Back
BackC117 Exam 3 Practice – Step-by-Step Chemistry Guidance
Study Guide - Smart Notes

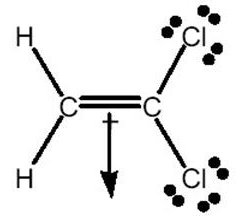
Q1. Which drawing best shows the direction of the dipole moment in H2C = CCl2?
Background
Topic: Molecular Polarity and Dipole Moments
This question tests your understanding of how to determine the direction of the molecular dipole moment based on the electronegativities of the atoms in a molecule.

Key Terms and Concepts:
Dipole Moment: A vector quantity that points from the positive (less electronegative) to the negative (more electronegative) region of a molecule.
Electronegativity: The ability of an atom to attract electrons in a bond. Chlorine is more electronegative than carbon, which is more electronegative than hydrogen.
Step-by-Step Guidance
Draw the Lewis structure for H2C = CCl2. Identify the atoms attached to each carbon.
Compare the electronegativities: Cl > C > H. The dipole moment will point toward the more electronegative atoms (the Cl atoms).
Consider the overall molecular geometry and the direction in which the electron density is pulled due to the Cl atoms.
Look at the provided diagrams and identify which arrow points toward the region with the highest electron density (toward the Cl atoms).
Try solving on your own before revealing the answer!
Final Answer: Diagram B (arrow points downward toward the Cl atoms)
The dipole moment points from the hydrogen side toward the chlorine side because chlorine is much more electronegative than both carbon and hydrogen.
Q2. Which of the following is the correct order of boiling points for KNO3, C2H5OH, C3H8, and He?
Background
Topic: Intermolecular Forces and Boiling Points
This question tests your ability to compare boiling points based on the types and strengths of intermolecular forces present in different substances.
Key Terms and Concepts:
Boiling Point: The temperature at which a substance changes from liquid to gas.
Intermolecular Forces: Includes dispersion (London) forces, dipole-dipole interactions, hydrogen bonding, and ionic forces.
General Trend: Stronger intermolecular forces lead to higher boiling points.
Step-by-Step Guidance
Identify the type of intermolecular forces present in each compound:
KNO3: Ionic forces (very strong)
C2H5OH: Hydrogen bonding
C3H8: Dispersion forces
He: Dispersion forces (very weak, as He is a noble gas)
Rank the compounds from weakest to strongest intermolecular forces.
Arrange the compounds in order of increasing boiling point based on the strength of their intermolecular forces.
Try solving on your own before revealing the answer!
Final Answer: He < C3H8 < C2H5OH < KNO3
KNO3 has the highest boiling point due to ionic forces, while He has the lowest due to very weak dispersion forces.
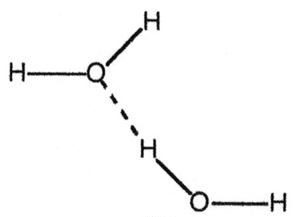
Q3. Which drawing best represents hydrogen bonding?
Background
Topic: Hydrogen Bonding
This question tests your ability to recognize hydrogen bonds in molecular diagrams, especially between molecules containing O-H, N-H, or F-H bonds.
Key Terms and Concepts:
Hydrogen Bond: A strong dipole-dipole attraction between a hydrogen atom bonded to a highly electronegative atom (O, N, or F) and a lone pair on another electronegative atom.
Step-by-Step Guidance
Look for a dashed line between a hydrogen atom attached to O, N, or F and another O, N, or F atom.
Check that the hydrogen bond is between two different molecules (intermolecular), not within the same molecule (intramolecular).
Identify which diagram shows this correct arrangement.
Try solving on your own before revealing the answer!
Final Answer: Diagram A (dashed line between H of one water molecule and O of another)
This diagram correctly shows a hydrogen bond between two water molecules.