 Back
BackAcids and Bases: Properties, Definitions, Equilibria, and pH Calculations
Study Guide - Smart Notes

Acids and Bases
Properties of Acids and Bases
Acids and bases are fundamental chemical species with distinct properties that are easily observed in laboratory and everyday contexts.
Acids: Sour taste, ability to dissolve metals, turn litmus paper red, neutralize bases.
Bases: Bitter taste, slippery feel, turn litmus paper blue, neutralize acids.
Definitions of Acids and Bases
There are several definitions for acids and bases, each with its own scope and application:
Arrhenius Definition: An acid produces H+ ions in aqueous solution; a base produces OH– ions in aqueous solution.
Brønsted–Lowry Definition: An acid donates H+ in a chemical reaction; a base accepts H+. This definition is more broadly applicable.
Lewis Definition: (To be covered later) Focuses on electron pair donation and acceptance.
Acid Dissociation and Conjugate Pairs
Acids dissociate in water, forming conjugate acid-base pairs. For example:
CH3COOH(aq) + H2O(l) → CH3COO–(aq) + H3O+(aq)
Acid (donates H+): CH3COOH
Base (accepts H+): H2O
Conjugate base: CH3COO–
Conjugate acid: H3O+
The weaker the acid, the stronger its conjugate base.
Acid and Base Strength
Acid Ionization Constant (Ka)
The equilibrium constant for acid dissociation is called the acid ionization constant (Ka):
General reaction: HA(aq) + H2O(l) → A–(aq) + H3O+(aq)
Ka expression:
The stronger the acid, the larger the Ka value, and the equilibrium favors products.
Strong vs. Weak Acids
Strong acids: Dissociate completely in water; equilibrium lies far to the right. Example: HCl(aq) + H2O(l) → Cl–(aq) + H3O+(aq)
Weak acids: Partially dissociate; equilibrium exists and Ka is used to quantify strength.
Base Ionization Constant (Kb)
For bases, the equilibrium constant is called Kb:
General reaction: B(aq) + H2O(l) → BH+(aq) + OH–(aq)
Kb expression:
The larger the Kb, the stronger the base.
Relationship Between Ka and Kb
For a conjugate acid-base pair:
At 25°C,
If Ka is known, Kb can be calculated for the conjugate base, and vice versa.
Autoionization of Water and the Ion-Product Constant (Kw)
Autoionization Reaction
Water can act as both an acid and a base (amphoteric). The autoionization reaction is:
2H2O(l) → H3O+(aq) + OH–(aq)
Equilibrium constant:
At 25°C,
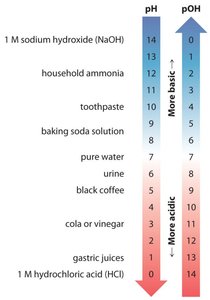
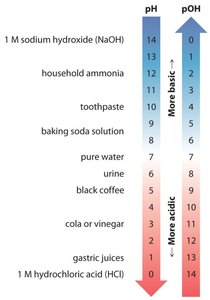
Acidic, Basic, and Neutral Solutions
If [H3O+] > [OH–], the solution is acidic.
If [H3O+] < [OH–], the solution is basic.
If [H3O+] = [OH–], the solution is neutral.
Concentrations in Pure Water
In pure water at 25°C: M
The pH and pOH Scales
Definition and Calculation
The pH scale is a logarithmic scale used to quantify acidity:
At 25°C: pH < 7.0 is acidic, pH = 7.0 is neutral, pH > 7.0 is basic.
pOH Scale
pOH is defined similarly:
Relationship Between pH and pOH
At 25°C:
General relationship:
pKa and pKb
Percent Ionization of Weak Acids
Definition and Calculation
Percent ionization quantifies the fraction of a weak acid that dissociates in water:
Example: For Ka = , [HA]in = 0.600 M, [H3O+]eq = M:
Effect of Concentration
As the concentration of a weak acid increases, percent ionization typically decreases.
Calculating pH from Ka
Procedure
To find the pH of a weak acid solution:
Write the balanced dissociation reaction.
Write the Ka expression.
Create an equilibrium table (ICE table).
Solve for [H3O+] using the Ka expression.
Check approximation validity or use the quadratic formula if necessary.
Calculate pH:
Example: For HNO2 with Ka = and [HNO2]initial = 0.200 M, pH = 2.02.
Summary Table: Acid/Base Strength and Equilibrium Constants
Type | Equilibrium Constant | Strength Indicator |
|---|---|---|
Acid | Ka | Larger Ka = Stronger Acid |
Base | Kb | Larger Kb = Stronger Base |
Conjugate Pair | Kw = Ka × Kb | Kw = 1.0 × 10–14 at 25°C |
Additional info: The notes above expand on the original lecture slides, providing full academic context, definitions, and stepwise procedures for calculations. All equations are formatted in LaTeX as required.
