 Back
BackBasic Concepts of Chemical Bonding: Structured Study Notes
Study Guide - Smart Notes

Basic Concepts of Chemical Bonding
Types of Chemical Bonds
Chemical bonds are the forces that hold atoms together in compounds. The three primary types are ionic, covalent, and metallic bonds.
Ionic Bonds: Formed by the electrostatic attraction between oppositely charged ions, typically between metals and nonmetals. Example: NaCl.
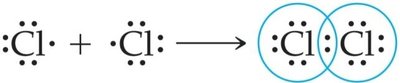
Covalent Bonds: Result from the sharing of electrons between atoms, usually between nonmetals. Example: Cl2.
Metallic Bonds: Involve metal nuclei floating in a 'sea of electrons,' characteristic of metallic elements. Example: Na.

Key Point: The nature of the bond affects the physical properties of the substance, such as melting point, conductivity, and solubility.
Lewis Symbols and the Octet Rule
Lewis symbols (or electron-dot symbols) are a visual representation of valence electrons around an atom's symbol. The octet rule states that atoms tend to gain, lose, or share electrons to achieve eight valence electrons, resembling the electron configuration of noble gases.
Lewis Symbols: Electrons are placed on four sides around the element's symbol.
Octet Rule: Atoms achieve stability by having eight valence electrons.


Example: Sodium (Na) has one valence electron, while chlorine (Cl) has seven. Both achieve an octet by forming NaCl.
Thermodynamics: Enthalpy and Energy
Enthalpy (H) is the sum of the internal energy (E) and the product of pressure and volume (PV) of a system. The change in enthalpy (ΔH) is measured during chemical reactions.
For most reactions, is small, so .
Endothermic reactions: (heat absorbed)
Exothermic reactions: (heat released)
Example: Formation of water: , kJ (exothermic).
Ionic Bonding and Lattice Energy
Ionic bonding involves the transfer of electrons from a metal to a nonmetal, resulting in the formation of ions. The stability of ionic compounds is largely due to lattice energy, the energy required to separate one mole of a solid ionic compound into gaseous ions.
Lattice Energy (): Depends on ion charge and size.
Coulomb's Law: , where and are ion charges, is the distance between ions, and is a constant.
Higher charge and smaller radius increase lattice energy.


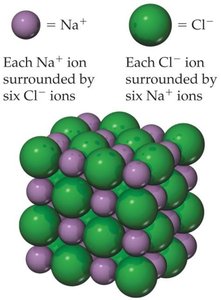
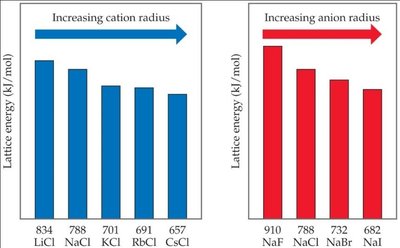
Example: NaCl formation is highly exothermic ( kJ/mol), and its crystal structure is regular and stable.
Covalent Bonding
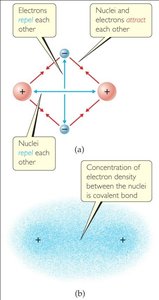
Covalent bonds are formed by sharing pairs of electrons between atoms. The strength and length of the bond depend on the number of shared electron pairs and the atoms involved.
Single Bond: One pair of electrons shared (e.g., H-H).
Double Bond: Two pairs shared (e.g., O=O).
Triple Bond: Three pairs shared (e.g., N≡N).
Bonding involves attractions and repulsions between electrons and nuclei.
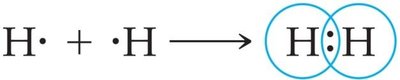
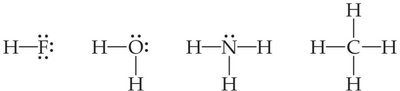
Example: The Lewis structure for methane (CH4) shows four single covalent bonds between carbon and hydrogen.
Bond Polarity and Electronegativity
Bond polarity describes the unequal sharing of electrons in a covalent bond. Electronegativity is the ability of an atom to attract electrons in a bond. The difference in electronegativity determines whether a bond is nonpolar, polar, or ionic.
Nonpolar Covalent: Electrons shared equally (e.g., F2).
Polar Covalent: Electrons shared unequally (e.g., HF).
Ionic: Large difference leads to electron transfer (e.g., NaBr).
Electronegativity increases across a period and decreases down a group.
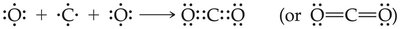
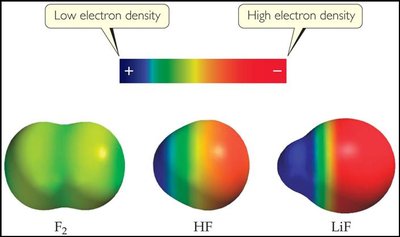
Example: The bond in HF is polar because fluorine is much more electronegative than hydrogen.
Drawing Lewis Structures and Formal Charge
Lewis structures represent the arrangement of electrons in molecules. Formal charge helps determine the most stable structure.
Sum valence electrons from all atoms.
Connect atoms with single bonds; central atom is least electronegative.
Complete octets for all atoms (except H).
Place leftover electrons on the central atom.
Form multiple bonds if needed for octet.
Calculate formal charges:
Example: The best Lewis structure has formal charges closest to zero and negative charges on the most electronegative atoms.
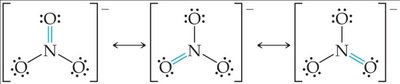
Resonance Structures
Some molecules cannot be represented by a single Lewis structure. Resonance structures are multiple valid Lewis structures for a molecule, and the actual structure is a hybrid of these.
Resonance occurs in molecules like O3, NO3-, SO3, and benzene.
Bonds in resonance structures are equivalent in length and strength.
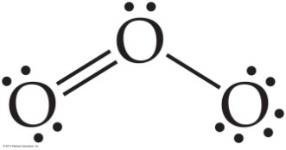
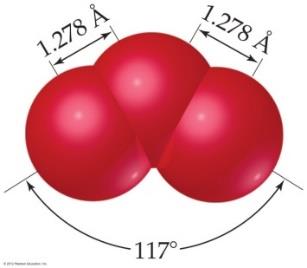

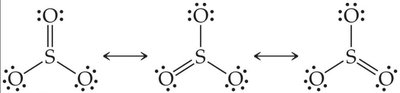
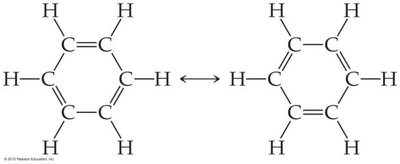
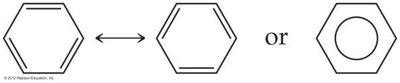
Example: Ozone (O3) has two resonance structures, but experimentally both bonds are identical.
Exceptions to the Octet Rule
Some molecules do not follow the octet rule. Exceptions include:
Odd Number of Electrons: Example: NO, NO2, ClO2.
Less than an Octet: Common in compounds of groups 1A, 2A, and 3A (e.g., BF3).
More than an Octet: Atoms from period 3 and beyond can have expanded octets (e.g., PF5, SF4, AsF6-).
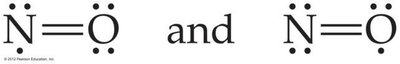
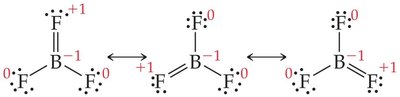
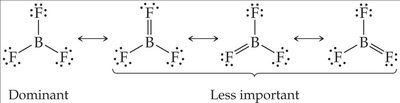
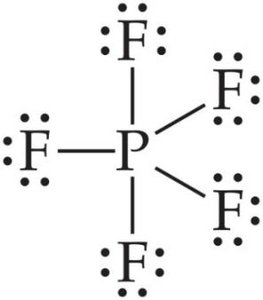
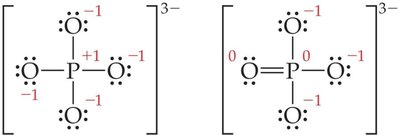
Note: Expanding the octet can help minimize formal charges, especially for central atoms in period 3 or below.
Strengths of Covalent Bonds: Bond Enthalpy
Bond enthalpy (D) is the energy required to break a specific covalent bond in one mole of a gaseous substance. It is always positive because bond breaking is endothermic.
Bond enthalpy depends on the number of electrons, atom sizes, and electronegativities.
Multiple bonds are stronger and shorter than single bonds.
Bond enthalpy is used to estimate reaction enthalpy:
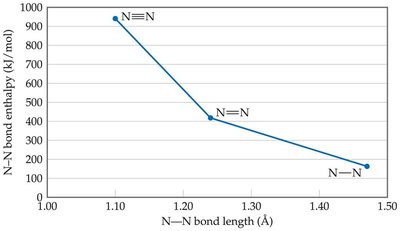
Example: The bond enthalpy for Cl2 is 242 kJ. For methane, the average C-H bond enthalpy is 415 kJ.
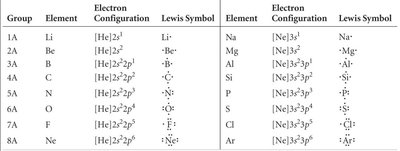
Summary Table: Lewis Symbols for Main Group Elements
This table summarizes the electron configurations and Lewis symbols for main group elements.
Group | Element | Electron Configuration | Lewis Symbol |
|---|---|---|---|
1A | Li | [He]2s1 | Li· |
2A | Be | [He]2s2 | Be·· |
3A | B | [He]2s22p1 | B··· |
4A | C | [He]2s22p2 | C···· |
5A | N | [He]2s22p3 | N····· |
6A | O | [He]2s22p4 | O······ |
7A | F | [He]2s22p5 | F······· |
8A | Ne | [He]2s22p6 | Ne········ |
Na | [Ne]3s1 | Na· | |
Mg | [Ne]3s2 | Mg·· | |
Al | [Ne]3s23p1 | Al··· | |
Si | [Ne]3s23p2 | Si···· | |
P | [Ne]3s23p3 | P····· | |
S | [Ne]3s23p4 | S······ | |
Cl | [Ne]3s23p5 | Cl······· | |
Ar | [Ne]3s23p6 | Ar········ |
Additional info: These notes expand on brief points from the original material, providing definitions, examples, and academic context for each topic. All included images are directly relevant to the explanations and reinforce key concepts.
