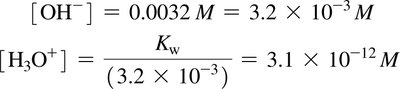
Acid and Base Equivalents
Normality and Equivalents
Normality (N) is the number of equivalents of acid or base per liter of solution.
Equivalent of acid: Amount containing 1 mole of H+
Equivalent of base: Amount containing 1 mole of OH–
Normality = Molarity × number of H+ or OH– ions per formula unit
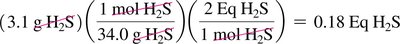
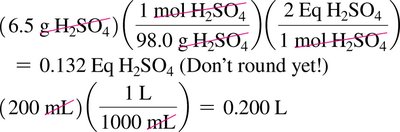
Common Acid-Base Reactions
Neutralization and Reactions with Carbonates
Acids react with hydroxide ions to form water and a salt.
Acids react with bicarbonate and carbonate ions to produce carbonic acid, which decomposes to CO2 and water.
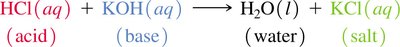
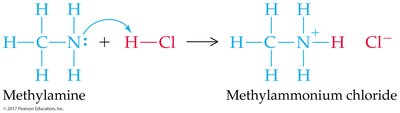
Reactions with Ammonia and Amines
Acids react with ammonia to yield ammonium salts.
Amines (nitrogen-containing compounds) react similarly, forming water-soluble salts.
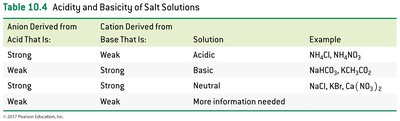
Acidity and Basicity of Salt Solutions
Classification of Salt Solutions
Salt solutions can be acidic, basic, or neutral depending on the ions present.
Salt of strong acid + weak base: Acidic
Salt of weak acid + strong base: Basic
Salt of strong acid + strong base: Neutral
Salt of weak acid + weak base: pH depends on relative strengths
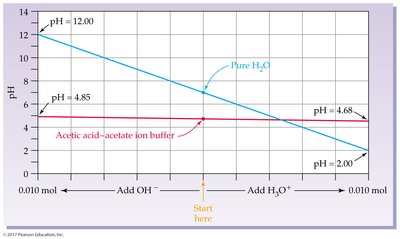
Buffer Solutions
Buffer Definition and Function
Buffers are combinations of substances (usually a weak acid and its conjugate base) that prevent drastic changes in pH.
If OH– is added, the acid component neutralizes it.
If H+ is added, the base component neutralizes it.
Henderson-Hasselbalch Equation
The pH of a buffer solution is calculated using:
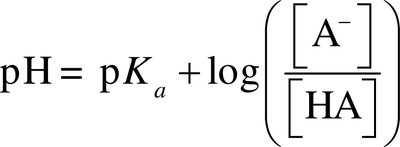

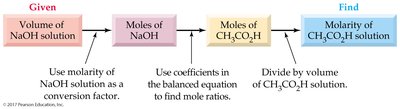
Titration
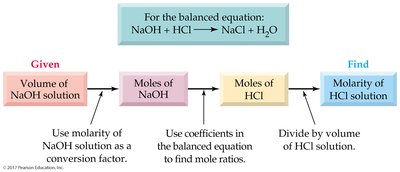
Acid-Base Titration Procedure
Titration is used to determine the total acid or base concentration in a solution.
A measured volume of acid is titrated with a base of known concentration until neutralization is complete (end point).
For 1:1 neutralization:
For reactions with different coefficients:
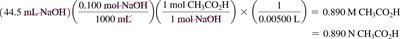
Concept Map