 Back
BackCh. 19
Study Guide - Smart Notes

Proteins and Amino Acids
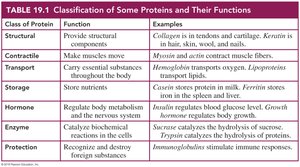
Functions and Importance of Proteins
Proteins are essential biomolecules that perform a wide variety of functions in living organisms. They serve as structural components, enzymes, transporters, hormones, and more. The diversity of protein function is a direct result of the variety and sequence of amino acids that compose them.
Structural proteins provide support and shape to cells and tissues.
Enzymatic proteins catalyze biochemical reactions.
Transport proteins carry substances throughout the body.
Hormonal proteins regulate physiological processes.
Protective proteins defend against foreign substances.
Amino Acids: Structure and Properties
General Structure of Amino Acids
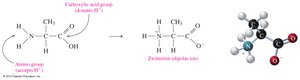
Amino acids are the building blocks of proteins. Each amino acid contains a central (alpha) carbon atom bonded to an amino group (−NH2), a carboxylic acid group (−COOH), a hydrogen atom, and a variable side chain (R group) that determines its unique properties. At physiological pH, amino acids exist as zwitterions, with the amino group protonated (−NH3+) and the carboxyl group deprotonated (−COO−).
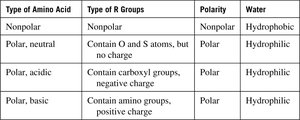
Classification of Amino Acids
Amino acids are classified based on the properties of their R groups:
Nonpolar (hydrophobic): R group is a hydrocarbon or aromatic group.
Polar, neutral (hydrophilic): R group contains O or S atoms but no charge.
Polar, acidic: R group contains a carboxyl group (negative charge).
Polar, basic: R group contains an amino group (positive charge).
Examples of Amino Acid Structures
The 20 common amino acids differ in their R groups, which confer specific chemical properties. These differences are crucial for protein structure and function.

Peptides and Protein Structure
Peptide Bond Formation
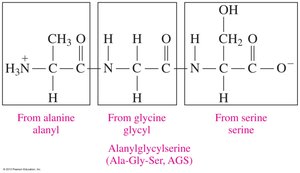
A peptide bond is an amide linkage formed between the carboxyl group of one amino acid and the amino group of another. This bond is the foundation of protein primary structure.
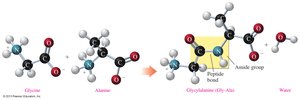
Naming Peptides
Peptides are named by listing the amino acid residues from the N-terminal (free amino group) to the C-terminal (free carboxyl group), with the N-terminal residue ending in "-yl" and the C-terminal residue retaining its full name.
Levels of Protein Structure
Primary Structure
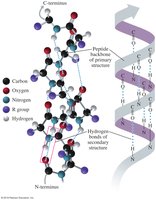
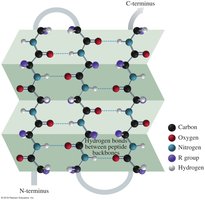
The primary structure of a protein is the unique sequence of amino acids in a polypeptide chain. This sequence determines all higher levels of protein structure and ultimately its function.
Secondary Structure
The secondary structure refers to local folding patterns stabilized by hydrogen bonds between backbone atoms. The two main types are:
Alpha helix (α-helix): A right-handed coil stabilized by hydrogen bonds between every fourth amino acid.
Beta-pleated sheet (β-sheet): Extended strands connected by hydrogen bonds, forming sheet-like structures.
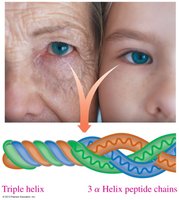
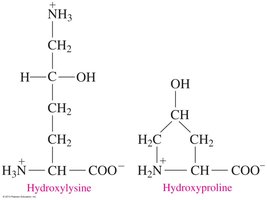
Triple Helix
A triple helix consists of three polypeptide chains woven together, commonly found in collagen. This structure is stabilized by hydrogen bonds involving hydroxyproline and hydroxylysine residues.
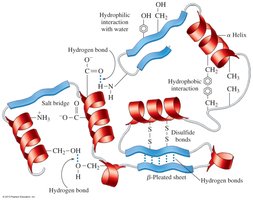
Tertiary Structure
The tertiary structure is the overall three-dimensional shape of a single polypeptide chain, stabilized by interactions among R groups, including:
Hydrophobic interactions
Hydrophilic interactions
Salt bridges (ionic bonds)
Hydrogen bonds
Disulfide bonds

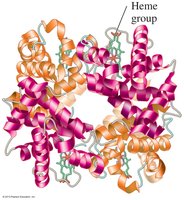
Quaternary Structure
The quaternary structure is the arrangement of two or more polypeptide subunits in a functional protein. Subunits are held together by the same types of interactions as in tertiary structure. Hemoglobin is a classic example, consisting of four subunits, each with a heme group for oxygen binding.
Summary Table: Structural Levels in Proteins
Structural Level | Characteristics |
|---|---|
Primary | Peptide bonds join amino acids in a specific sequence in a polypeptide. |
Secondary | Alpha helix or beta-pleated sheet forms by hydrogen bonding between backbone atoms. |
Tertiary | Polypeptide folds into a compact, three-dimensional shape stabilized by R group interactions. |
Quaternary | Two or more protein subunits combine and are stabilized by interactions to form a biologically active protein. |

Protein Hydrolysis and Denaturation
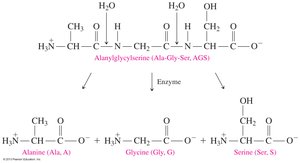
Protein Hydrolysis
Protein hydrolysis is the cleavage of peptide bonds, resulting in smaller peptides or free amino acids. This process occurs during digestion and cellular protein turnover. In the laboratory, hydrolysis requires acid or base, water, and heat; in the body, it is catalyzed by enzymes.
Protein Denaturation
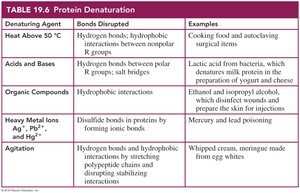
Denaturation is the loss of a protein's native structure due to the disruption of secondary, tertiary, or quaternary interactions. Denaturation can be caused by heat, acids and bases, organic compounds, heavy metal ions, or agitation. Denatured proteins lose their biological activity.
Denaturing Agent | Bonds Disrupted | Examples |
|---|---|---|
Heat Above 50°C | Hydrogen bonds, hydrophobic interactions | Cooking food, autoclaving surgical items |
Acids and Bases | Hydrogen bonds, salt bridges | Lactic acid in bacteria, cheese production |
Organic Compounds | Hydrophobic interactions | Ethanol, isopropyl alcohol (disinfectants) |
Heavy Metal Ions | Disulfide bonds | Mercury, lead poisoning |
Agitation | Hydrogen bonds, hydrophobic interactions | Whipped cream, meringue |
Key Concepts and Applications
Proteins are polymers of amino acids linked by peptide bonds.
Amino acids are classified by the properties of their side chains (R groups).
Protein structure is organized into four levels: primary, secondary, tertiary, and quaternary.
Hydrolysis breaks peptide bonds, while denaturation disrupts higher-order structures without breaking peptide bonds.
