 Back
BackChapter 6: Gases – Properties, Laws, and Real Gas Behavior
Study Guide - Smart Notes

Gases: Characteristics and Physical Properties
General Properties of Gases
Gases are distinguished from other states of matter by their high degree of disorder and ability to fill containers completely. They possess a large amount of free space, can expand infinitely, and mix rapidly and randomly. The physical properties of gases are fundamentally linked to their molecular motion and collisions.
Free space: Gas molecules are far apart compared to solids and liquids.
Expansion: Gases expand to fill any container.
Uniformity: Gases occupy containers uniformly and completely.
Mixing: Gases mix rapidly and randomly.
Disorder: Gases are the most disordered phase of matter.
Four Basic Properties of Gases
The behavior of gases is described by four interrelated properties: pressure, volume, temperature, and amount (moles). Changes in one property affect the others.
Pressure (P): Measured in atmospheres (atm), pascals (Pa), torr, or psi.
Volume (V): Measured in liters (L).
Temperature (T): Measured in kelvins (K).
Amount (n): Measured in moles.
Gas Pressure
Definition and Origin of Pressure
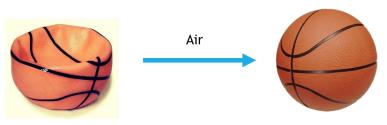
Gas pressure is the force exerted per unit area by gas molecules as they strike the surfaces around them. It results from the constant movement and collisions of gas molecules.
Pressure formula:
Concentration effect: Higher concentration of gas molecules increases pressure.
Volume effect: Increasing volume decreases concentration and pressure.

Common Pressure Units
Unit | Abbreviation | Average Air Pressure at Sea Level |
|---|---|---|
Pascal | Pa | 101,325 Pa |
Pounds per square inch | psi | 14.7 psi |
Torr (1 mmHg) | torr | 760 torr |
Inches of mercury | in Hg | 29.92 in Hg |
Atmosphere | atm | 1 atm |
Gas Laws
Boyle’s Law: Pressure and Volume
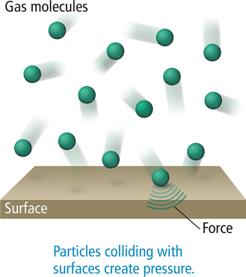
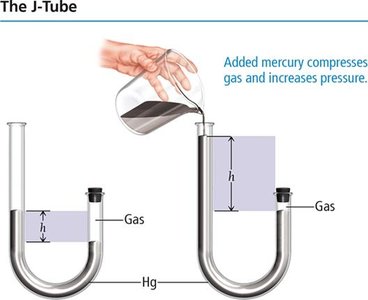
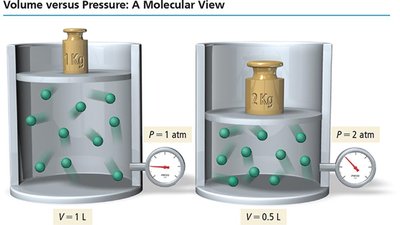
Boyle’s Law describes the inverse relationship between pressure and volume for a fixed amount of gas at constant temperature. As volume decreases, pressure increases due to more frequent collisions.
Mathematical expression:
Inverse relationship: (at constant n and T)
Charles’s Law: Volume and Temperature
Charles’s Law states that the volume of a gas is directly proportional to its temperature (in kelvins) at constant pressure and amount. As temperature increases, kinetic energy and volume increase.
Mathematical expression:
Direct relationship: (at constant n and P)
Kelvin conversion:
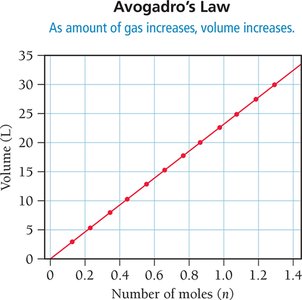
Avogadro’s Law: Volume and Amount (Moles)
Avogadro’s Law states that the volume of a gas is directly proportional to the number of moles at constant pressure and temperature. More gas particles require more space.
Mathematical expression:
Direct relationship: (at constant P and T)
Equal volumes: Equal volumes of gases contain equal numbers of molecules at constant P and T.
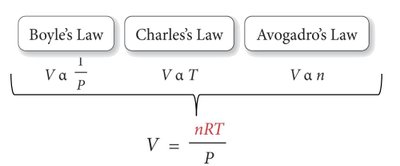
The Ideal Gas Law
The Ideal Gas Law combines Boyle’s, Charles’s, and Avogadro’s laws into a single equation for ideal gases. It relates pressure, volume, temperature, and amount of gas.
Equation:
Gas constant:
Applications: Used to calculate any property if the others are known.
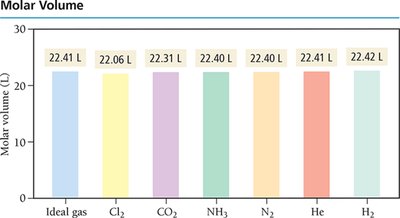
Standard Conditions (STP) and Molar Volume
Standard Temperature and Pressure (STP) are defined as 0°C (273 K) and 1.00 atm. At STP, one mole of an ideal gas occupies 22.4 L, known as the molar volume.
Molar volume at STP: 22.4 L for 1 mol of any ideal gas.
Identity of gas: Molar volume is independent of gas identity at STP.

Density and Molar Mass of Gases
Density of a Gas at STP
Density is the ratio of mass to volume. At STP, the density of a gas depends on its molar mass and the molar volume (22.4 L).
Density formula:
Greater molar mass: Higher density.
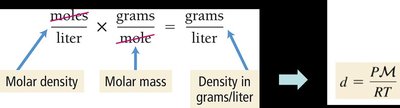
Calculating Density at Any Condition
The Ideal Gas Law can be rearranged to calculate density at any condition:
Formula:
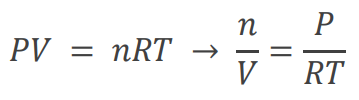
Molar Mass of a Gas
The molar mass of an unknown gas can be determined by measuring its mass, volume, pressure, and temperature, then using the Ideal Gas Law to find the number of moles.
Formula:
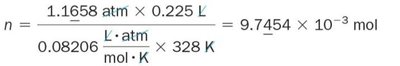
Mixtures of Gases and Partial Pressures
Partial Pressure and Dalton’s Law
In a mixture, each gas exerts a partial pressure independently. The total pressure is the sum of the partial pressures of all gases present.
Dalton’s Law:
Partial pressure: Calculated using the Ideal Gas Law for each component.
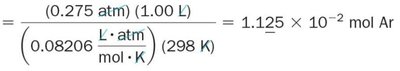
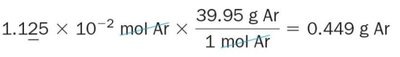
Mole Fraction
The mole fraction is the ratio of the number of moles of a component to the total number of moles in the mixture. It is used to calculate partial pressures.
Formula:
Partial pressure:
Reactions Involving Gases
Stoichiometry with Gases
Coefficients in chemical equations can be used as conversion factors. At STP, 1 mole of gas = 22.4 L. The Ideal Gas Law is used to relate moles and volume for reactions involving gases.
Conversion steps: mass A → moles A → moles B → mass B

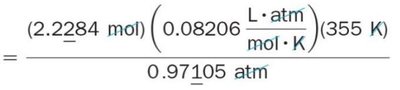
Kinetic Molecular Theory
Basic Assumptions
The kinetic molecular theory models gases as particles in constant motion. It explains gas properties and behavior based on three main assumptions:
Negligible size: Gas particles occupy no volume compared to the space between them.
Kinetic energy proportional to temperature: Higher temperature means faster motion.
Elastic collisions: Collisions exchange energy without loss.

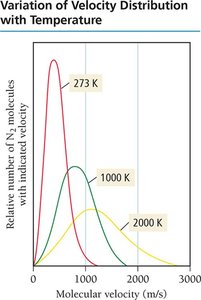
Temperature and Molecular Velocities
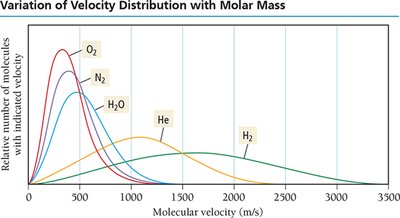
At a given temperature, particles have the same average kinetic energy but different velocities due to varying masses. Lighter particles travel faster; speed increases with temperature.
Root mean square velocity:
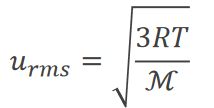
Mean Free Path, Diffusion, and Effusion
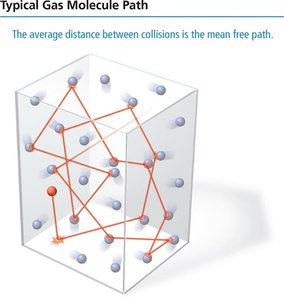
Mean Free Path
The mean free path is the average distance a molecule travels between collisions. It decreases as pressure increases.
Diffusion and Effusion
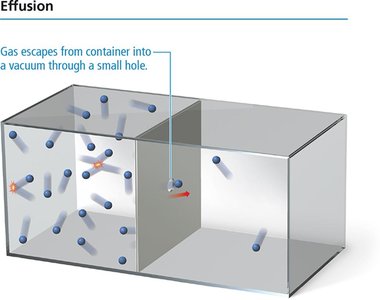
Diffusion is the spreading of molecules from high to low concentration. Effusion is the escape of molecules through a small hole into a vacuum. Both rates are related to molecular velocity; heavier molecules diffuse and effuse slower.
Graham’s Law of Effusion
Graham’s Law relates the rates of effusion of two gases to their molar masses:
Formula:
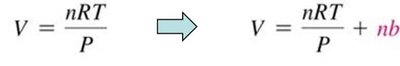
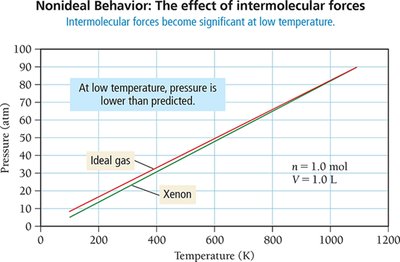
Real Gases and Deviations from Ideal Behavior
Nonideal Behavior at High Pressure and Low Temperature
Real gases deviate from ideal behavior at high pressure and low temperature due to finite particle volume and intermolecular forces. The ideal gas law assumes negligible particle volume and no intermolecular forces, which is valid at STP for most gases.
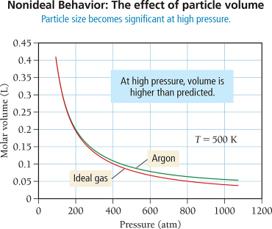
Van der Waals Equation
Johannes van der Waals modified the ideal gas equation to account for real gas behavior. The equation includes corrections for molecular volume (b) and intermolecular attractions (a):
Correction for volume:
Correction for pressure:
Combined equation:


Additional info: The notes above expand on the original content by providing definitions, formulas, and academic context for each law and concept. All images included are directly relevant to the adjacent explanations, visually reinforcing key points about gas properties, laws, and real gas behavior.
