 Back
BackAqueous Ionic Equilibrium: Buffers, Titrations, and Solubility
Study Guide - Smart Notes

Aqueous Ionic Equilibrium
Acidosis and Buffering in Blood
Aqueous ionic equilibrium is crucial for maintaining physiological pH, especially in blood. The body uses buffers to resist changes in pH, preventing conditions like acidosis, which can compromise oxygen transport.
Acidosis: Occurs when blood pH drops below normal, often due to accumulation of acidic metabolites such as glycolic acid.
Buffering System: The primary buffer in blood is a mixture of carbonic acid (H2CO3) and bicarbonate (HCO3-).
Example: Ethylene glycol (antifreeze) is metabolized to glycolic acid, overwhelming the blood's buffer and causing acidosis.
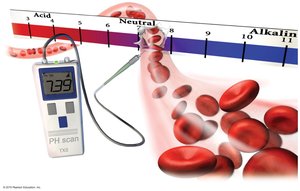
Additional info: The pH of blood is tightly regulated around 7.4; deviations can be life-threatening.
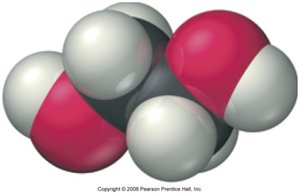
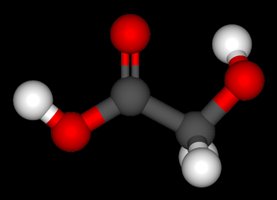
Ethylene Glycol and Glycolic Acid
Ethylene glycol is a common component of antifreeze. It is metabolized in the liver to glycolic acid, which is toxic due to its effect on blood pH.
Ethylene Glycol: A diol with the formula HOCH2CH2OH.
Glycolic Acid: An alpha-hydroxy acid (HOCH2COOH) formed from ethylene glycol metabolism.
Toxicity Mechanism: Glycolic acid lowers blood pH, leading to acidosis.
Buffer Solutions
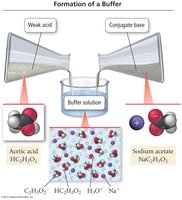
Definition and Function
Buffers are solutions that resist changes in pH when small amounts of acid or base are added. They are typically made by mixing a weak acid with its conjugate base or a weak base with its conjugate acid.
Weak Acid/Conjugate Base Buffer: Example: Acetic acid (HC2H3O2) and sodium acetate (NaC2H3O2).
Weak Base/Conjugate Acid Buffer: Example: Ammonia (NH3) and ammonium chloride (NH4Cl).
Buffer Action: Addition of Acid or Base
Buffers neutralize added acids or bases by shifting equilibrium according to Le Châtelier’s Principle.
Addition of Base: HA + OH- → A- + H2O
Addition of Acid: H+ + A- → HA
Common Ion Effect
The common ion effect occurs when a salt containing the conjugate base of the buffer acid is added, shifting the equilibrium and increasing the pH.
Example: Adding NaA to HA(aq) + H2O(l) ⇌ A-(aq) + H3O+(aq) shifts equilibrium left.

Buffer Calculations: Acetic Acid Example
To calculate the pH of a buffer, use the equilibrium expression for the weak acid and its conjugate base.
Equilibrium Expression:
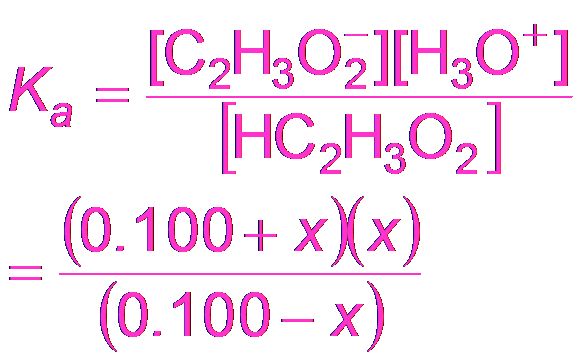
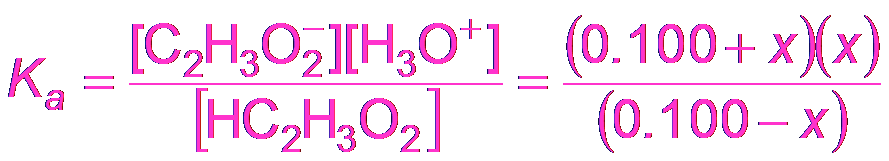
Approximation: If x is small, M
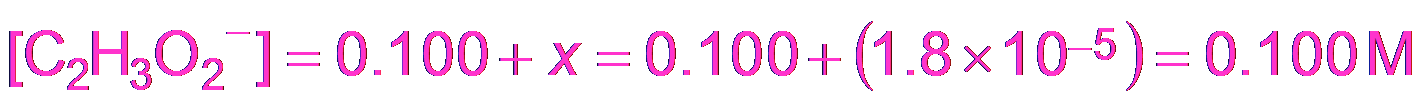
Buffer Calculations: Hydrofluoric Acid Example
Similar calculations apply for buffers made from HF and KF.
Equilibrium Expression:
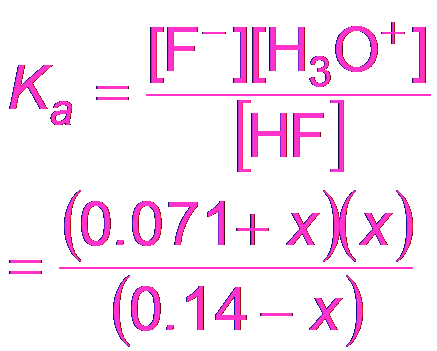
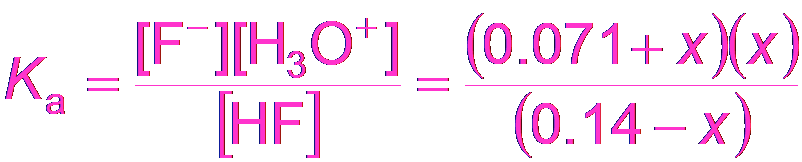
Henderson-Hasselbalch Equation
The Henderson-Hasselbalch equation simplifies buffer pH calculations when the "x is small" approximation is valid.
Equation:

Buffer Calculations: Benzoic Acid Example
For a buffer made from benzoic acid and sodium benzoate:
Example Calculation:
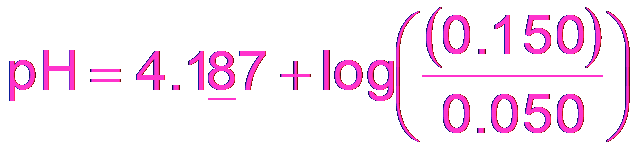
Buffer Calculations: HF Example
For a buffer made from HF and KF:
Example Calculation:
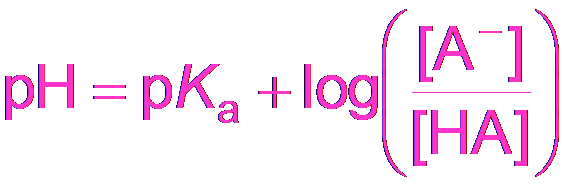
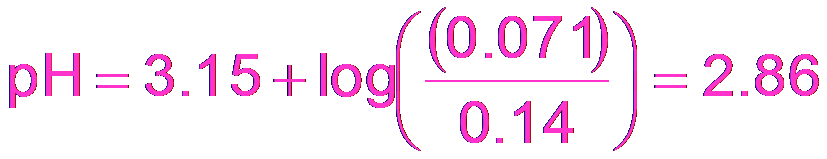
Buffer Response to Added Acid or Base
When acid or base is added to a buffer, a stoichiometric calculation is performed to determine the new concentrations, followed by an equilibrium calculation for the new pH.
Example: Adding NaOH to an acetic acid/acetate buffer.
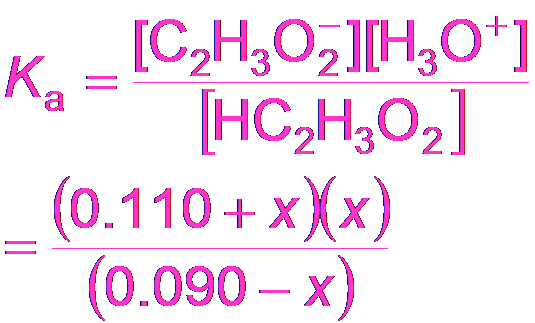
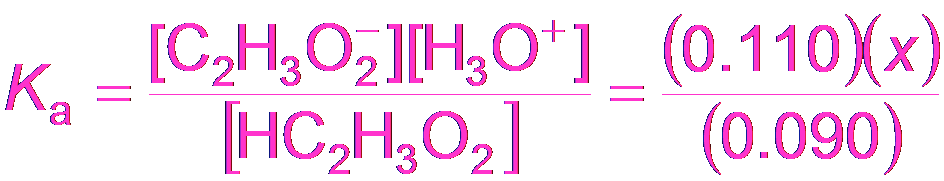
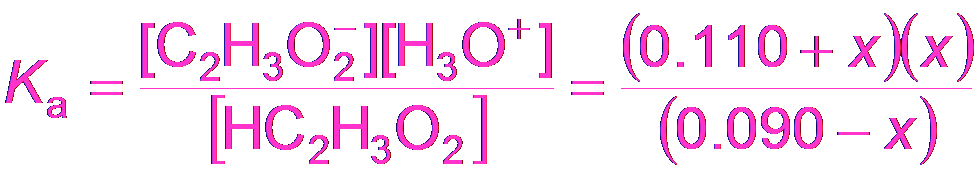
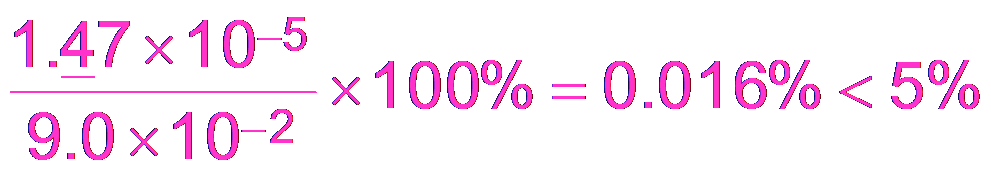
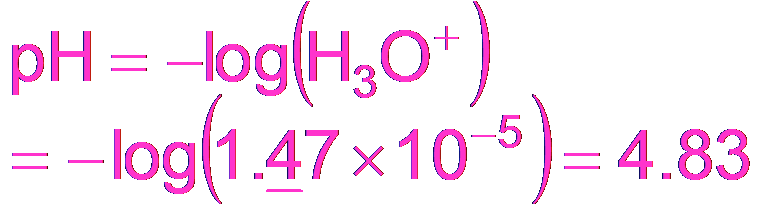
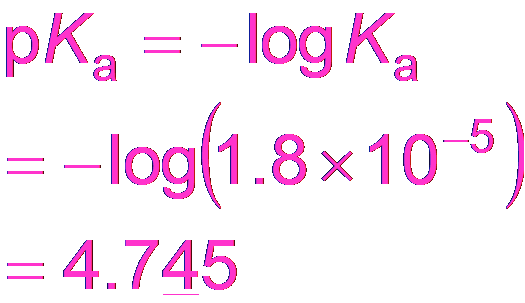
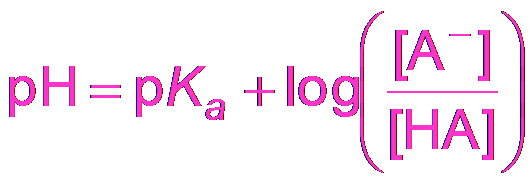
Buffering Effectiveness
Relative and Absolute Concentrations
The effectiveness of a buffer depends on the relative and absolute concentrations of acid and base.
Relative Amounts: Most effective when [acid] = [base].
Absolute Concentrations: Most effective when both concentrations are large.
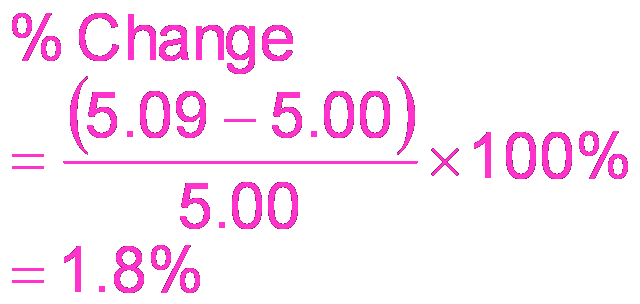
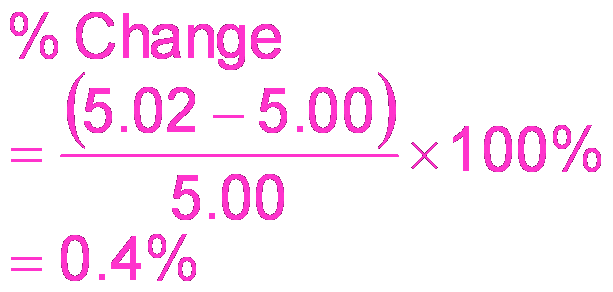
Buffering Capacity and Range
Buffering capacity is the amount of acid or base a buffer can neutralize. The effective pH range is pKa ± 1.
Buffering Capacity: Increases with higher concentrations.
Buffering Range: Effective when .
Titration and Indicators
Acid-Base Titration
Titration is a technique to determine the concentration of an unknown solution by reacting it with a solution of known concentration.
Endpoint: The point at which the reaction is complete, often indicated by a color change.
Equivalence Point: When moles of acid equal moles of base.


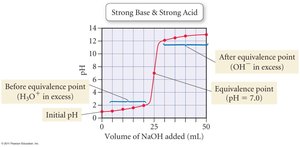
Titration Curves
A titration curve plots pH versus volume of titrant added. The inflection point is the equivalence point.
Strong Acid/Strong Base: Equivalence point at pH 7.
Weak Acid/Strong Base: Equivalence point above pH 7.
Weak Base/Strong Acid: Equivalence point below pH 7.
Indicators
Indicators are weak acids or bases that change color depending on the pH of the solution. They are used to visually determine the endpoint of a titration.
Phenolphthalein: Colorless in acidic, pink in basic solutions.
Methyl Red: Red in acidic, yellow in basic solutions.
Solubility Equilibria
Solubility Product (Ksp)
The solubility product, Ksp, is the equilibrium constant for the dissolution of an ionic solid in water.
General Form: For MnXm(s) ⇌ nMm+(aq) + mXn-(aq),
Example: PbCl2(s) ⇌ Pb2+(aq) + 2 Cl-(aq),
Common Ion Effect on Solubility
Addition of a soluble salt containing a common ion decreases the solubility of an "insoluble" salt.
Example: Adding NaCl to PbCl2 equilibrium decreases PbCl2 solubility.
Effect of pH on Solubility
Solubility of ionic hydroxides decreases with increasing pH, while solubility of salts containing weak acid anions increases with decreasing pH.
Example: M(OH)n(s) ⇌ Mn+(aq) + nOH-(aq)
Example: M2(CO3)n(s) ⇌ 2 Mn+(aq) + nCO32-(aq)
Precipitation and Selective Precipitation
Precipitation occurs when the ion product Q exceeds Ksp. Selective precipitation is used to separate ions based on their differing Ksp values.
Q = Ksp: Solution is saturated, no precipitation.
Q < Ksp: Solution is unsaturated, no precipitation.
Q > Ksp: Precipitation occurs.
Complex Ion Formation
Complex Ions and Formation Constants
Transition metals often form complex ions with ligands. The equilibrium constant for complex ion formation is called the formation constant, Kf.
Example: Ag+(aq) + 2 NH3(aq) ⇌ Ag(NH3)2+(aq)
Effect of Complex Ion Formation on Solubility
The solubility of an ionic compound increases in the presence of ligands that form strong complexes with the metal ion.
Example: Adding NH3 to AgCl(s) increases Ag+ solubility.
Amphoteric Metal Hydroxides
Some metal hydroxides are amphoteric, meaning they can dissolve in both acidic and basic solutions by acting as either an acid or a base.
Examples: Al3+, Cr3+, Zn2+, Pb2+, Sb2+
Additional info: Amphoteric behavior is important in qualitative analysis and environmental chemistry.
