 Back
BackAtoms and the Periodic Table: Structure, Properties, and Electron Configurations
Study Guide - Smart Notes

Atoms and the Periodic Table
Atomic Theory and the Structure of Atoms
The foundation of modern chemistry is atomic theory, which describes the nature and structure of atoms. Atoms are the smalles. t units of matter that retain the identity of an element. Atomic theory is based on four key assumptions:
All matter is composed of atoms.
Atoms of a given element differ from those of other elements.
Chemical compounds consist of atoms combined in specific ratios.
Chemical reactions change only the way atoms are combined in compounds.
Atoms are made up of three types of subatomic particles: protons, neutrons, and electrons. Protons carry a positive charge, neutrons are neutral, and electrons carry a negative charge. The mass of these particles is measured in atomic mass units (amu), with electrons being much lighter than protons and neutrons.
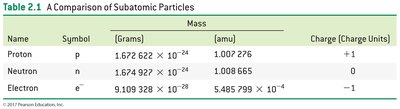
The nucleus, composed of protons and neutrons, is extremely small and dense compared to the overall size of the atom, which is mostly empty space occupied by rapidly moving electrons.
The relative size of the nucleus compared to the atom is similar to a pea in the middle of a stadium.

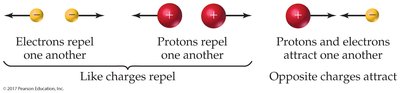
Electrostatic forces govern the behavior of these particles: opposite charges attract, while like charges repel.
Elements and Atomic Number
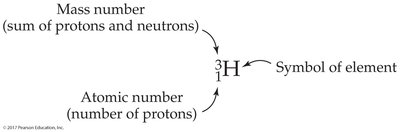
Each element is defined by its atomic number (Z), which is the number of protons in its nucleus. The mass number (A) is the sum of protons and neutrons. In a neutral atom, the number of electrons equals the number of protons.
Atomic Number (Z): Number of protons
Mass Number (A): Number of protons + neutrons
To find the number of neutrons:

Isotopes and Atomic Weight
Isotopes are atoms of the same element (same Z) but with different mass numbers (A) due to varying numbers of neutrons. For example, hydrogen has three isotopes: protium, deuterium, and tritium.
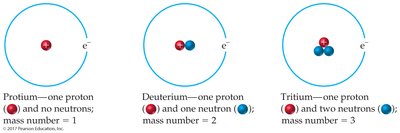
Isotopes are represented with their mass number as a superscript and atomic number as a subscript before the atomic symbol:
The atomic weight of an element is the weighted average mass of its naturally occurring isotopes, calculated as:
The Periodic Table
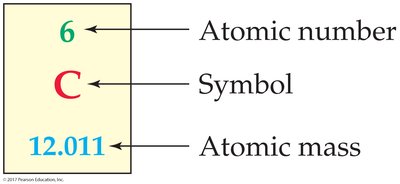
The periodic table organizes all known elements by increasing atomic number and groups elements with similar properties together. Each element's box contains its atomic number, symbol, and atomic mass.
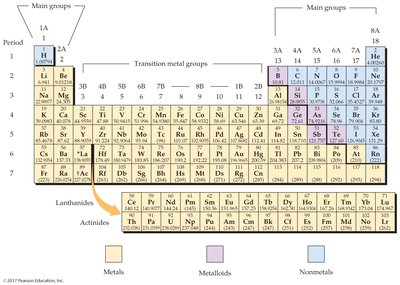
Elements are classified as metals, nonmetals, or metalloids based on their physical and chemical properties:
Metals: Malleable, lustrous, good conductors; found on the left side of the table.
Nonmetals: Poor conductors; found on the upper-right side.
Metalloids: Intermediate properties; located in a zigzag band between metals and nonmetals.



Elements in the same vertical column (group) have similar chemical properties and are categorized as main group elements, transition metals, or inner transition metals.
Characteristics of Different Groups
The periodic table exhibits periodicity, a repeating pattern of properties such as atomic radius.
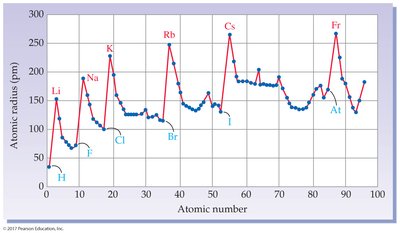
Group 1A (Alkali metals): Shiny, soft, highly reactive metals; never found pure in nature.
Group 2A (Alkaline earth metals): Lustrous, silvery, less reactive than alkali metals.
Group 7A (Halogens): Colorful, corrosive nonmetals; found only in compounds.
Group 8A (Noble gases): Colorless, chemically inert gases.
Electronic Structure of Atoms
The arrangement of electrons in an atom determines its chemical properties. The quantum mechanical model describes electrons as having both particle and wave properties, restricted to quantized energy levels.

Electrons are organized into shells, subshells, and orbitals:
Shells: Groupings by energy level (numbered 1, 2, 3, 4, ...)
Subshells: Types within shells (s, p, d, f)
Orbitals: Regions where electrons are likely found; s has 1, p has 3, d has 5, f has 7
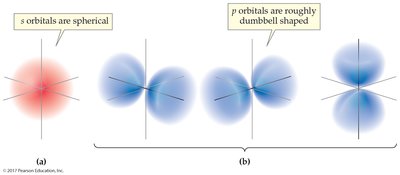
Each orbital holds two electrons with opposite spins. The farther a shell is from the nucleus, the more electrons it can hold and the higher their energy.
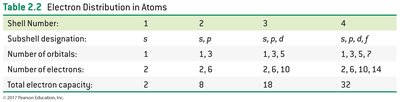
Shell Number | Subshells | Number of Orbitals | Electron Capacity |
|---|---|---|---|
1 | s | 1 | 2 |
2 | s, p | 1, 3 | 8 |
3 | s, p, d | 1, 3, 5 | 18 |
4 | s, p, d, f | 1, 3, 5, 7 | 32 |
Electron Configurations
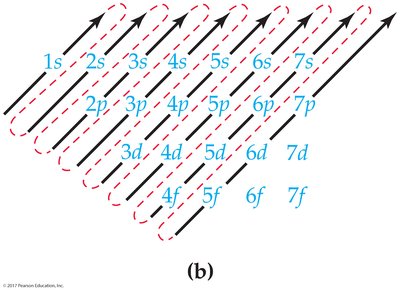
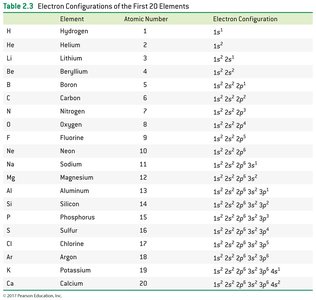
The electron configuration describes the arrangement of electrons in an atom's shells and subshells. Three rules govern electron configurations:
Electrons occupy the lowest energy orbitals available.
Each orbital holds only two electrons of opposite spin.
Orbitals of equal energy are half-filled before any is completely filled.
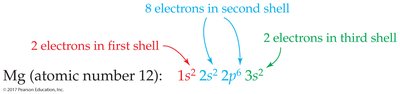
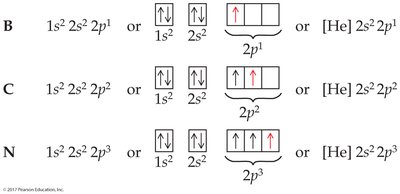
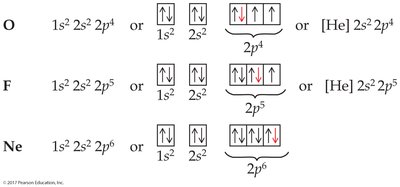
Electron configurations can be written in full or shorthand notation using noble gas symbols. Orbital diagrams use arrows to represent electron spins.
Electron Configurations and the Periodic Table
The periodic table is divided into blocks (s, p, d, f) based on the subshell filled last. Elements in the same group have similar valence shell electron configurations.
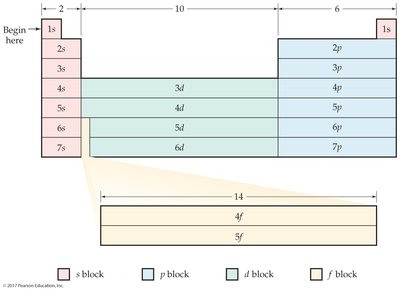
Valence electrons are those in the outermost shell and determine chemical reactivity.
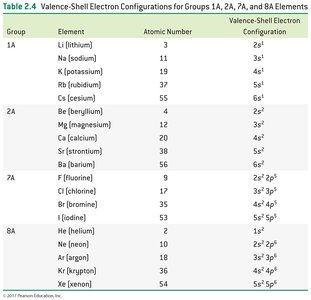
Group | Element | Valence-Shell Configuration |
|---|---|---|
1A | Li, Na, K, Rb, Cs | ns1 |
2A | Be, Mg, Ca, Sr, Ba | ns2 |
7A | F, Cl, Br, I | ns2np5 |
8A | He, Ne, Ar, Kr, Xe | ns2np6 |
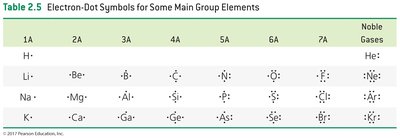
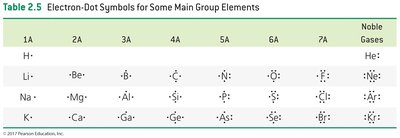
Electron-Dot Symbols (Lewis Symbols)
Electron-dot symbols (Lewis symbols) represent the valence electrons of an atom as dots around the atomic symbol. The number of dots equals the number of valence electrons.
Group | 1A | 2A | 3A | 4A | 5A | 6A | 7A | Noble Gases |
|---|---|---|---|---|---|---|---|---|
H | •H | |||||||
Li | •Li | |||||||
Be | •Be• | |||||||
B | •B•• | |||||||
C | •C••• | |||||||
N | •N•••• | |||||||
O | •O••••• | |||||||
F | •F•••••• | |||||||
Ne | •Ne••••••• |
Example: For group 5A elements, the electron-dot symbol shows five valence electrons, with the first four distributed singly and the fifth paired.
