 Back
BackAqueous Ionic Equilibrium: Buffers, Titrations, and Solubility
Study Guide - Smart Notes

Chapter 17 – Aqueous Ionic Equilibrium
Buffers: Solutions that Resist pH Changes
Buffer solutions are mixtures of a weak acid and its conjugate base (or a weak base and its conjugate acid) that resist changes in pH when small amounts of acid or base are added. The ability of buffers to maintain pH is crucial in many chemical and biological systems.
Buffer Definition: A solution that resists significant changes in pH upon addition of small amounts of acid or base.
Components: High concentrations of a weak acid/base and its conjugate counterpart.
Mechanism: Buffers neutralize added acids or bases by shifting equilibrium.
Example: Acetic acid (CH3COOH) and sodium acetate (NaCH3COO).
Common Ion Effect
The common ion effect occurs when a compound containing an ion already present in the solution is added, shifting the equilibrium and reducing the solubility or ionization of the weak acid/base.
Le Chatelier’s Principle: Addition of a common ion shifts equilibrium to reduce ionization.
Example: Adding sodium acetate to acetic acid solution decreases [H3O+].
Calculating Buffer pH: Henderson-Hasselbalch Equation
When both the concentration of weak acid and conjugate base are known, and the acid dissociation constant (Ka) is small, the pH can be calculated using the Henderson-Hasselbalch equation:
Equation:
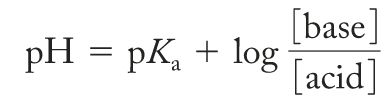
Buffer Calculations and Examples
Buffer calculations often involve two steps: stoichiometry (to account for added acid/base) and equilibrium (to determine final pH).
Step 1: Stoichiometry calculation (moles).
Step 2: Equilibrium calculation (molarity).
Alternative: If K is small and volume change is negligible, use the Henderson-Hasselbalch equation with updated concentrations.
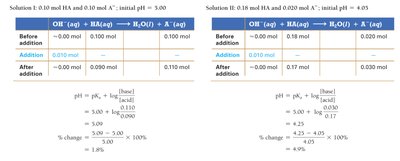

Buffer Effectiveness: Range and Capacity
Buffer capacity is the amount of acid or base a buffer can neutralize before the pH changes significantly. The effectiveness depends on the concentrations and ratio of acid to base.
Effective Buffer Ratio: should be between 0.1 and 10.
Concentration: Higher concentrations increase buffer capacity.
Effective Range: One pH unit on either side of pKa.
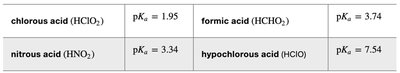
Choosing an Acid: Select an acid with a pKa close to the desired pH.
Titrations and pH Curves
Titration is a technique where a solution of known concentration (titrant) is added to a solution of unknown concentration (analyte) to determine its concentration. The pH is monitored throughout the process, and the equivalence point is where moles of acid equal moles of base.
Equivalence Point: The point at which stoichiometrically equivalent amounts of acid and base have reacted.
Titration Curve: A plot of pH versus volume of titrant added, showing how pH changes during titration.
Strong Acid–Strong Base Titrations
In these titrations, the pH changes rapidly near the equivalence point. Calculations involve determining moles of acid and base, neutralization, and resulting concentrations.
Initial pH: Determined by the concentration of strong acid.
After Addition: Calculate moles neutralized and new [H3O+].
Weak Acid–Strong Base Titrations
Weak acid titrations differ in that the pH at the equivalence point is above 7 due to the production of the conjugate base. The titration curve is less steep near the equivalence point.
Half-Equivalence Point: [HA] = [A–], pH = pKa.
Equivalence Point: All acid converted to conjugate base.
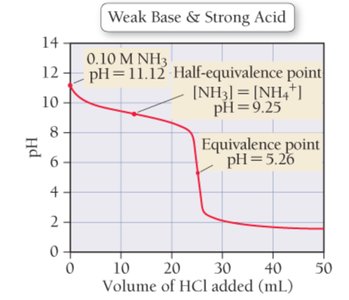
Weak Base–Strong Acid Titrations
These titrations start with a basic solution and become acidic after the equivalence point. The curve is similar to weak acid–strong base titrations but inverted.
Titrations of Polyprotic Acids
Polyprotic acids have more than one ionizable proton and can exhibit multiple equivalence points in titration curves, each corresponding to the loss of a proton.
Example: Sulfurous acid (H2SO3) has two dissociation steps with different Ka values.
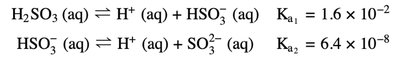
Solubility Equilibria and the Solubility-Product Constant (Ksp)
Solubility equilibria describe the dissolution of ionic compounds in water. The solubility-product constant (Ksp) quantifies the equilibrium between the solid and its ions in solution.
Ksp Definition: for a salt MmAn.
Molar Solubility: The concentration of dissolved solid (mol/L).
Comparing Ksp: Only valid for compounds with similar stoichiometry.

Common Ion Effect on Solubility
The presence of a common ion reduces the solubility of a salt by shifting the dissolution equilibrium.
Example: Solubility of PbCO3 decreases in a solution containing Pb(NO3)2.
Effect of pH on Solubility
The solubility of salts with basic anions increases in acidic solutions due to the neutralization of the anion.
Example: BaF2 is more soluble in acidic solution than in neutral solution.
Precipitation and Predicting Precipitate Formation
Precipitation occurs when the product of ion concentrations (Q) exceeds the Ksp. The comparison of Q and Ksp determines whether a precipitate forms.
If Q < Ksp: No precipitate forms (unsaturated).
If Q > Ksp: Precipitate forms (supersaturated).
If Q = Ksp: Solution is saturated.
Complex Ion Equilibria
Transition metal ions often form complex ions with Lewis bases, increasing their solubility in solution. The formation constant (Kf) quantifies the stability of the complex ion.
Complex Ion: A metal ion surrounded by ligands (Lewis bases).
Formation Constant:
Effect on Solubility: Complex ion formation increases the solubility of metal salts.
Common Lewis Bases: Ammonia, water, chloride, cyanide.
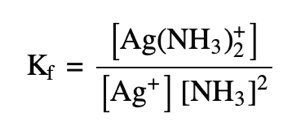
Additional info: All equations are provided in LaTeX format for clarity. Tables and curves are included only when directly relevant to the explanation. The notes cover buffer systems, titrations, solubility equilibria, precipitation, and complex ion formation, as outlined in Chapter 17 of a general chemistry course.
