 Back
BackAcids, Bases, Buffers, and Titration – General Chemistry Study Guide
Study Guide - Smart Notes

Q1. Buffer Solution and HCl Addition
Background
Topic: Buffers and Acid-Base Reactions
This question tests your understanding of how buffer solutions resist changes in pH when a strong acid is added.
Key Terms:
Buffer: A solution that resists changes in pH when small amounts of acid or base are added.
Conjugate Acid-Base Pair: The acid and base that differ by one proton (H+).
Step-by-Step Guidance
Recall that a buffer is made from a weak acid and its conjugate base (or vice versa).
When HCl (a strong acid) is added, it provides extra H+ ions to the solution.
Think about which component of the buffer will react with the added H+ to minimize the pH change.
Consider the chemical equation for the buffer system:
Try solving on your own before revealing the answer!
Q2. Calculate the pH of a Carbonic Acid/Sodium Bicarbonate Solution
Background
Topic: Buffer Calculations (Henderson-Hasselbalch Equation)
This question tests your ability to calculate the pH of a buffer solution using the concentrations of a weak acid and its conjugate base.
Key Formula:
Henderson-Hasselbalch Equation:
= concentration of sodium bicarbonate ()
= concentration of carbonic acid ()
= acid dissociation constant for
Step-by-Step Guidance
Calculate from the given value:
Identify the concentrations: ,
Plug these values into the Henderson-Hasselbalch equation.
Set up the expression for but do not calculate the final value yet.
Try solving on your own before revealing the answer!
Q3. pH at the Equivalence Point of a Weak Acid–Strong Base Titration
Background
Topic: Titration Curves and Equivalence Point Calculations
This question tests your understanding of how to determine the pH at the equivalence point when titrating a weak acid with a strong base.
Key Concepts:
At the equivalence point, all the weak acid has been neutralized to form its conjugate base.
The pH is determined by the hydrolysis of the conjugate base.
Key Formula:
To find pH: Calculate at equivalence, then use for :
Step-by-Step Guidance
Calculate the moles of acetic acid and NaOH used at equivalence.
Determine the total volume of the solution at equivalence.
Find the concentration of formed.
Set up the hydrolysis equation for and write the expression for .
Try solving on your own before revealing the answer!
Q4. Destroying a Buffer Solution
Background
Topic: Buffer Capacity and Buffer Destruction
This question tests your understanding of what can destroy a buffer solution by overwhelming its capacity.
Key Concepts:
A buffer is destroyed if enough strong acid or base is added to react completely with one component of the buffer.
Step-by-Step Guidance
Recall the buffer components: (acid) and (base).
Consider what happens if you add an amount of strong acid or base equal to the amount of one buffer component.
Think about which addition would neutralize all of either the acid or base in the buffer, leaving only one component.
Try solving on your own before revealing the answer!
Q5. Volume of KOH Required for Neutralization
Background
Topic: Stoichiometry of Acid-Base Neutralization
This question tests your ability to use stoichiometry to determine the volume of base needed to neutralize a given amount of acid.
Key Formula:
(for monoprotic acid and base)
= molarity of acid
= volume of acid
= molarity of base
= volume of base (what you're solving for)
Step-by-Step Guidance
Write the balanced equation:
Set up the equation using the given values.
Rearrange to solve for (volume of KOH needed).
Try solving on your own before revealing the answer!
Q6. pH at the Equivalence Point of a Weak Acid–Strong Base Titration
Background
Topic: Titration Curves
This question tests your understanding of the pH at the equivalence point for different types of titrations.
Key Concept:
At the equivalence point, the weak acid has been converted to its conjugate base, which hydrolyzes to make the solution basic.
Step-by-Step Guidance
Recall what happens to the conjugate base of a weak acid in water.
Think about whether the resulting solution will be acidic, neutral, or basic.
Try solving on your own before revealing the answer!
Q7. Strongest Acid Identification
Background
Topic: Acid Strength
This question tests your ability to compare the strengths of different acids based on their molecular structure and position in the periodic table.
Key Concept:
Acid strength increases with the ability to donate a proton easily; for binary acids, this generally increases down a group.
Step-by-Step Guidance
Recall the trend for acid strength among hydrides (e.g., HF, H2O, NH3, CH4).
Identify which molecule is most likely to donate a proton (H+).
Try solving on your own before revealing the answer!
Q8. Strongest Base Identification
Background
Topic: Base Strength
This question tests your understanding of how to compare the strengths of different bases.
Key Concept:
The strongest base is the one most likely to accept a proton (H+).
Step-by-Step Guidance
Recall the relationship between conjugate acids and bases.
Consider which ion is the conjugate base of the weakest acid.
Try solving on your own before revealing the answer!
Q9. Ranking Acid Strengths
Background
Topic: Acid Strength Trends
This question tests your ability to rank hydrohalic acids (HI, HBr, HCl, HF) from strongest to weakest.
Key Concept:
For binary acids, acid strength increases as bond strength decreases (down the group in the periodic table).
Step-by-Step Guidance
Recall the trend for acid strength among the halogen hydrides.
Arrange the acids in order from strongest to weakest based on periodic trends.
Try solving on your own before revealing the answer!
Q10. Conjugate Acid–Base Pair Identification
Background
Topic: Conjugate Acid–Base Pairs
This question tests your ability to identify pairs that differ by one proton (H+).
Key Concept:
A conjugate acid–base pair consists of two species that differ by a single proton.
Step-by-Step Guidance
For each pair, determine if one is the acid form and the other is the base form (differing by H+).
Eliminate pairs that do not fit this definition.
Try solving on your own before revealing the answer!
Q11. Acid and Conjugate Base Strength Relationship
Background
Topic: Acid-Base Strength Relationships
This question tests your understanding of the relationship between the strength of an acid and its conjugate base.
Key Concept:
The stronger the acid, the weaker its conjugate base, and vice versa.
Step-by-Step Guidance
Recall the inverse relationship between acid and conjugate base strength.
Identify which statement correctly describes this relationship.
Try solving on your own before revealing the answer!
Q12. Effect of Adding NH₄Cl to NH₃ Solution
Background
Topic: Common Ion Effect and Buffer Solutions
This question tests your understanding of how adding a salt containing a common ion affects the pH of a buffer solution.
Key Concept:
Adding a salt with a common ion shifts the equilibrium and affects the pH.
Step-by-Step Guidance
Identify the common ion between NH3 and NH4Cl.
Predict how adding NH4Cl will shift the equilibrium and affect the pH.
Try solving on your own before revealing the answer!
Q13. Effect of Adding NaF to HF Solution
Background
Topic: Common Ion Effect
This question tests your understanding of how adding a salt containing the conjugate base affects the pH of a weak acid solution.
Key Concept:
Adding NaF increases the concentration of F-, shifting the equilibrium and affecting the pH.
Step-by-Step Guidance
Write the equilibrium for HF dissociation.
Predict how adding F- (from NaF) will shift the equilibrium and affect the pH.
Try solving on your own before revealing the answer!
Q14. pH of a NaF Solution
Background
Topic: Salt Hydrolysis
This question tests your understanding of whether a salt solution is acidic, basic, or neutral.
Key Concept:
NaF is the salt of a strong base (NaOH) and a weak acid (HF), so its solution will be basic.
Step-by-Step Guidance
Recall the hydrolysis reaction for F- in water.
Predict whether the solution will be acidic, basic, or neutral.
Try solving on your own before revealing the answer!
Q15. Salt Producing a Basic Solution
Background
Topic: Salt Hydrolysis
This question tests your ability to predict whether a salt will produce an acidic, basic, or neutral solution.
Key Concept:
A salt formed from a weak acid and strong base will produce a basic solution.
Step-by-Step Guidance
Identify the acid and base from which each salt is derived.
Determine which salt will hydrolyze to produce a basic solution.
Try solving on your own before revealing the answer!
Q16. Brønsted-Lowry Acid and Conjugate Base Identification
Background
Topic: Brønsted-Lowry Acid-Base Theory
This question tests your ability to identify the acid and its conjugate base in a chemical reaction.
Key Concept:
The acid donates a proton; the conjugate base is what remains after the acid donates a proton.
Step-by-Step Guidance
Identify which species donates a proton in the reaction.
Determine which species is the conjugate base (the product after proton donation).
Try solving on your own before revealing the answer!
Q17. Lewis Acid and Conjugate Base Identification
Background
Topic: Lewis Acid-Base Theory
This question tests your understanding of Lewis acids (electron pair acceptors) and their conjugate bases.
Key Concept:
Lewis acids accept electron pairs; Lewis bases donate electron pairs.
Step-by-Step Guidance
Identify which species acts as the electron pair acceptor (Lewis acid).
Determine which species is the conjugate base after the acid accepts an electron pair.
Try solving on your own before revealing the answer!
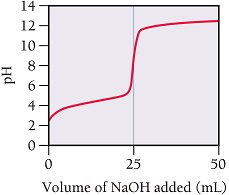
Q18. Determining from a Titration Curve
Background
Topic: Titration Curves and Weak Acid Dissociation Constant
This question tests your ability to use a titration curve to determine the of a weak acid.
Key Concepts and Steps:
The pH at the half-equivalence point equals the of the weak acid.
Step-by-Step Guidance
Locate the equivalence point on the titration curve (where the steepest rise occurs).
Find the volume at the equivalence point (from the graph, it's where the vertical jump is centered).
Determine the half-equivalence point (half the volume to equivalence).
Read the pH at the half-equivalence point from the graph. This pH equals the of the acid.
Try solving on your own before revealing the answer!
