 Back
BackAcids and Bases: Properties, Definitions, Equilibria, and pH Calculations
Study Guide - Smart Notes

Acids and Bases
Properties of Acids and Bases
Acids and bases are fundamental chemical species with distinct properties that are easily observed in laboratory and everyday contexts.
Acids: Sour taste, ability to dissolve metals, turn litmus paper red, neutralize bases.
Bases: Bitter taste, slippery feel, turn litmus paper blue, neutralize acids.
Definitions of Acids and Bases
There are several definitions for acids and bases, each with its own scope and application:
Arrhenius Definition: An acid produces H+ ions in aqueous solution; a base produces OH– ions in aqueous solution.
Brønsted–Lowry Definition: An acid donates H+ in a chemical reaction; a base accepts H+. This definition is more broadly applicable.
Lewis Definition: (To be covered later) Focuses on electron pair donation and acceptance.
Acid Dissociation and Conjugate Pairs
Acids dissociate in water, forming conjugate acid-base pairs. For example:
CH3COOH(aq) + H2O(l) → CH3COO–(aq) + H3O+(aq)
Acid (donates H+): CH3COOH
Base (accepts H+): H2O
Conjugate base: CH3COO–
Conjugate acid: H3O+
The weaker the acid, the stronger its conjugate base.
Acid and Base Strength
Acid Ionization Constant (Ka)
The equilibrium constant for acid dissociation is called the acid ionization constant, Ka:
Strong acids: Equilibrium lies far to the right; reaction goes to completion. Ka is not discussed for strong acids.
Weak acids: Equilibrium exists; Ka is used to quantify strength.
Stronger acid: Larger Ka value.
Base Ionization Constant (Kb)
Bases also have an equilibrium constant, Kb:
Stronger base: Larger Kb value.
Relationship Between Ka and Kb
For a conjugate acid-base pair:
Where is the ion-product constant for water.
Autoionization of Water and Kw
Autoionization Reaction
Water can act as both an acid and a base (amphoteric). When two water molecules react:
The equilibrium constant is:
At 25°C, .
If , the solution is acidic.
If , the solution is basic.
If , the solution is neutral.
Hydronium and Hydroxide Concentrations in Pure Water
In pure water at 25°C:
The pH and pOH Scales
pH Scale
The pH scale is a logarithmic scale used to quantify acidity:
pH < 7.0: Acidic
pH = 7.0: Neutral
pH > 7.0: Basic
pOH Scale
pOH is defined similarly:
Relationship between pH and pOH:
(at 25°C)
Converting Between pH, pOH, [H3O+], and [OH–]
Given one value, others can be calculated using and logarithmic relationships.
pKa and pKb
pKa and pKb Definitions
pKa and pKb are logarithmic measures of Ka and Kb:
Lower pKa means stronger acid.
Percent Ionization of Weak Acids
Definition and Calculation
Percent ionization quantifies how much a weak acid dissociates:
As acid concentration increases, percent ionization typically decreases.
Example Calculation
For a weak acid with and , :
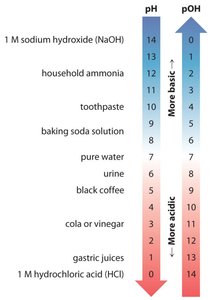
Summary Table: pH and pOH Values for Common Substances
Substance | pH | pOH |
|---|---|---|
1 M NaOH | 14 | 0 |
Household ammonia | 11 | 3 |
Toothpaste | 9 | 5 |
Baking soda solution | 8 | 6 |
Pure water | 7 | 7 |
Urine | 6 | 8 |
Black coffee | 5 | 9 |
Cola or vinegar | 3 | 11 |
Gastric juices | 1 | 13 |
1 M HCl | 0 | 14 |

Practice and Application
Write balanced acid-base reactions and identify conjugate pairs.
Use equilibrium tables and Ka/Kb expressions to solve for unknown concentrations.
Calculate pH, pOH, and percent ionization for strong and weak acids.
Example: For HNO2 with Ka = 4.6 x 10–4 and [HNO2]initial = 0.200 M, pH = 2.02.
Additional info: The notes cover all major aspects of acid-base chemistry relevant to a general chemistry course, including equilibrium, calculations, and conceptual understanding.
