 Back
BackChapter 15: Chemical Equilibrium – Structured Study Notes
Study Guide - Smart Notes

Chemical Equilibrium
Introduction to Chemical Equilibrium
Chemical equilibrium is a fundamental concept in chemistry describing the state in which the rates of the forward and reverse reactions are equal, resulting in constant concentrations of reactants and products. This chapter explores the principles, calculations, and applications of equilibrium in chemical systems.
Hemoglobin and Equilibrium
Hemoglobin Equilibrium System
Hemoglobin (Hb) is a protein in red blood cells that binds oxygen (O2) to facilitate its transport. The reaction is:
Hb + O2 → HbO2
The system is in dynamic equilibrium, meaning the concentrations of Hb, O2, and HbO2 are interdependent.
The equilibrium constant, K, quantifies the relative concentrations at equilibrium.
A large K indicates a product-favored equilibrium.
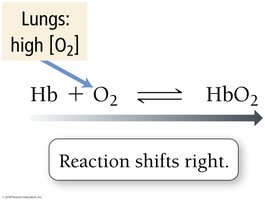
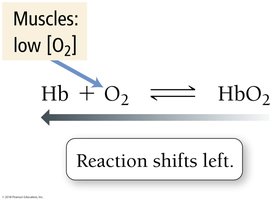
O2 Transport: Lungs vs. Muscles
In the lungs, high O2 concentration shifts equilibrium right, forming more HbO2.
In muscles, low O2 concentration shifts equilibrium left, releasing O2 from HbO2.
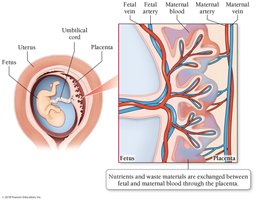
Fetal Hemoglobin
Fetal hemoglobin (HbF) has a higher equilibrium constant than adult hemoglobin, making it more efficient at binding O2.
O2 is transferred from maternal to fetal hemoglobin in the placenta.
Dynamic Equilibrium
Definition and Characteristics
Dynamic equilibrium occurs when the rates of the forward and reverse reactions are equal, and concentrations remain constant.
Reactant concentrations decrease as products form.
Reverse reaction rate increases as product concentration increases.
Equilibrium does not mean equal amounts of reactants and products.

The Equilibrium Constant (K)
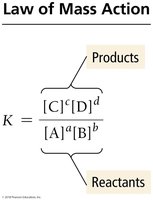
Law of Mass Action
The equilibrium constant, K, relates the concentrations of products and reactants at equilibrium for a given reaction:
For a general reaction: aA + bB → cC + dD
The expression is:
K is dimensionless and always products over reactants.
Writing Equilibrium Constant Expressions
For 2 N2O5(g) → 4 NO2(g) + O2(g):
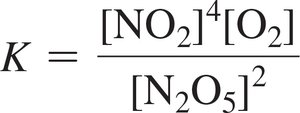
Interpreting the Value of K
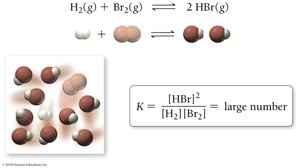
If K >> 1: Product-favored equilibrium (more products than reactants).
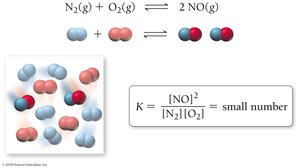
If K << 1: Reactant-favored equilibrium (more reactants than products).
Manipulating Equilibrium Constants
Reversing the reaction inverts K:
Multiplying coefficients by n raises K to the nth power.
Adding reactions multiplies their equilibrium constants:
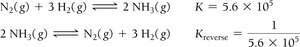
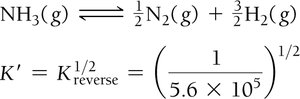
Equilibrium Constants for Gaseous Reactions
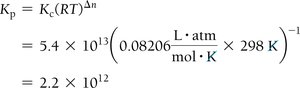
Kc and Kp
Kc uses concentrations (mol/L), Kp uses partial pressures (atm).
Relationship: , where Δn = moles of gaseous products - moles of gaseous reactants.


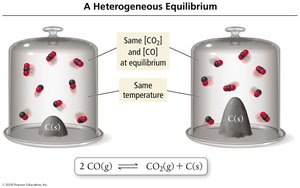
Heterogeneous Equilibria
Solids and Liquids in Equilibrium Expressions
Concentrations of pure solids and liquids are constant and omitted from equilibrium expressions.
Example: For CaCO3(s) → CaO(s) + CO2(g),
Calculating Equilibrium Constants
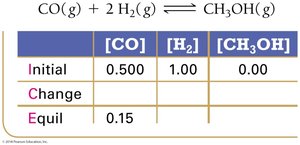
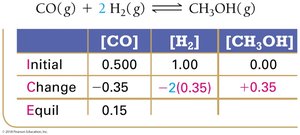
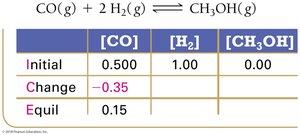
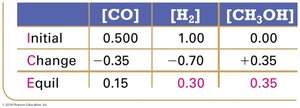
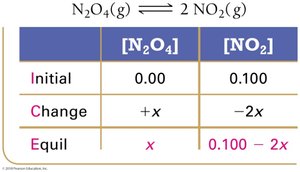
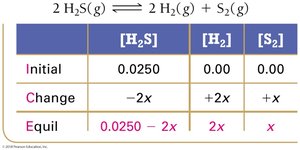
Using Experimental Data and ICE Tables
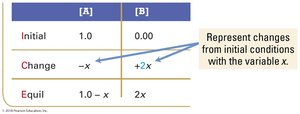
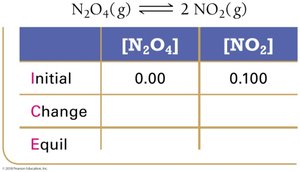
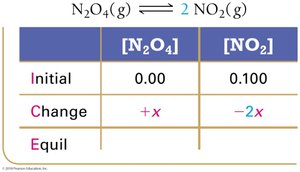
ICE tables (Initial, Change, Equilibrium) are used to organize and calculate equilibrium concentrations.
Step 1: Set up ICE table with initial concentrations.
Step 2: Calculate changes based on stoichiometry.
Step 3: Sum columns to find equilibrium concentrations.
Step 4: Substitute into equilibrium expression to solve for K.
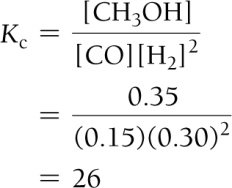
The Reaction Quotient (Q)
Definition and Use
Q is calculated like K but with current (not equilibrium) concentrations.
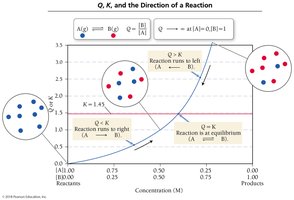
Compare Q to K to predict reaction direction:
Q > K: Reaction shifts left (reverse).
Q < K: Reaction shifts right (forward).
Q = K: System is at equilibrium.
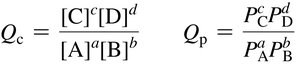
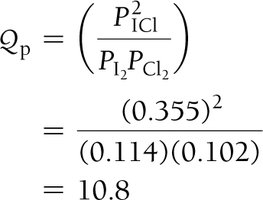
Finding Equilibrium Concentrations Using ICE Tables
Procedure and Quadratic Equations
Set up ICE table, define changes with variable x.
Write equilibrium expressions in terms of x.
Solve for x using algebra or quadratic formula.
Check for physically realistic answers (no negative concentrations).
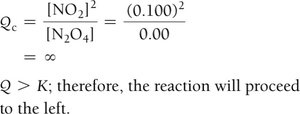
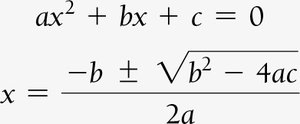
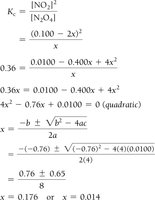
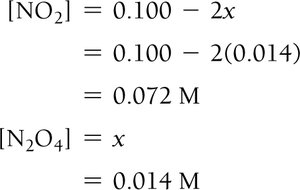
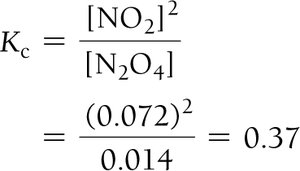
Approximations in Equilibrium Calculations
5% Rule
If x is less than 5% of the initial concentration, it can be neglected for simplification.
Used when K is very small and equilibrium favors reactants.
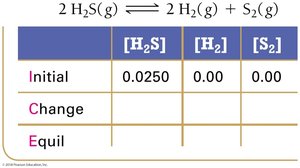
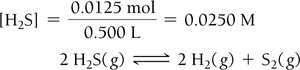
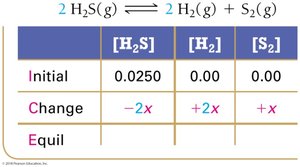
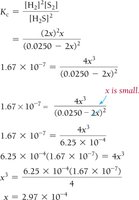

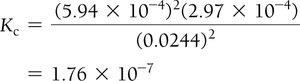
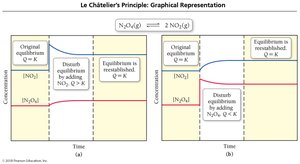
Le Châtelier’s Principle
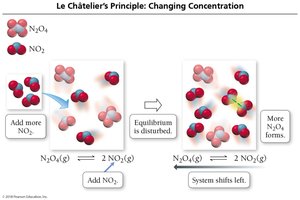
Disturbing and Restoring Equilibrium
If a system at equilibrium is disturbed, it shifts to minimize the disturbance.
Adding reactants shifts equilibrium right; removing reactants shifts it left.
Adding products shifts equilibrium left; removing products shifts it right.
Adding solids or liquids does not affect equilibrium.
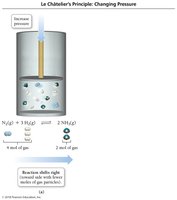
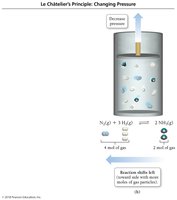
Effect of Volume and Pressure Changes
Decreasing volume increases pressure and shifts equilibrium toward the side with fewer gas molecules.
Increasing volume decreases pressure and shifts equilibrium toward the side with more gas molecules.
Adding an inert gas at constant volume has no effect.
Effect of Temperature Changes
Exothermic reactions: Heat is a product. Increasing temperature shifts equilibrium left and decreases K.
Endothermic reactions: Heat is a reactant. Increasing temperature shifts equilibrium right and increases K.
Effect of Catalysts
Catalysts increase the rate of both forward and reverse reactions equally.
Catalysts do not affect the position of equilibrium.
