 Back
BackChapter 7: Periodic Properties of the Elements - Comprehensive Study Notes
Study Guide - Smart Notes

Periodic Properties of the Elements
Development of the Periodic Table
The periodic table is a fundamental tool in chemistry, organizing elements according to their atomic structure and properties. Its development was driven by the need to classify elements in a meaningful way, reflecting recurring trends in their chemical and physical behavior.
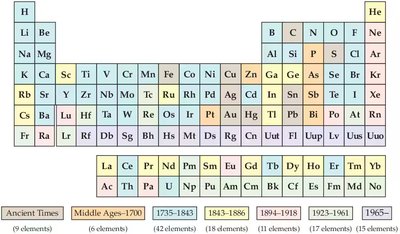
Early Attempts: Mendeleev and Meyer arranged elements by increasing atomic weight, but some elements did not fit the pattern.
Mendeleev's Predictions: Mendeleev predicted the existence and properties of undiscovered elements, such as germanium (eka-silicon).
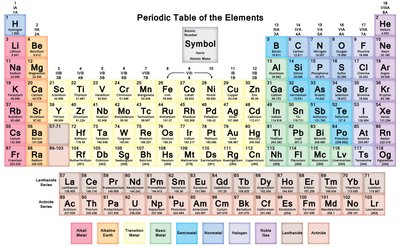
Modern Table: Elements are now arranged by increasing atomic number, which resolves earlier inconsistencies.

Property | Mendeleev's Predictions for Eka-Silicon (1871) | Observed Properties of Germanium (1886) |
|---|---|---|
Atomic weight | 72 | 72.59 |
Density (g/cm3) | 5.5 | 5.35 |
Specific heat (J/g·K) | 0.305 | 0.309 |
Melting point (°C) | High | 947 |
Color | Dark gray | Grayish white |
Formula of oxide | XO2 | GeO2 |
Density of oxide (g/cm3) | 4.7 | 4.70 |
Formula of chloride | XCl4 | GeCl4 |
Boiling point of chloride (°C) | A little under 100 | 84 |
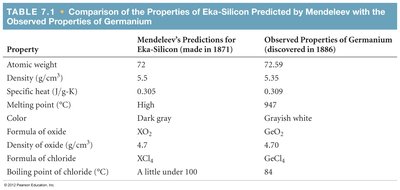
Example: Mendeleev's prediction of germanium's properties was remarkably accurate, demonstrating the power of periodic trends.
Effective Nuclear Charge
In multi-electron atoms, electrons experience both attraction to the nucleus and repulsion from other electrons. The net positive charge felt by an electron is called the effective nuclear charge (Zeff).
Definition: where Z is the atomic number and S is the screening constant (number of inner electrons).
Shielding Effect: Inner electrons shield outer electrons from the full nuclear charge, reducing Zeff.
Factors Affecting Zeff: Distance from nucleus, electron-electron repulsion, and orbital type.
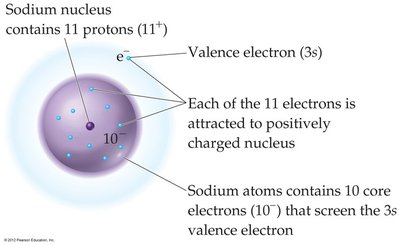
Example: In sodium, the 3s valence electron is shielded by 10 core electrons, so Zeff is less than the full nuclear charge.
Sizes of Atoms and Ions
Atomic Radii
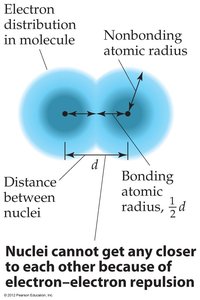
The size of an atom is typically measured by its bonding atomic radius, defined as half the distance between covalently bonded nuclei.
Trends: Atomic radius decreases across a period (left to right) due to increasing Zeff, and increases down a group due to higher principal quantum number (n).
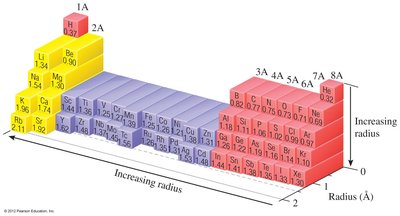
Ionic Radii
Ionic size depends on nuclear charge, number of electrons, and electron orbitals.
Cations: Smaller than parent atoms due to loss of electrons and reduced repulsion.
Anions: Larger than parent atoms due to gain of electrons and increased repulsion.
Trend: Ionic size increases down a group (higher n).
Isoelectronic Series: Ions with the same number of electrons; size decreases with increasing nuclear charge.
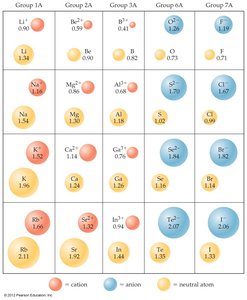
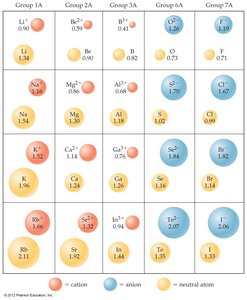
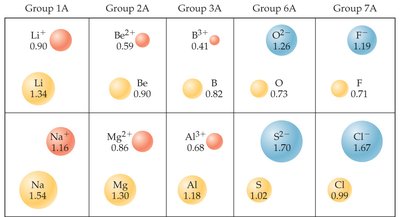
Example: In an isoelectronic series, O2− is larger than F− because it has fewer protons attracting the same number of electrons.
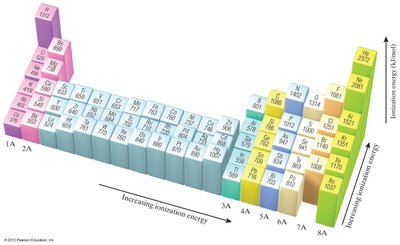
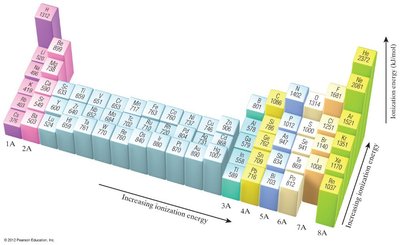
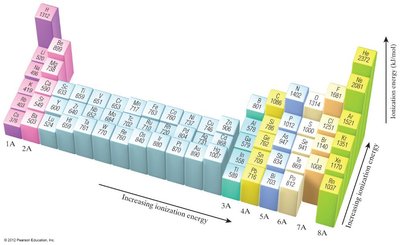
Ionization Energy
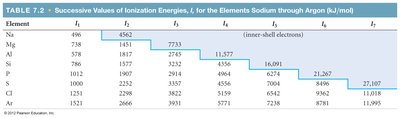
Ionization energy (IE) is the energy required to remove an electron from a gaseous atom or ion. Successive ionization energies increase as more electrons are removed.
First Ionization Energy: Energy to remove the first electron.
Successive Ionization Energies: Each additional electron requires more energy, especially after all valence electrons are removed.
Trends: IE decreases down a group and increases across a period due to increasing Zeff.
Exceptions: Discontinuities occur when removing the first p electron (Group IIIA) or a doubly occupied p orbital (Group VIA).

Element | I1 | I2 | I3 | I4 | I5 | I6 | I7 |
|---|---|---|---|---|---|---|---|
Na | 496 | 4562 | 6912 | 9544 | 13352 | 16613 | 21267 |
Mg | 738 | 1451 | 7733 | 10542 | 13630 | 18020 | 21700 |
Al | 578 | 1817 | 2745 | 11577 | 14842 | 18379 | 23326 |
Si | 786 | 1577 | 3232 | 4366 | 6274 | 16101 | 21067 |
P | 1012 | 1907 | 2914 | 4952 | 6276 | 8134 | 9800 |
S | 1002 | 2252 | 3400 | 4556 | 7004 | 8495 | 10459 |
Cl | 1251 | 2666 | 3860 | 5158 | 6548 | 9362 | 11018 |
Ar | 1521 | 2666 | 3931 | 5771 | 7238 | 8781 | 11995 |
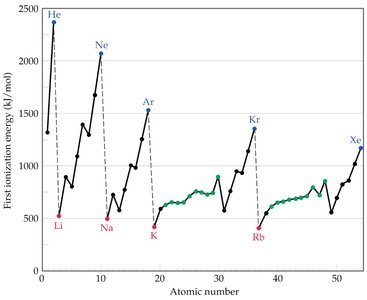
Example: The sharp increase in ionization energy after removal of all valence electrons indicates the start of inner-shell electron removal.
Electron Affinity
Electron affinity (EA) is the energy change when an electron is added to a gaseous atom. Negative values indicate exothermic processes (energy released).
Trends: EA becomes more exothermic across a period, but there are discontinuities between Groups IA/IIA and IVA/VA.
Exceptions: Noble gases have positive EA, meaning their anions are unstable.
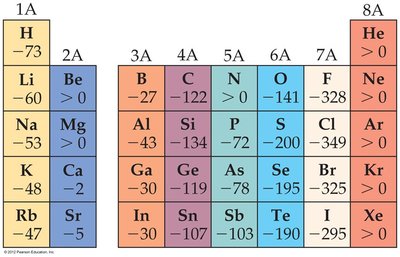
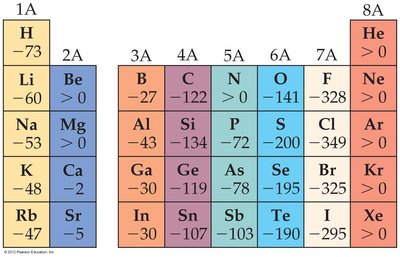
Example: Chlorine has a highly negative EA, making it a strong oxidizer, while noble gases do not form stable anions.
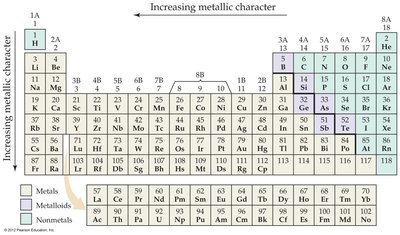
Metals, Nonmetals, and Metalloids
Elements are classified as metals, nonmetals, or metalloids based on their physical and chemical properties.
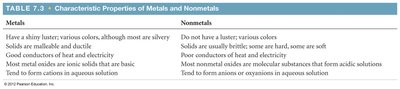
Metals: Shiny, malleable, good conductors, tend to form cations, and basic oxides.
Nonmetals: Diverse, poor conductors, tend to form anions, and acidic oxides.
Metalloids: Intermediate properties; e.g., silicon is shiny but brittle.
Metals | Nonmetals |
|---|---|
Shiny luster, various colors | No luster, various colors |
Solids are malleable and ductile | Solids are usually brittle |
Good conductors of heat/electricity | Poor conductors |
Basic oxides | Acidic oxides |
Form cations in solution | Form anions in solution |

Example: Brass and steel are metals, while sulfur and antimony are nonmetals.
Trends for Group 1A and Group 2A Metals
Group 1A: Alkali Metals
Alkali metals are soft, highly reactive solids dominated by the loss of their single s electron. Their reactivity increases down the group.
Reactions: React with water to form hydroxides and hydrogen gas; reactions are exothermic.
Physical Properties: Low density, low melting point, low ionization energy.
Oxides: Form peroxides and superoxides (except Li).
Flame Colors: Produce bright colors in flame tests.
Example: Sodium reacts vigorously with water, producing NaOH and H2 gas.
Group 2A: Alkaline Earth Metals
Alkaline earth metals are harder and denser than alkali metals, with higher melting points and ionization energies. Their reactivity increases down the group.
Reactivity: Beryllium does not react with water; magnesium reacts only with steam; others react readily with water.
Example: Calcium reacts with water to form Ca(OH)2 and H2 gas.
Trends for Selected Nonmetals
Hydrogen
Hydrogen is a colorless diatomic gas, capable of forming both H− and H+ ions. Its aqueous chemistry is dominated by the proton H+(aq).
Group 6A: The Oxygen Group
Metallic character increases down the group. Oxygen exists as O2 and ozone (O3), both potent oxidizers. Sulfur forms S8 rings and is a weaker oxidizer than oxygen.
Example: Ozone is prepared from oxygen and is toxic and unstable.
Group 7A: The Halogens
Halogens are prototypical nonmetals, highly reactive, and form salts by gaining electrons. Fluorine is the most reactive, and chlorine is industrially important.
Electron Affinity: Large, negative values; strong oxidizers.
Reactions: React with metals to form halides; chlorine disinfects water.
Example: Chlorine reacts with water to produce hypochlorous acid, a disinfectant.
Group 8A: The Noble Gases
Noble gases have very high ionization energies and positive electron affinities, making them chemically inert. They exist as monatomic gases and rarely form compounds.
Example: Xenon forms compounds such as XeF2, XeF4, and XeF6 due to its relatively low ionization energy.
Chapter Checklist
Describe the development of the Periodic Table.
Explain electron shells and atomic size.
Define ionization energy and electron affinity; describe their trends.
Classify metals, nonmetals, and metalloids; explain their distribution.
Describe group trends for active metals (Groups 1A, 2A) and selected nonmetals (Groups 6A, 7A, 8A, hydrogen).
