 Back
BackAnalyzing Percent Dissociation of Acids Using Particle Diagrams
Study Guide - Smart Notes

Q19. Which acid has the lowest percent dissociation? The following pictures represent aqueous solutions of three acids HA (A = X, Y, or Z); water molecules have been omitted for clarity.
Background
Topic: Acid Dissociation and Particle Diagrams
This question tests your understanding of how to interpret particle diagrams to determine the relative percent dissociation of different acids in aqueous solution. Percent dissociation refers to the fraction of acid molecules that ionize to produce hydronium ions () and conjugate base ions () in water.
Key Terms and Concepts:
Percent Dissociation: The percentage of acid molecules that dissociate into ions in solution.
HA: Generic formula for a monoprotic acid.
: Hydronium ion, formed when an acid donates a proton to water.
: Conjugate base of the acid.
Step-by-Step Guidance
Examine each particle diagram (HX, HY, HZ) and count the number of undissociated acid molecules (HA), hydronium ions (), and conjugate base ions ().
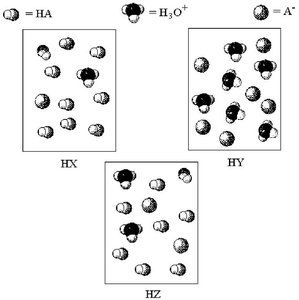
Recall that a higher number of and particles relative to HA indicates a higher percent dissociation.
For each acid (HX, HY, HZ), estimate the ratio of dissociated particles ( and ) to the total number of acid particles (HA + ).
Identify which diagram has the lowest ratio of dissociated ions to undissociated acid molecules. This acid will have the lowest percent dissociation.
Try solving on your own before revealing the answer!
