 Back
BackChapter 21: Radioactivity and Nuclear Chemistry – Study Notes
Study Guide - Smart Notes

Radioactivity and Nuclear Chemistry
Introduction to Nuclear Chemistry
Nuclear chemistry focuses on the changes that occur within the nucleus of an atom, distinguishing it from chemical processes that involve only the electrons. Nuclear reactions can result in the transformation of one element into another and are responsible for phenomena such as radioactivity, nuclear fission, and fusion.
Chemical vs. Nuclear Processes
Chemical reactions involve changes in the electronic structure of atoms (gaining, losing, or sharing electrons) without altering the nucleus.
Nuclear reactions involve changes in the nucleus, including the number of protons and neutrons, which can result in the formation of different elements.
Discovery of Radioactivity
Marie Curie and the Nature of Radioactivity
Marie Curie discovered that certain elements emit rays from their nuclei, a phenomenon she named radioactivity. Her work led to the discovery of new elements, such as radium and polonium, and earned her Nobel Prizes in Physics and Chemistry.
Types of Radioactivity
Radioactivity is the spontaneous emission of high-energy particles or gamma rays from an unstable atomic nucleus. These emissions can ionize matter, penetrate materials, and cause phosphorescent substances to glow.
Ionizing Power: The ability of radiation to ionize other molecules and atoms. Alpha particles have the highest ionizing power.
Penetrating Power: The ability of radiation to pass through matter. Gamma rays are the most penetrating, while alpha particles are the least.

Important Atomic Symbols
Understanding nuclear chemistry requires familiarity with the symbols for subatomic particles and nuclear notation.
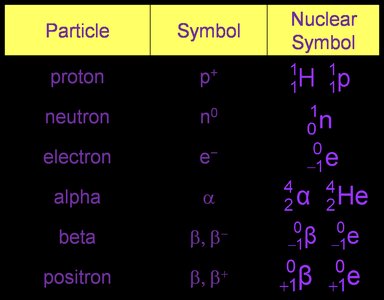
Nuclear Notation
Nuclear notation expresses the identity of a nuclide using its atomic number (Z), mass number (A), and chemical symbol (X):

Transmutation and Nuclear Equations
During radioactive decay, a parent nuclide transforms into a daughter nuclide. Nuclear equations must conserve both atomic number and mass number.
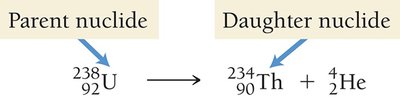
Types of Radioactive Decay
Alpha Decay (α-decay)
Alpha decay occurs when an unstable nucleus emits an alpha particle (two protons and two neutrons, identical to a helium-4 nucleus). This process decreases the atomic number by 2 and the mass number by 4. Alpha particles are highly ionizing but have low penetrating power.
Beta Decay (β-decay)
Beta decay involves the emission of an electron from the nucleus. The atomic number increases by 1, while the mass number remains unchanged. Beta particles are more penetrating than alpha particles but less ionizing.
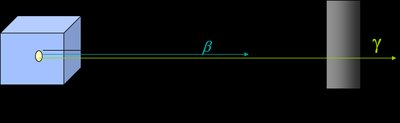
Gamma Emission (γ-emission)
Gamma rays are high-energy photons emitted from the nucleus, usually following another type of decay. Gamma emission does not change the atomic or mass number but is the most penetrating and least ionizing form of radiation.
Positron Emission
A positron is the antiparticle of the electron, with a charge of +1. When a nucleus emits a positron, its atomic number decreases by 1, but the mass number remains the same. This process results from a proton converting into a neutron.
Electron Capture
Electron capture occurs when an inner orbital electron is drawn into the nucleus, where it combines with a proton to form a neutron. The atomic number decreases by 1, and the mass number remains unchanged. No particle is emitted, but the atom changes identity.
Kinetics of Radioactive Decay
Radioactive decay follows first-order kinetics, meaning the rate depends on the number of radioactive nuclei present. The decay rate is given by:
where k is the decay constant and N is the number of radioactive nuclei.
The half-life (t1/2) is the time required for half of the radioactive nuclei to decay:
The shorter the half-life, the more rapidly the sample decays.
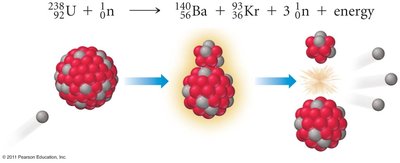
Nuclear Fission
Fission is the splitting of a large nucleus into smaller nuclei when struck by a neutron, releasing a tremendous amount of energy. This process is the basis for nuclear power and atomic bombs.
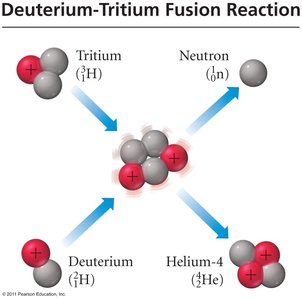
Nuclear Fusion
Fusion is the process by which small nuclei combine to form a larger nucleus, overcoming electrostatic repulsion at extremely high temperatures. Fusion releases even more energy per gram than fission and powers the sun and hydrogen bombs. However, achieving controlled fusion for electricity generation is currently very costly and technologically challenging.
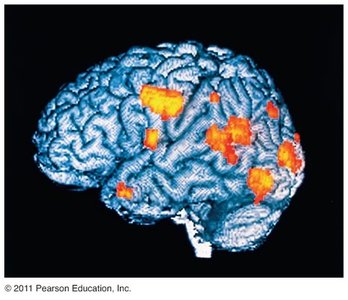
Applications of Radioactivity in Medicine
Radiotracers and PET Scans
Radioactive nuclides, or radiotracers, are used in medicine to track the movement of substances within the body. Positron emission tomography (PET) scans employ positron-emitting nuclides, such as fluorine-18, to visualize brain activity and diagnose diseases.
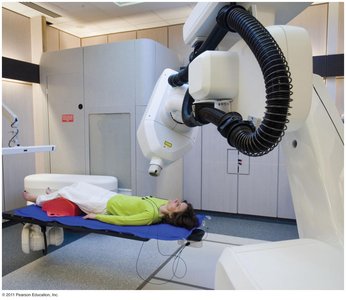
Gamma Ray Treatment (Radiotherapy)
Gamma rays are used in radiotherapy to treat cancer by targeting and destroying malignant cells with high-energy radiation.
Summary Table: Types of Radioactive Decay
Decay Type | Particle Emitted | Change in Atomic Number | Change in Mass Number | Penetrating Power | Ionizing Power |
|---|---|---|---|---|---|
Alpha (α) | Helium-4 nucleus () | -2 | -4 | Low | High |
Beta (β) | Electron () | +1 | 0 | Moderate | Moderate |
Gamma (γ) | Photon | 0 | 0 | High | Low |
Positron | Positron () | -1 | 0 | Moderate | Moderate |
Electron Capture | None (electron absorbed) | -1 | 0 | N/A | N/A |
Key Equations
First-order decay rate:
Half-life:
Comparison: Nuclear Fission vs. Fusion
Process | Description | Energy Released | Example |
|---|---|---|---|
Fission | Splitting of a large nucleus into smaller nuclei | High (per event) | Nuclear power plants, atomic bombs |
Fusion | Combining of small nuclei to form a larger nucleus | Very high (per gram) | Sun, hydrogen bomb |
