 Back
BackAcids and Bases: Polyfunctional Acids, Acid Strength, and Chemical Structure
Study Guide - Smart Notes

Acids and Bases
Polyfunctional Acids and Bases
Polyfunctional acids and bases are important in general chemistry due to their ability to donate or accept multiple protons. Understanding their dissociation behavior and calculations is essential for predicting solution pH and species concentrations.
Polyfunctional Acids: These acids can donate two or more protons and are often symbolized as HnA, where n is the number of ionizable protons. Examples include phosphoric acid (H3PO4), maleic acid (C2H2(COOH)2), and carbonic acid (H2CO3).
Polyfunctional Bases: These can either have more than one nitrogen-containing functional group (with available lone pairs) or be conjugate bases of polyprotic weak acids, capable of accepting up to n protons. Examples include ethylenediamine and phosphate ion (PO43−).
In water, polyfunctional acids dissociate stepwise, each step having its own dissociation constant (Ka). For example, phosphoric acid dissociates in three steps:
Step 1: Ka1 =
Step 2: Ka2 =
Step 3: Ka3 =
Generally, the first dissociation constant is much larger than the subsequent ones because the first proton is removed from a neutral molecule, while later protons are removed from increasingly negatively charged ions.
Calculations with Polyprotic Acids: If successive Ka values differ by a factor of 103 or more, pH calculations can often be simplified by considering only the first dissociation step, similar to monoprotic acids.
Example: Ascorbic Acid (H2C6H6O6)
First dissociation: Ka1 =
Second dissociation: Ka2 =
Since Ka1 >> Ka2, only the first dissociation is considered for pH calculation. The ICE table method is used to solve for [H3O+], and the assumption of negligible x is checked for validity.
Example: Oxalic Acid (H2C2O4)
First dissociation: Ka1 =
Second dissociation: Ka2 =
When the assumption of negligible x is not valid, the quadratic equation must be used to solve for [H3O+].
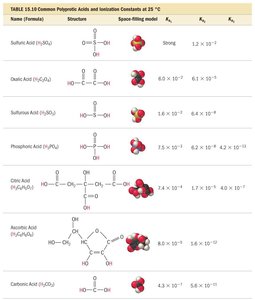
Key Table: Common Polyprotic Acids and Ionization Constants
Name (Formula) | Structure | Ka1 | Ka2 | Ka3 |
|---|---|---|---|---|
Sulfuric Acid (H2SO4) | Strong | 1.2 × 100 | 2.4 × 10−2 | — |
Oxalic Acid (H2C2O4) | — | 6.7 × 10−2 | 6.4 × 10−5 | — |
Phosphoric Acid (H3PO4) | — | 7.5 × 10−3 | 6.2 × 10−8 | 4.2 × 10−13 |
Ascorbic Acid (H2C6H6O6) | — | 8.5 × 10−5 | 1.6 × 10−12 | — |
Carbonic Acid (H2CO3) | — | 4.3 × 10−7 | 4.8 × 10−11 | — |
Acid Strength and Chemical Structure
Binary Acids
Binary acids consist of hydrogen and one other element (HY, H2Y, H3Y, H4Y). Their acidity is influenced by the electronegativity and size of the other element (Y).
Electronegativity: As Y becomes more electronegative (across a period), acidity increases.
Bond Strength: As the bond strength decreases (down a group), acidity increases.
For hydrides of Groups 6A and 7A, acidity increases both with increasing electronegativity and decreasing bond strength.

Bond energy is a key factor in determining acid strength:
Bond | Bond Energy (kJ/mol) | Acid Strength (aqueous solution) |
|---|---|---|
H-F | 565 | Weak |
H-Cl | 427 | Strong |
H-Br | 363 | Strong |
H-I | 295 | Strong |
Oxyacids (Oxoacids)
Oxyacids have a hydrogen atom bonded to an oxygen atom, which is bonded to a central non-metal atom. Their acid strength is influenced by the electronegativity of the central atom and the number of oxygen atoms attached.
Electronegativity: For oxyacids with the same number of oxygen atoms, a more electronegative central atom increases acid strength.
Number of Oxygen Atoms: For oxyacids with different numbers of oxygen atoms, acid strength increases with more oxygen atoms.
Examples:
Acid Series | # O atoms | Ka | Acid Type |
|---|---|---|---|
HOClO3 | 4 | 1.0 × 107 | Strong |
HOClO2 | 3 | 1.0 | Strong |
HOClO | 2 | 1.1 × 10−2 | Weak |
HOCl | 1 | 2.9 × 10−8 | Weak |
Carboxylic Acids
Carboxylic acids are organic acids with the general formula RCOOH, where R is the rest of the molecule. The strength of a carboxylic acid depends on the nature of the R group.
Electronegative Substituents: Electronegative atoms (e.g., Cl) in the R group pull electron density toward themselves, making the O-H bond more polar and the H easier to ionize, thus increasing acid strength.
Ka Values: Chloroacetic acid (R = CH2Cl) has a higher Ka than acetic acid (R = CH3), indicating stronger acidity.
Structures:


Comparison Table:
Acid Name | R Identity | Ka |
|---|---|---|
Acetic acid | CH3 | 1.8 × 10−5 |
Chloroacetic acid | CH2Cl | 1.4 × 10−3 |
----------------------------------------
