 Back
BackChapter 19: Chemical Thermodynamics – Study Notes
Study Guide - Smart Notes

Chemical Thermodynamics
Introduction to Thermodynamics
Chemical thermodynamics studies the energy changes and spontaneity of chemical processes. It helps predict whether a reaction will occur under given conditions and explains the direction of energy flow in chemical systems.
First Law of Thermodynamics: Energy cannot be created or destroyed; it can only be transferred or converted from one form to another. The total energy of the universe remains constant.
Enthalpy (H): The heat absorbed or released by a system at constant pressure.
Entropy (S): A measure of the randomness or disorder in a system. Both enthalpy and entropy are crucial in determining whether a process is spontaneous.
Spontaneous Processes
Definition and Characteristics
A spontaneous process proceeds without outside assistance. If a process is spontaneous in one direction, it is nonspontaneous in the reverse direction (e.g., an egg breaking is spontaneous, but reassembling is not).
Spontaneity does not imply speed; some spontaneous reactions are slow.
Nonspontaneous does not mean impossible; energy input can drive nonspontaneous reactions.
Temperature and pressure can affect spontaneity (e.g., ice melting/freezing depends on temperature).
Reversible and Irreversible Processes
Reversible process: The system and surroundings can be returned to their original states by exactly reversing the process. This maximizes the work done by the system.
Irreversible process: Cannot be undone by exactly reversing the change. All real, spontaneous processes are irreversible.
Entropy and the Laws of Thermodynamics
Second Law of Thermodynamics
The Second Law of Thermodynamics states that the entropy of the universe increases in any spontaneous process. Entropy is a state function and can be calculated by heat transfer at a given temperature.
Spontaneous processes increase the disorder (entropy) of the universe.
Third Law of Thermodynamics
The Third Law of Thermodynamics states that the entropy of a pure crystalline substance at absolute zero (0 K) is zero. At this temperature, there is only one possible microstate (perfect order).
Statistical Thermodynamics and Microstates
Microstate: A specific arrangement of the positions and energies of molecules in a system.
The number of microstates (W) is related to entropy by the Boltzmann equation:
Where S is entropy, k is the Boltzmann constant, and W is the number of microstates.
More microstates mean higher entropy (more disorder).
Molecular Motions and Entropy
Translational motion: Movement of the entire molecule from one place to another.
Vibrational motion: Periodic motion of atoms within a molecule.
Rotational motion: Rotation of the molecule about an axis.
Entropy increases with more possible molecular motions (degrees of freedom).
Factors Affecting Entropy
Increasing volume increases entropy (more positions for molecules).
Increasing temperature increases entropy (greater distribution of molecular speeds).
Increasing the number of atoms/molecules increases entropy (more degrees of freedom).
Entropy and Physical States
Entropy increases with the freedom of motion of molecules:
Gases > Liquids > Solids
Processes that increase entropy:
Formation of gases from solids or liquids
Formation of liquids or solutions from solids
Increase in the number of gas molecules during a reaction
Entropy Changes in Chemical Reactions
Calculating Entropy Changes
Entropy changes for reactions can be calculated similarly to enthalpy changes:
S° is the standard molar entropy.
n and m are the stoichiometric coefficients from the balanced equation.
Standard molar entropy values increase with molar mass and number of atoms.
Entropy Changes in Surroundings
Heat flow into or out of the system changes the entropy of the surroundings.
For an isothermal process at constant pressure:
For spontaneous processes, the total entropy change (system + surroundings) is positive.
Gibbs Free Energy
Definition and Equation
Gibbs Free Energy (G) combines enthalpy and entropy to predict spontaneity:
If , the process is spontaneous.
If , the system is at equilibrium.
If , the process is nonspontaneous (spontaneous in the reverse direction).
Standard Free Energy of Formation
Analogous to standard enthalpy of formation, the standard free energy of formation () is used to calculate the free energy change for reactions:
n and m are stoichiometric coefficients.
Free Energy and Temperature
Spontaneity depends on the signs and magnitudes of and , and the temperature (T):
ΔH | ΔS | Spontaneity |
|---|---|---|
- | + | Always spontaneous |
+ | - | Never spontaneous |
- | - | Spontaneous at low T |
+ | + | Spontaneous at high T |
Free Energy and the Equilibrium Constant
Relationship Between ΔG and K
The standard free energy change is related to the equilibrium constant (K):
At equilibrium ():
R is the universal gas constant (8.314 J/mol·K).
If Q < K, the reaction shifts right (toward products).
If Q > K, the reaction shifts left (toward reactants).
Driving Nonspontaneous Reactions
Coupled Reactions in Biological Systems
Many important reactions are nonspontaneous as written. In biological systems, nonspontaneous reactions are driven by coupling them to spontaneous ones, such as the hydrolysis of ATP.
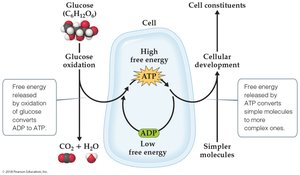
Free energy released by oxidation of glucose is used to convert ADP to ATP.
ATP hydrolysis provides free energy to drive cellular processes that would otherwise be nonspontaneous.
