 Back
BackBalancing Chemical Equations and Identifying Redox Species
Study Guide - Smart Notes

Q11. Balance each chemical equation:
Background
Topic: Chemical Equations & Balancing
This question tests your ability to balance chemical equations, ensuring the same number of each type of atom appears on both sides of the equation. This is a fundamental skill in general chemistry, reflecting the law of conservation of mass.
Key Terms and Concepts:
Reactants: Substances present before the reaction.
Products: Substances formed as a result of the reaction.
Balancing: Adjusting coefficients to ensure equal numbers of each atom on both sides.
Step-by-Step Guidance
For each equation, list all elements present on both sides of the equation.
Count the number of atoms of each element on both sides.
Adjust coefficients (the numbers in front of compounds or elements) to balance one element at a time, starting with the most complex molecule.
Repeat the process for each element, checking after each adjustment to ensure previous elements remain balanced.
Continue until all elements are balanced. Double-check your work by recounting all atoms on both sides.
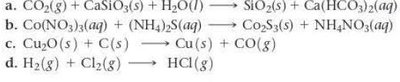
Try solving on your own before revealing the answer!
Q13. Assign oxidation states to the atoms in the following reaction and identify oxidized/reduced species and agents:
Background
Topic: Redox Reactions & Oxidation States
This question tests your understanding of how to assign oxidation states to elements in compounds and identify which species are oxidized or reduced in a redox reaction. You are also asked to identify the oxidizing and reducing agents.
Key Terms and Concepts:
Oxidation State: A number assigned to an element in chemical combination that represents the number of electrons lost or gained.
Oxidized Species: The species that loses electrons (increase in oxidation state).
Reduced Species: The species that gains electrons (decrease in oxidation state).
Oxidizing Agent: The species that causes oxidation (itself is reduced).
Reducing Agent: The species that causes reduction (itself is oxidized).
Step-by-Step Guidance
Assign oxidation states to each atom in all reactants and products using standard rules (e.g., group 1 metals are +1, oxygen is usually -2, hydrogen is +1, etc.).
Compare the oxidation states of each element before and after the reaction to determine which species' oxidation state increases (oxidized) and which decreases (reduced).
Identify the oxidizing agent (the species that is reduced) and the reducing agent (the species that is oxidized).

Try solving on your own before revealing the answer!
