 Back
BackChapter 6: Electronic Structure of Atoms – Study Notes
Study Guide - Smart Notes

Electronic Structure of Atoms
6.1 The Wave Nature of Light
Electromagnetic radiation, also known as radiant energy, exhibits wave-like behavior. This includes visible light, radio waves, and infrared radiation. Understanding the properties of waves is essential for describing the behavior of light and its interaction with matter.
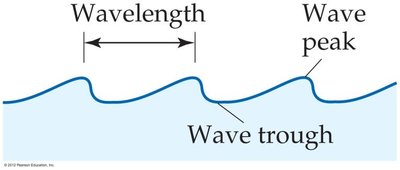
Wavelength (\(\lambda\)): The distance between two consecutive peaks (crests) or troughs in a wave.
Frequency (\(\nu\)): The number of complete wavelengths that pass a given point per second, measured in hertz (Hz, or s-1).
Speed of Light (\(c\)): All electromagnetic radiation travels at \(3.00 \times 10^8\) m/s in a vacuum.
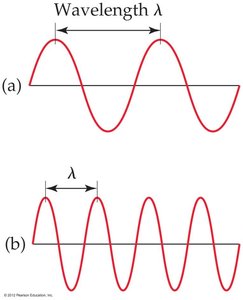
There is an inverse relationship between wavelength and frequency: as one increases, the other decreases.

The mathematical relationship is given by:
Where \(c\) is the speed of light, \(\lambda\) is wavelength, and \(\nu\) is frequency.
High-frequency waves have short wavelengths, and low-frequency waves have long wavelengths.
The electromagnetic spectrum encompasses all types of electromagnetic radiation, from gamma rays to radio waves. The visible region is only a small part of this spectrum.
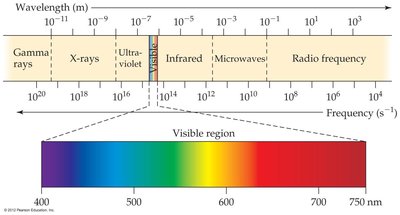
Frequency and energy are directly related, while frequency and wavelength are inversely related.
6.2 Quantized Energy and Photons
The wave model of light cannot explain all phenomena, such as the quantization of energy and the photoelectric effect.
Quantization of Energy (Planck's Hypothesis): Atoms absorb or emit energy in discrete amounts called quanta.
\(E\): energy of a quantum (Joules)
\(h\): Planck's constant (\(6.626 \times 10^{-34}\) J·s)
\(\nu\): frequency (Hz)
Photoelectric Effect (Einstein): Light energy is quantized in particles called photons. When photons of sufficient energy strike a metal, electrons are emitted.
Energy can only be absorbed or emitted in whole-number multiples of \(h\nu\).
6.3 Line Spectra and the Bohr Model
When radiation is separated into its component wavelengths, a spectrum is produced. A continuous spectrum contains all wavelengths, while a line spectrum contains only specific wavelengths.
Monochromatic: single wavelength (e.g., laser)
Polychromatic: multiple wavelengths (e.g., white light)
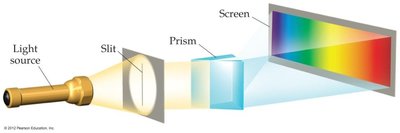
Some sources, like excited gases, produce line spectra, not continuous spectra.

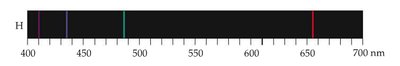
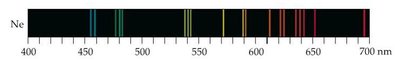
The Bohr Model explains the line spectra of hydrogen by proposing that electrons occupy only certain allowed orbits with specific energies. Transitions between these orbits result in the emission or absorption of photons.
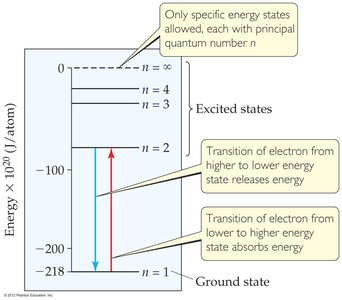
Energy is emitted or absorbed only when an electron changes from one allowed energy state to another:
or
J
\(n\): principal quantum number (1, 2, 3, ...)
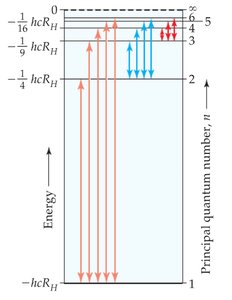
The energy difference for a transition is:
J
If \(\Delta E\) is negative, energy is emitted; if positive, energy is absorbed.
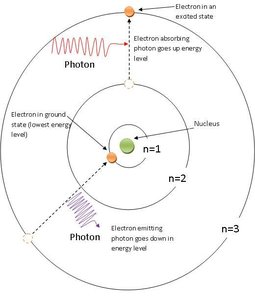
The Rydberg Equation allows calculation of the wavelength of emitted or absorbed light for hydrogen:
\(R_H = 1.0968 \times 10^7\) m-1
\(n_2 > n_1\), both integers
6.4 The Wave Behavior of Matter
Louis de Broglie proposed that particles, such as electrons, have wave-like properties. The wavelength of a particle is given by:
\(m\): mass (kg)
\(v\): velocity (m/s)
This concept is significant for subatomic particles, but negligible for macroscopic objects.
6.5 Quantum Mechanics and Atomic Orbitals
Quantum mechanics, developed by Schrödinger, describes electrons as wave functions (\(\psi\)). The square of the wave function (\(\psi^2\)) gives the probability density of finding an electron in a particular region.
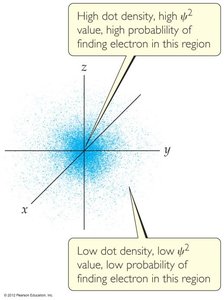
Solutions to Schrödinger's equation are called orbitals, each described by a set of quantum numbers:
Principal quantum number (n): Indicates size and energy level (n = 1, 2, 3, ...).
Angular momentum quantum number (l): Indicates shape (l = 0 to n-1; s, p, d, f).
Magnetic quantum number (ml): Indicates orientation (-l to +l).
Spin quantum number (ms): Indicates electron spin (+1/2 or -1/2).
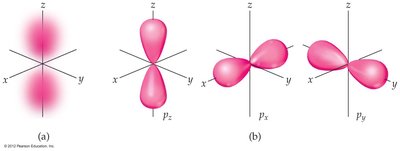
6.6 Representations of Orbitals
The radial probability function describes the probability of finding an electron at a certain distance from the nucleus. The s, p, and d orbitals have distinct shapes and orientations.
s orbitals: Spherical shape
p orbitals: Dumbbell shape, oriented along x, y, or z axes
d orbitals: More complex shapes
6.7 Many-Electron Atoms and the Pauli Exclusion Principle
The Pauli exclusion principle states that no two electrons in an atom can have the same set of four quantum numbers. Each orbital can hold a maximum of two electrons with opposite spins.
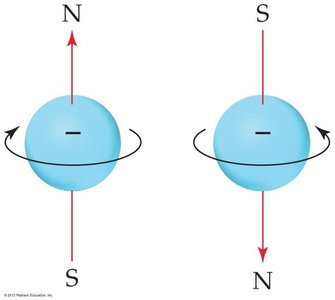
Quantum numbers are used to describe the allowed states of electrons in atoms.
6.8 Electron Configurations
The electron configuration of an atom describes the distribution of electrons among orbitals. The Aufbau principle states that electrons fill orbitals in order of increasing energy. The Pauli exclusion principle and Hund's rule also govern electron arrangements:
Aufbau principle: Fill lowest energy orbitals first.
Pauli exclusion principle: Maximum two electrons per orbital, with opposite spins.
Hund's rule: Electrons occupy degenerate orbitals singly before pairing up.
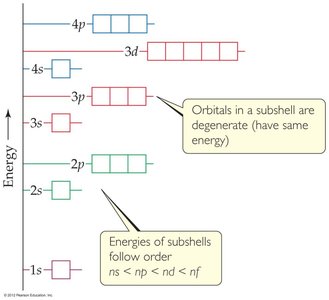
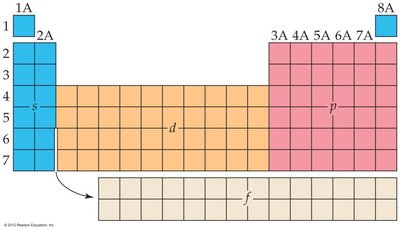
Electron configurations can be written in full or condensed (noble-gas) notation.

Core electrons: Inner electrons, not involved in bonding.
Valence electrons: Outermost electrons, involved in bonding and chemical reactions.
Practice and Applications
Write electron configurations for elements and ions.
Identify core and valence electrons.
Recognize isoelectronic species (atoms/ions with the same electron configuration).
Summary Table: Quantum Numbers and Orbitals
n | Possible Values of l | Subshell Designation | Possible Values of ml | Number of Orbitals in Subshell | Total Number of Orbitals in Shell |
|---|---|---|---|---|---|
1 | 0 | 1s | 0 | 1 | 1 |
2 | 0, 1 | 2s, 2p | 0; 1, 0, -1 | 1; 3 | 4 |
3 | 0, 1, 2 | 3s, 3p, 3d | 0; 1, 0, -1; 2, 1, 0, -1, -2 | 1; 3; 5 | 9 |
4 | 0, 1, 2, 3 | 4s, 4p, 4d, 4f | 0; 1, 0, -1; 2, 1, 0, -1, -2; 3, 2, 1, 0, -1, -2, -3 | 1; 3; 5; 7 | 16 |
