 Back
BackChapter 19: Free Energy and Thermodynamics – Study Notes
Study Guide - Smart Notes

Free Energy and Thermodynamics
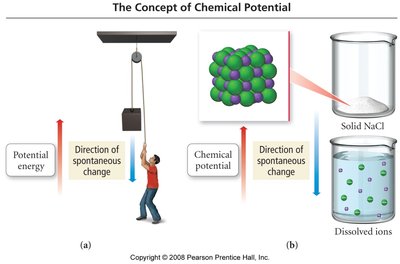
Spontaneity and Chemical Potential
Spontaneity in chemical processes refers to whether a reaction or change occurs naturally without continuous external intervention. The concept of chemical potential is analogous to potential energy in physics and helps predict the direction of spontaneous change in chemical systems.
Spontaneous process: Occurs without ongoing outside intervention.
Non-spontaneous process: Requires continuous energy input to proceed.
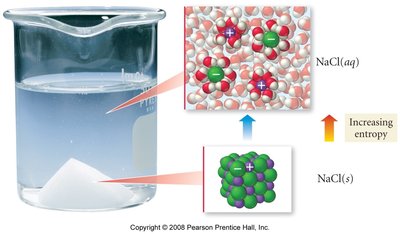
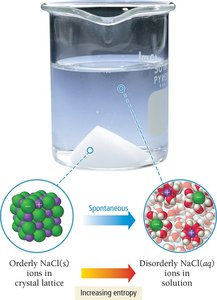
Example: Dissolving NaCl in water is spontaneous due to the increase in entropy (randomness) as ions disperse in solution.
Examples of Spontaneous and Nonspontaneous Processes
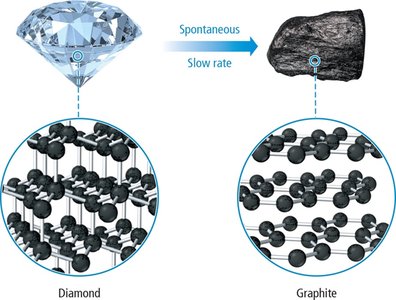
Some processes are spontaneous but may occur at extremely slow rates. For example, the conversion of diamond to graphite is thermodynamically spontaneous but kinetically very slow.
Diamond to graphite: Spontaneous but occurs over geological timescales.
Exothermic reactions: Often spontaneous, but not always (other factors like entropy matter).
Enthalpy, Entropy, and Spontaneity
Enthalpy Changes (ΔH)
Enthalpy (H) is the heat content of a system at constant pressure. Changes in enthalpy (ΔH) can be:
Exothermic (ΔH < 0): Releases heat to surroundings; often favors spontaneity.
Endothermic (ΔH > 0): Absorbs heat from surroundings; may still be spontaneous if entropy increases sufficiently.



Entropy (S) and the Second Law of Thermodynamics
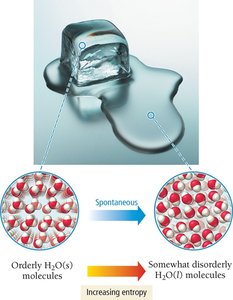
Entropy (S) is a measure of the disorder or randomness of a system. The second law of thermodynamics states that for any spontaneous process, the entropy of the universe increases:
ΔS = S_{final} - S_{initial}
Boltzmann Equation: where J/K and is the number of microstates.
For a process to be spontaneous:

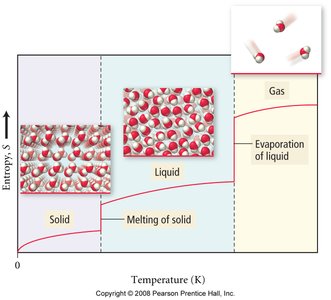
Entropy Changes with State and Temperature
Entropy increases with:
Phase changes:
Increasing temperature
Increasing volume
Mixing substances
Factors Affecting Spontaneity
Two main thermodynamic factors determine spontaneity:
ΔH (Enthalpy change): Related to heat exchange and bond energies.
ΔS (Entropy change): Related to disorder and energy dispersal.
Spontaneous process:

Quantifying Entropy and Enthalpy Changes
Entropy Change in the Surroundings
The entropy change in the surroundings is related to the heat exchanged and the temperature:
Exothermic reactions () increase ; endothermic reactions decrease it.
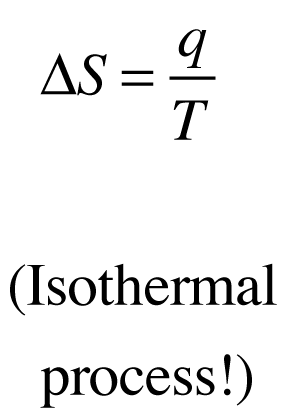
Gibbs Free Energy (ΔG)
Definition and Equation
Gibbs free energy (G) combines enthalpy and entropy to predict spontaneity at constant temperature and pressure:
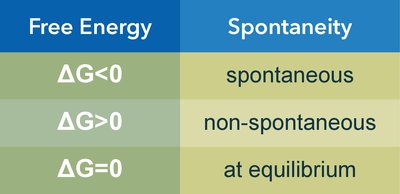
If , the process is spontaneous.
If , the process is non-spontaneous.
If , the system is at equilibrium.
Temperature Dependence of Spontaneity
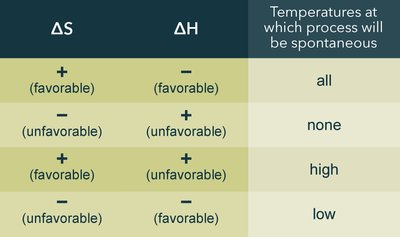
The sign of ΔH and ΔS determines whether a process is spontaneous at all, some, or no temperatures:
ΔS | ΔH | Spontaneity |
|---|---|---|
+ | - | All temperatures |
- | + | None |
+ | + | High temperatures |
- | - | Low temperatures |
Entropy and Free Energy in Chemical Reactions
Standard Entropy and Free Energy Changes
Standard entropy () and free energy () changes are calculated using tabulated values:
Hess's Law for Free Energy
Free energy changes for overall reactions can be determined by combining stepwise reactions, similar to Hess's Law for enthalpy.
Free Energy and Nonstandard Conditions
Free Energy Under Nonstandard Conditions
When reactants and products are not in standard states, the free energy change is:
Where is the reaction quotient, is the gas constant, and is temperature in Kelvin.
Free Energy and Equilibrium
Relationship Between ΔG and the Equilibrium Constant (K)
At equilibrium, and (the equilibrium constant):
If , (products favored at equilibrium).
If , (reactants favored at equilibrium).
Temperature Dependence of the Equilibrium Constant
The equilibrium constant changes with temperature according to:
A plot of versus yields a straight line (van 't Hoff equation).
Entropy: Molecular View and Trends
Microstates and Entropy
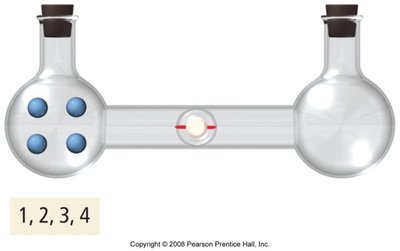
Entropy increases with the number of possible microstates (ways to arrange particles):
Mixing increases entropy.
Phase changes (solid to liquid to gas) increase entropy.








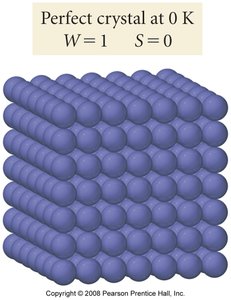
Third Law of Thermodynamics
The third law states that the entropy of a perfect crystal at absolute zero (0 K) is zero:
At 0 K, only one microstate exists (), so .
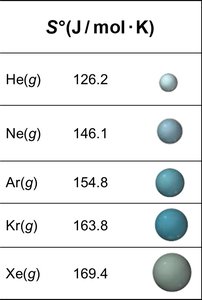
Trends in Standard Entropies
Heavier atoms and more complex molecules have higher entropy.
Less constrained structures (e.g., graphite vs. diamond) have higher entropy.
Dissolved ions have higher entropy than solids.


Summary Table: Spontaneity, Enthalpy, and Entropy
ΔS | ΔH | Spontaneity |
|---|---|---|
+ | - | All temperatures |
- | + | None |
+ | + | High temperatures |
- | - | Low temperatures |

Practice Problems and Applications
Calculate and using standard values.
Predict spontaneity at different temperatures using the sign of ΔH and ΔS.
Relate to the equilibrium constant .
Additional info: These notes integrate key concepts from thermodynamics, including the laws of thermodynamics, entropy, enthalpy, and free energy, with practical examples and molecular-level illustrations to reinforce understanding for general chemistry students.
