 Back
BackAcids and Bases: Properties, Definitions, and Calculations
Study Guide - Smart Notes

Chapter 17: Acids and Bases
Properties of Acids
Acids are a class of compounds with distinct chemical and physical properties. They are commonly found in foods, industrial processes, and biological systems.
Sour taste (e.g., citrus fruits contain citric acid)
Ability to dissolve many metals (e.g., hydrochloric acid dissolves zinc)
Ability to neutralize bases (react with bases to form water and salts)
Change blue litmus paper to red
Common Acids and Their Uses
Acids are widely used in industry, food, and biological systems. Some common examples include:
Name | Formula | Occurrence / Uses |
|---|---|---|
Hydrochloric acid | HCl | Metal cleaning, food preparation, stomach acid |
Sulfuric acid | H2SO4 | Fertilizer, explosives, batteries |
Nitric acid | HNO3 | Fertilizer, explosives, dyes |
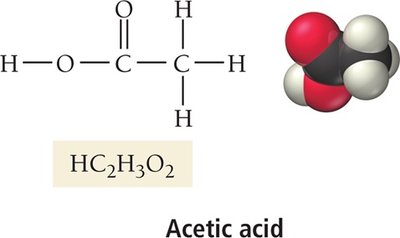
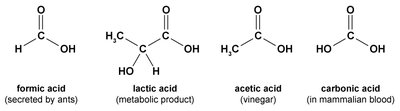
Acetic acid | HC2H3O2 | Vinegar, food preservation |
Citric acid | H3C6H5O7 | Citrus fruits, pH adjustment in foods |
Carbonic acid | H2CO3 | Carbonated beverages |
Hydrofluoric acid | HF | Glass etching, metal cleaning |
Phosphoric acid | H3PO4 | Fertilizers, soft drinks |

Structures of Acids
Binary acids: Hydrogen attached to a nonmetal (e.g., HCl, HF)
Oxyacids: Hydrogen attached to an oxygen atom (e.g., H2SO4, HNO3)
Carboxylic acids: Contain the carboxyl group (–COOH); only the hydrogen in the –COOH is acidic (e.g., acetic acid, citric acid)
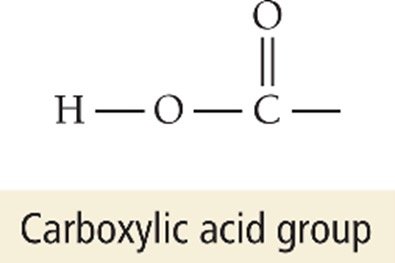
Properties of Bases
Bases are substances with characteristic properties and important industrial and household uses.
Bitter taste
Often poisonous
Slippery feel (e.g., soap)
Ability to neutralize acids
Turn red litmus paper blue

Common Bases and Their Uses
Name | Formula | Occurrence / Uses |
|---|---|---|
Sodium hydroxide | NaOH | Soap, plastic manufacturing, petroleum processing |
Potassium hydroxide | KOH | Soap, batteries, cotton processing |
Sodium bicarbonate | NaHCO3 | Baking soda, antacid |
Sodium carbonate | Na2CO3 | Glass, soap manufacturing |
Ammonia | NH3 | Fertilizer, cleaning agent |
Definitions of Acids and Bases
Arrhenius Model
The Arrhenius definition is the earliest and most restrictive model for acids and bases:
Acid: Produces H+ ions in aqueous solution
Base: Produces OH− ions in aqueous solution
Example reaction:
Brønsted–Lowry Theory
This model expands the definition to include proton transfer reactions:
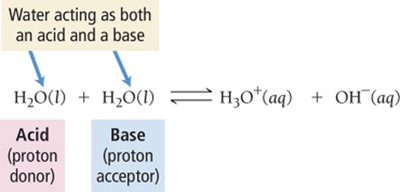
Acid: Proton (H+) donor
Base: Proton (H+) acceptor
Amphoteric substances: Can act as either acid or base (e.g., H2O)
Example:
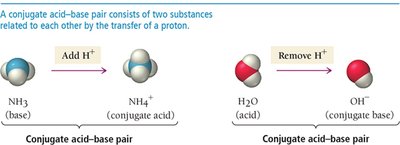
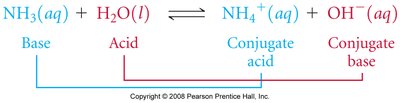
Conjugate Acid–Base Pairs
In Brønsted–Lowry reactions, each acid and base forms a conjugate pair:
Conjugate acid: Formed when a base gains a proton
Conjugate base: Formed when an acid loses a proton
Acid and Base Strength
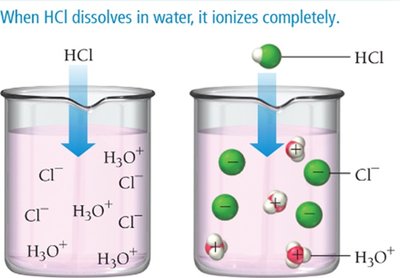
Strong and Weak Acids
Strong acids: Completely ionize in water (e.g., HCl, HNO3, H2SO4)
Weak acids: Only partially ionize in water (e.g., HF, acetic acid)
Acid Ionization Constant (Ka)
The strength of an acid is measured by its acid ionization constant, Ka:
Larger Ka = stronger acid
Smaller Ka = weaker acid
Autoionization of Water and pH
Water can act as both an acid and a base, leading to autoionization:
The ion product of water is:
at 25°C
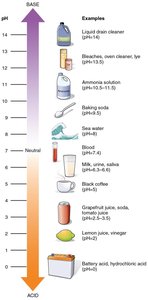
pH and pOH
pH:
pOH:
At 25°C,
pH < 7: acidic; pH = 7: neutral; pH > 7: basic
Calculating pH of Strong and Weak Acids
For strong acids:
For weak acids: Use an ICE table and solve for equilibrium concentrations using Ka
Example for weak acid:
Set up ICE table, solve for x, then
Percent Ionization
Percent ionization measures the fraction of acid molecules that ionize:
Strong acids: >99%
Weak acids: <5%
Acid–Base Properties of Salts
Salts in Solution
Cations from strong bases and anions from strong acids yield neutral solutions (e.g., NaCl)
Anions from weak acids yield basic solutions (e.g., NaF)
Cations from weak bases yield acidic solutions (e.g., NH4Cl)
Highly charged metal cations can also make solutions acidic (e.g., Al(NO3)3)
Relationship Between Ka and Kb
For a conjugate acid–base pair:
Polyprotic Acids
Polyprotic acids can donate more than one proton, with each step having its own Ka value:
Molecular Structure and Acid Strength
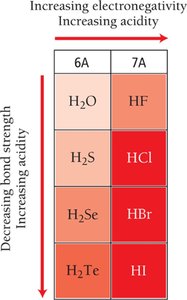
Binary Acids
Acid strength increases across a period (left to right) and down a group (top to bottom) in the periodic table
More polarized and weaker H–X bonds lead to stronger acids
Oxyacids
Acid strength increases with the number of oxygen atoms bonded to the central atom
Greater electronegativity of the central atom also increases acidity

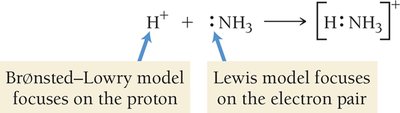
Lewis Acid–Base Theory
The Lewis definition broadens the concept of acids and bases:
Lewis acid: Electron pair acceptor
Lewis base: Electron pair donor
All Brønsted–Lowry acids are Lewis acids, but not all Lewis acids are Brønsted–Lowry acids.
Acid Rain and Environmental Impact
Combustion of fossil fuels produces nonmetal oxides (CO2, SO2, NO2) that dissolve in water to form acids, contributing to acid rain. Acid rain damages buildings, aquatic life, and soil quality.

