 Back
BackAqueous Equilibria: Buffers, Titrations, and Solubility
Study Guide - Smart Notes

Buffer Solutions
Definition and Properties
Buffer solutions are aqueous systems that resist significant changes in pH upon the addition of small amounts of acid or base. They are composed of a weak acid and its conjugate base, or a weak base and its conjugate acid. Buffers are essential in many biological and chemical processes where maintaining a stable pH is crucial.
Key Point 1: Buffers contain a weak acid to neutralize added bases and a weak base to neutralize added acids.
Key Point 2: The components of a buffer must not react with each other directly.
Key Point 3: Buffers are typically prepared with nearly equal concentrations of the conjugate acid-base pair.
Example: A solution containing 0.5 M acetic acid (CH3COOH) and 0.5 M sodium acetate (CH3COONa) acts as a buffer. When small amounts of HCl or NaOH are added, the pH changes only slightly compared to pure water.

Buffer Action
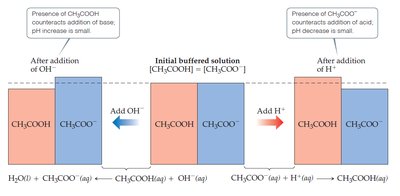
The ability of a buffer to resist pH changes is due to the presence of both a weak acid and its conjugate base. When acid (H+) is added, the conjugate base neutralizes it; when base (OH-) is added, the weak acid neutralizes it.
Key Point: The buffer's effectiveness depends on the concentrations of the acid and base components.
Biological Example: Blood Buffer System
Blood is buffered at pH 7.40 ± 0.05. The main buffer system in blood involves carbonic acid (H2CO3) and bicarbonate (HCO3-):
CO2(aq) + H2O(l) ↔ H2CO3(aq)
H2CO3(aq) + H2O(l) ↔ H3O+(aq) + HCO3-(aq)
Normal concentrations: [H2CO3] = 0.0025 M, [HCO3-] = 0.025 M.
The pH of Buffer Solutions
Henderson-Hasselbalch Equation
The pH of a buffer solution is determined by the ratio of the concentrations of the conjugate base ([A-]) to the weak acid ([HA]), not their absolute amounts. The Henderson-Hasselbalch equation is used to calculate buffer pH:
pKa is the negative logarithm of the acid dissociation constant (Ka).
When [HA] = [A-], pH = pKa because .


Buffer Range and Selection
The useful buffer range is typically pH = pKa ± 1, corresponding to a [A-]/[HA] ratio between 0.1 and 10. Select a buffer system with a pKa close to the desired pH.
Buffer Capacity
Definition and Factors
Buffer capacity is the amount of acid or base that can be added to a buffer without causing a significant change in pH. It depends on the absolute concentrations of the buffer components.
Key Point: The greater the concentrations of [A-] and [HA], the higher the buffer capacity.


Addition of Acid or Base to a Buffer
Stoichiometry and Equilibrium Calculations
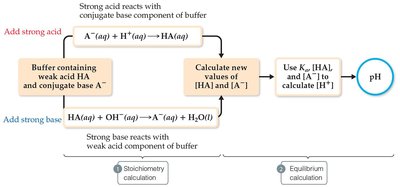
When a strong acid or base is added to a buffer, the buffer components react to minimize pH changes. The process involves two steps: a stoichiometry calculation (to determine new concentrations) and an equilibrium calculation (to find the new pH).
Example: Adding NaOH to a buffer of NaH2PO4 and Na2HPO4 converts H2PO4- to HPO42-, changing the [A-]/[HA] ratio and thus the pH.
Acid-Base Titrations
Principles and Apparatus
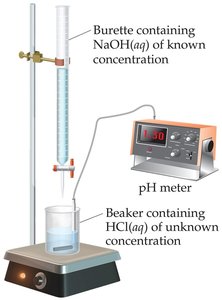
Acid-base titrations are used to determine the concentration of an unknown acid or base by reacting it with a standard solution (titrant) of known concentration. The titration is monitored using a pH meter or an indicator that changes color near the equivalence point.
Equivalence Point: The point at which stoichiometrically equivalent amounts of acid and base have reacted.
End Point: The point at which the indicator changes color, ideally close to the equivalence point.
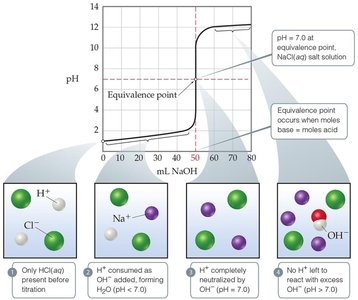
Titration Curves
Titration curves plot pH versus volume of titrant added. The shape of the curve depends on the strengths of the acid and base involved.
Strong Acid–Strong Base: Sharp rise in pH at equivalence point (pH = 7 for monoprotic acids).
Weak Acid–Strong Base: Equivalence point at pH > 7 due to the formation of a weak conjugate base.
Weak Base–Strong Acid: Equivalence point at pH < 7 due to the formation of a weak conjugate acid.
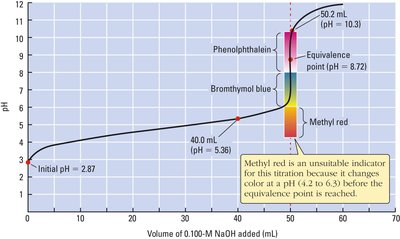
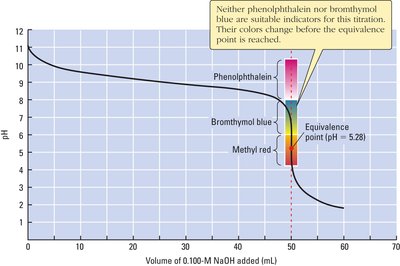
Indicators and Detection of Equivalence Point
Indicators are weak acids or bases that change color over a specific pH range. The choice of indicator depends on the expected pH at the equivalence point.
Example: Methyl red (pKa ≈ 5.0), bromothymol blue (pKa ≈ 7.1), and phenolphthalein (pKa ≈ 9.3) are common indicators for different titration types.

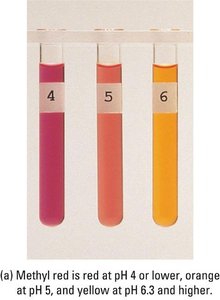
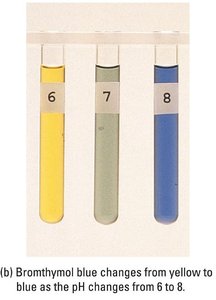
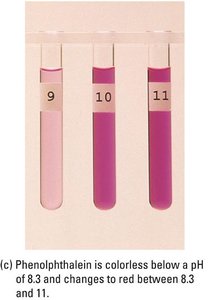
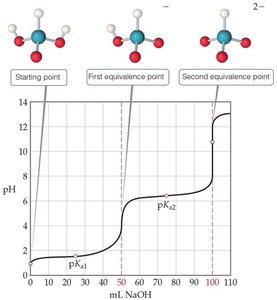
Polyprotic Acid Titrations
Multiple Equivalence Points
Polyprotic acids (acids with more than one ionizable proton) exhibit multiple equivalence points in titration curves, each corresponding to the neutralization of one proton.
Solubility Equilibria and Ksp
Solubility Product Constant (Ksp)
The solubility product constant, Ksp, describes the equilibrium between a solid and its ions in a saturated solution. For a salt AxBy:
Example: For AgCl,


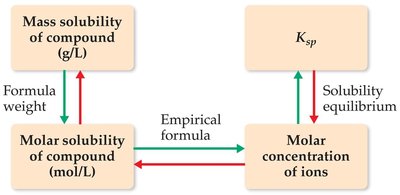
Calculating Solubility from Ksp
Solubility (S) can be calculated from Ksp by setting up an equilibrium expression. For AgCl:
Example: If , then M.

Factors Affecting Solubility
pH: Salts with basic anions are more soluble in acidic solutions due to the removal of the anion by reaction with H+.
Common Ion Effect: The presence of a common ion decreases solubility. For example, AgCl is less soluble in NaCl solution than in pure water.


Complex Ion Formation
Some metal ions form complex ions with ligands, increasing their solubility. The overall equilibrium is described by the product of Ksp and the formation constant (Kf).
Example: AgBr(s) + 2 S2O32-(aq) → [Ag(S2O3)2]3-(aq) + Br-(aq)
Precipitation and Selective Precipitation
Predicting Precipitation
To determine if a precipitate will form, calculate the ion product Q and compare it to Ksp:
If Q > Ksp: Precipitation occurs.
If Q = Ksp: Solution is saturated (at equilibrium).
If Q < Ksp: No precipitation; more solid can dissolve.
Selective Precipitation
Selective precipitation is used to separate ions based on their differing solubilities. By carefully controlling the concentration of added ions, one ion can be precipitated while others remain in solution.
Additional info: These notes cover the core concepts of aqueous equilibria, buffer systems, titrations, and solubility equilibria, as outlined in a standard general chemistry curriculum. All images included are directly relevant to the explanations provided and reinforce key concepts visually.
