 Back
BackAtoms: The Quantum World – Electronic Structure and Atomic Theory
Study Guide - Smart Notes

Atoms: The Quantum World
Modern View of Atomic Structure
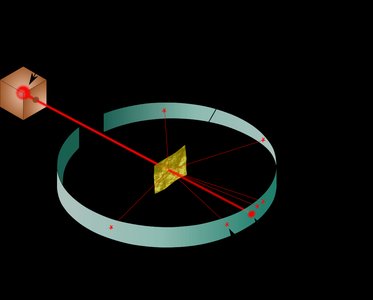
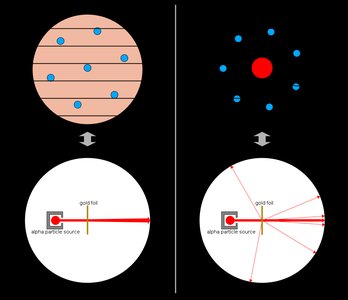
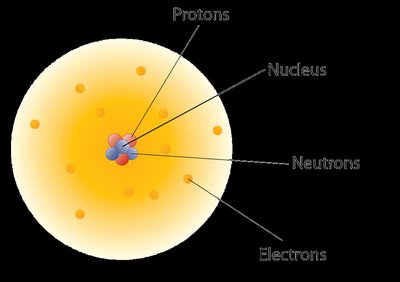
The modern atomic theory describes atoms as consisting of a dense nucleus containing protons and neutrons, surrounded by electrons occupying the space outside the nucleus. The discovery of the atomic nucleus was a pivotal moment in chemistry, leading to our current understanding of atomic structure.
Protons: Positively charged particles found in the nucleus.
Neutrons: Neutral particles also located in the nucleus.
Electrons: Negatively charged particles occupying the space outside the nucleus.
Atomic radius: Approximately 100 pm (1 x 10-10 m).
Nuclear radius: Approximately 5 x 10-3 pm (5 x 10-15 m).

Electrons in Atoms and Electromagnetic Radiation
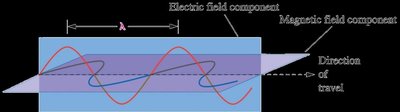
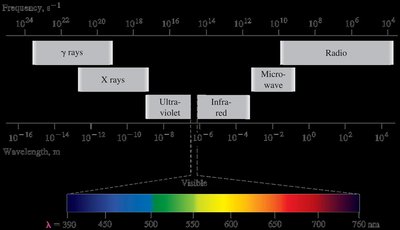
Electrons are the primary participants in chemical reactions (excluding nuclear reactions). The study of their behavior is closely linked to the interaction of electromagnetic (EM) radiation with matter. EM radiation is a form of energy transmission, propagating as waves of electric and magnetic fields.
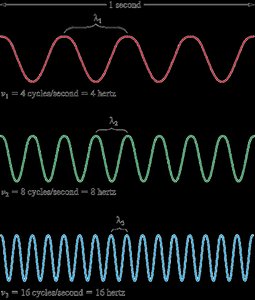
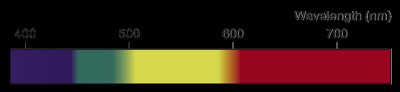
Wavelength (\(\lambda\)): The distance between identical points on successive waves. Units: m, cm, μm, nm, Å, pm.
Frequency (\(\nu\)): The number of waves passing a point per second. Unit: Hertz (Hz or s-1).
Velocity (c): Speed of light, \(c = 2.997925 \times 10^8\) m/s.
Relationship:
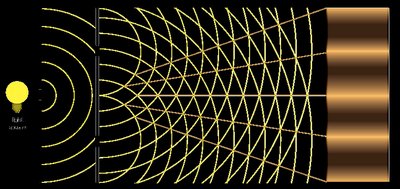
Wave Properties: Diffraction and Interference
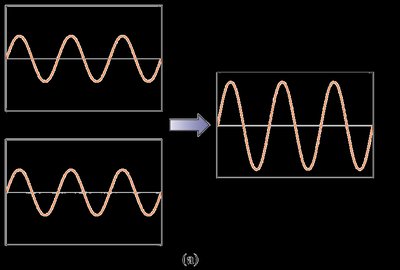
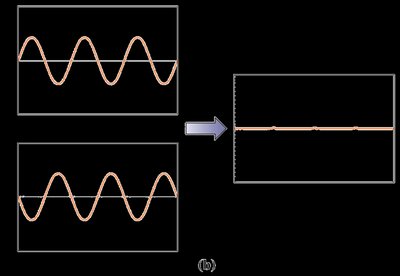
Waves exhibit properties such as diffraction and interference. Diffraction is the bending of waves around obstacles or through apertures, while interference is the combination of two or more waves to form a composite wave.
Constructive interference: Waves in phase add together.
Destructive interference: Waves out of phase cancel each other.


Quantum Theory and the Photoelectric Effect
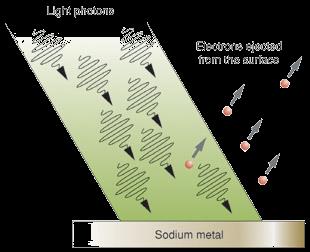
Classical physics could not explain certain phenomena, such as the photoelectric effect. Max Planck proposed that energy is quantized, and Albert Einstein extended this to suggest that light consists of particles called photons.
Planck's equation: (where \(h\) is Planck's constant, \(6.62607 \times 10^{-34}\) J·s)
Photoelectric effect: Electrons are ejected from metal surfaces when struck by light of sufficient energy.
Photon energy: If photon energy exceeds the work function, electrons are liberated.
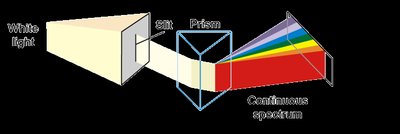
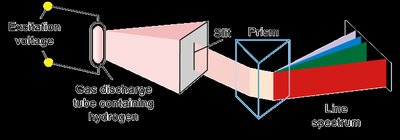
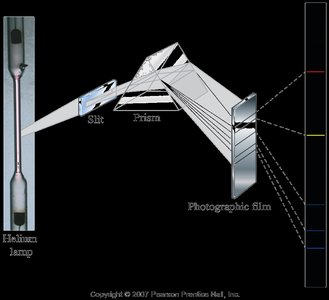
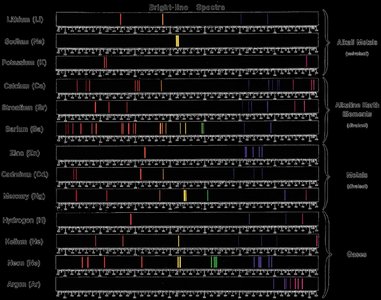
Atomic Emission Spectra
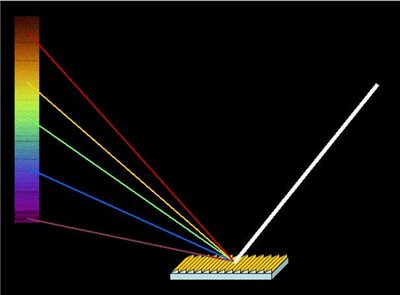
When elements are heated, their electrons are excited to higher energy levels. As electrons return to lower levels, they emit photons, producing characteristic line spectra unique to each element.
Line spectra: Discrete wavelengths emitted by excited atoms.
Flame tests: Used to identify elements by the color of emitted light.

The Bohr Model of the Atom
Niels Bohr proposed a model for the hydrogen atom, where electrons move in fixed orbits with quantized energies. Energy is emitted or absorbed when electrons transition between orbits.
Quantized energy levels: (where J)
Radius of orbit: (where pm)
Energy transitions:
Energy Levels, Spectra, and Ionization Energy
Electrons in atoms occupy quantized energy levels. The ground state is the lowest energy level (n=1), and excited states correspond to higher levels. Ionization energy is the energy required to remove an electron from the atom.
Energy-level diagram: Shows allowed transitions and corresponding spectral lines.
Ionization energy for hydrogen:
For hydrogen-like ions:
Wave-Particle Duality and Quantum Mechanics
Electrons and other particles exhibit both wave-like and particle-like properties. The de Broglie relation connects wavelength and momentum, and the Heisenberg Uncertainty Principle states that position and momentum cannot both be precisely measured.
de Broglie wavelength:
Heisenberg Uncertainty Principle:
Schrödinger Equation and Atomic Orbitals
The Schrödinger equation describes the behavior of electrons in atoms, yielding wave functions (orbitals) and quantized energy levels. Orbitals are defined by three quantum numbers: principal (n), angular momentum (l), and magnetic (ml).
Principal quantum number (n): Size and energy of the orbital.
Angular momentum quantum number (l): Shape of the orbital.
Magnetic quantum number (ml): Orientation of the orbital.
Electron spin quantum number (ms): +½ or -½, describes electron spin.
Multi-electron Atoms: Shielding and Penetration
In atoms with more than one electron, inner electrons shield outer electrons from the full nuclear charge, reducing the effective nuclear charge (Zeff). Electrons in s orbitals penetrate closer to the nucleus and are more effective at shielding.
Shielding effect: Reduces the attraction between nucleus and outer electrons.
Penetration: s > p > d (order of ability to approach the nucleus).
Energy order: In multi-electron atoms, subshells within the same principal shell are not degenerate.
Summary Table: Properties of Subatomic Particles
Particle | Electric Charge (SI) | Electric Charge (Atomic) | Mass (SI) | Mass (Atomic u) |
|---|---|---|---|---|
Proton | +1.6022 × 10-19 C | +1 | 1.6726 × 10-24 g | 1.0073 |
Neutron | 0 | 0 | 1.6749 × 10-24 g | 1.0087 |
Electron | -1.6022 × 10-19 C | -1 | 9.1094 × 10-28 g | 0.00054858 |

Example: Calculating Photon Energy
For radiation of wavelength 242.4 nm, the energy of one photon is:
Where J·s, m/s, m
Additional info: These notes provide foundational knowledge for understanding electronic structure, atomic theory, and quantum mechanics, which are essential for further study in general chemistry.
