 Back
BackAqueous Ionic Equilibrium: Buffers, Titrations, and Solubility
Study Guide - Smart Notes

Chapter 16 – Aqueous Ionic Equilibrium
Buffers
Buffer solutions are essential in chemistry for maintaining a stable pH when small amounts of acid or base are added. They are composed of a weak acid and its conjugate base, or a weak base and its conjugate acid, and are only effective when both components are present in similar concentrations (within a 1:10 ratio).
Definition: A buffer is a solution that resists changes in pH upon addition of small amounts of strong acid or strong base.
Composition: Contains both members of a conjugate acid/base pair.
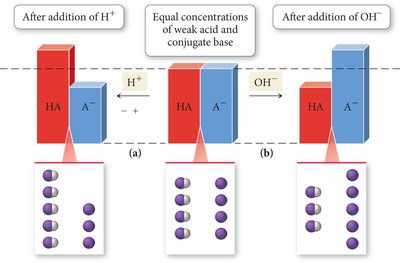
Mechanism: Added H3O+ is neutralized by the basic component; added OH– is neutralized by the acidic component.
Example: Acetic acid (CH3COOH) and sodium acetate (CH3COONa) form a buffer system.
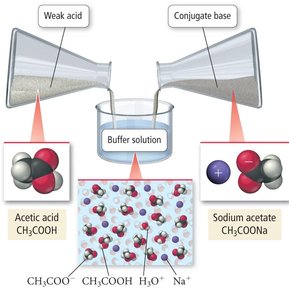
Buffer Action and the Common Ion Effect
Buffer solutions operate through equilibrium reactions. The presence of a common ion suppresses the ionization of the weak acid or base, a phenomenon known as the common ion effect.
Equilibrium Example: CH3COOH(aq) + H2O(l) ↔ CH3COO–(aq) + H3O+(aq)
Common Ion Effect: The addition of CH3COO– (from sodium acetate) suppresses the ionization of acetic acid.
Calculating Buffer pH: Henderson-Hasselbalch Equation
The Henderson-Hasselbalch equation provides a simplified method for calculating the pH of a buffer, assuming the "x is small" approximation is valid (percent ionization < 5%).
Equation:
Application: If [A–] and [HA] are equal, pH = pKa.
Rule of Thumb: The equation is valid if and .
Buffer Calculations: Stoichiometry and Equilibrium
When a strong acid or base is added to a buffer, the calculation involves two steps: a stoichiometry calculation to determine the new amounts of buffer components, followed by an equilibrium calculation to determine the new pH.
Stoichiometry: Added acid reacts with A– to form HA; added base reacts with HA to form A–.
Equilibrium: Use the Henderson-Hasselbalch equation with updated concentrations.
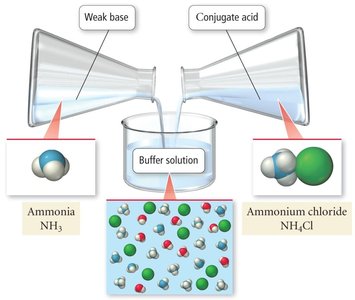
Basic Buffers
Buffers can also be made from a weak base and its conjugate acid. The calculations are analogous, but use Kb and pKb values. The relationship between pKa and pKb is given by:
Equation: at 25°C
Example: Ammonia (NH3) and ammonium chloride (NH4Cl) form a basic buffer.
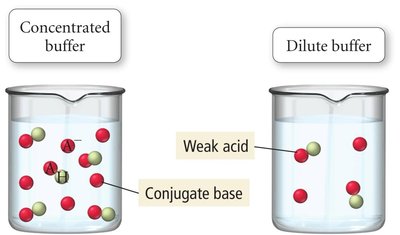
Buffer Effectiveness
The effectiveness of a buffer depends on the relative and absolute concentrations of its components. A buffer is most effective when the concentrations of acid and conjugate base are equal and high.
Relative Concentrations: Should not differ by more than a factor of 10.
Absolute Concentrations: Higher concentrations increase buffer capacity.
Buffer Range: Effective within pH = pKa ± 1.
Acid-Base Titrations
Titrations are used to determine the concentration of an acid or base by neutralizing it with a titrant. The titration curve shows pH versus volume of titrant added, with distinct regions: initial conditions, before equivalence point, at equivalence point, and after equivalence point.
Strong Acid + Strong Base: Equivalence point at pH = 7.00.
Weak Acid + Strong Base: Equivalence point at pH > 7.00.
Weak Base + Strong Acid: Equivalence point at pH < 7.00.

Indicators
Acid-base indicators are substances whose color changes at specific pH ranges, marking the endpoint of a titration. The choice of indicator depends on the steepest part of the titration curve.
Transition Range: Color change occurs at indicator pKa ± 1.
Indicator Error: Difference between observed endpoint and true equivalence point.
Solubility and the Solubility Product Constant (Ksp)
Solubility describes how much of a compound dissolves in water. The solubility product constant (Ksp) quantifies the equilibrium between a solid and its ions in solution.
Definition:
Molar Solubility: The number of moles of solute that dissolve per liter of solution.
Example: For CaF2:
The Common Ion Effect on Solubility
The presence of a common ion decreases the solubility of an ionic compound due to Le Châtelier’s principle. For example, adding NaCl to a PbCl2 solution decreases PbCl2 solubility.
Calculation: Use an ICE table and Ksp expression, accounting for the initial concentration of the common ion.
The Effect of pH on Solubility
The solubility of salts containing basic anions increases in acidic conditions. Acid neutralizes the basic anion, shifting the equilibrium to dissolve more solid.
Example: Mg(OH)2 is more soluble in acidic media because H3O+ neutralizes OH–.
Precipitation and Selective Precipitation
Precipitation occurs when the reaction quotient (Q) exceeds Ksp. Selective precipitation is used to separate ions by adding a reagent that precipitates one ion before others.
Q < Ksp: No precipitation; solution is unsaturated.
Q > Ksp: Precipitation occurs; solution is supersaturated.
Selective Precipitation: Used to separate ions based on differences in Ksp values.
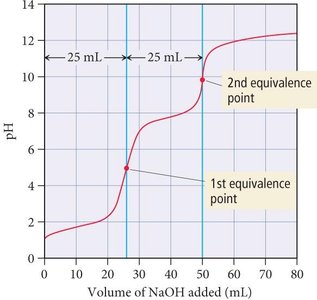
Polyprotic Acid Titration
Polyprotic acids have multiple equivalence points in titration, each corresponding to the removal of a proton. The volume required for each equivalence point is consistent.
Example: Titration of H2SO3 by NaOH shows two equivalence points.
Summary Table: Buffer Effectiveness
The following tables illustrate the effect of relative and absolute concentrations on buffer effectiveness:
Condition | Before Addition | Addition | After Addition |
|---|---|---|---|
Solution I: 0.10 mol HA and 0.10 mol A–; initial pH = 5.00 | 0.100 mol HA, 0.100 mol A– | 0.010 mol OH– | 0.090 mol HA, 0.110 mol A– |
Solution II: 0.18 mol HA and 0.020 mol A–; initial pH = 4.05 | 0.18 mol HA, 0.020 mol A– | 0.010 mol OH– | 0.17 mol HA, 0.030 mol A– |


Condition | Before Addition | Addition | After Addition |
|---|---|---|---|
Solution I: 0.50 mol HA and 0.50 mol A–; initial pH = 5.00 | 0.50 mol HA, 0.50 mol A– | 0.010 mol OH– | 0.49 mol HA, 0.51 mol A– |
Solution II: 0.050 mol HA and 0.050 mol A–; initial pH = 5.00 | 0.050 mol HA, 0.050 mol A– | 0.010 mol OH– | 0.040 mol HA, 0.060 mol A– |


Additional info: This guide covers the core concepts of aqueous ionic equilibrium, including buffer systems, titration curves, solubility, and precipitation, with relevant examples and equations for exam preparation.
