 Back
BackChapter 7: Thermochemistry – Energy, Heat, and Chemical Reactions
Study Guide - Smart Notes

Thermochemistry: The Study of Energy in Chemical Processes
Introduction to Thermochemistry
Thermochemistry explores the relationship between energy and matter, focusing on how energy is transferred during chemical reactions and physical changes. Understanding these concepts is essential for predicting reaction behavior and energy requirements in chemical systems.
Nature and Types of Energy
Definitions and Forms of Energy
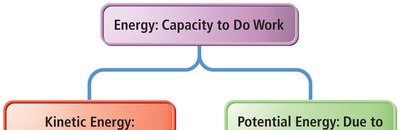
Energy is the capacity to do work or produce heat.
Work is a force acting over a distance:
Heat is the flow of energy due to a temperature difference between objects.
Classification of Energy
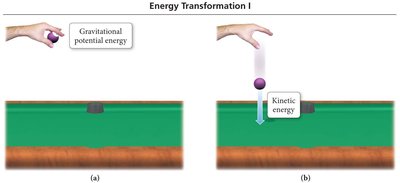
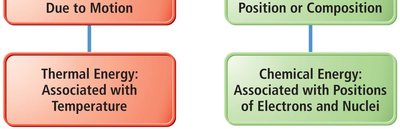
Kinetic Energy: Energy of motion or energy being transferred. Thermal energy is a form of kinetic energy associated with temperature.
Potential Energy: Stored energy due to position or composition. Chemical potential energy is stored in the structure of compounds.
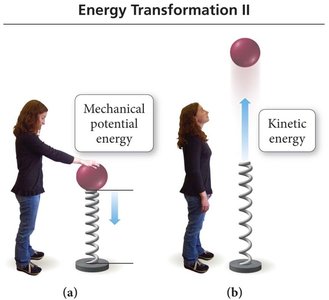
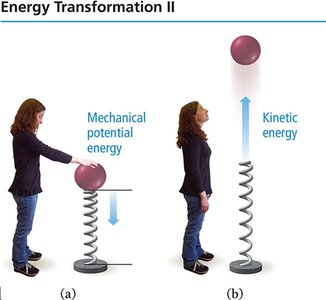
Examples of Energy Forms
Electrical energy: Kinetic energy from the flow of charge.
Thermal energy: Kinetic energy from molecular motion.
Light (radiant) energy: Kinetic energy from atomic transitions.
Nuclear energy: Potential energy in atomic nuclei.
Chemical energy: Potential energy from atomic and molecular structure.
Conservation of Energy and Energy Transfer
Law of Conservation of Energy
Energy cannot be created or destroyed; it can only be transferred or converted.
Total energy before and after a process remains constant.
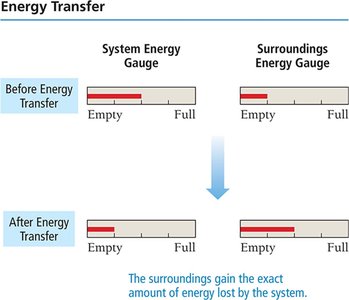
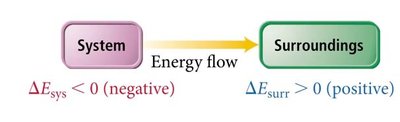
System and Surroundings
System: The part of the universe under study (e.g., chemicals in a reaction).
Surroundings: Everything else that can exchange energy with the system.
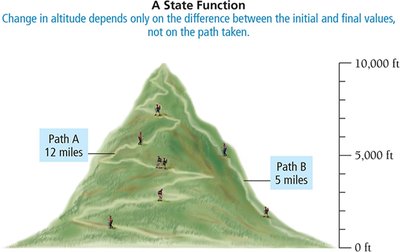
Energy Transfer and State Functions
Energy gained or lost by the system equals the energy lost or gained by the surroundings.
State function: A property dependent only on the initial and final states, not the path taken (e.g., internal energy, altitude).
Units of Energy
Joules and Calories
Joule (J):
Calorie (cal): Energy to raise 1 g of water by 1°C.
Calorie (Cal or kcal):
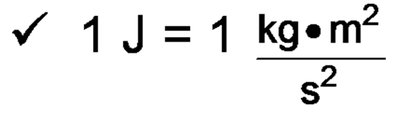

Internal Energy and Its Changes
Internal Energy (E)
Sum of kinetic and potential energies of all particles in a system.
Change in internal energy:
For reactions:
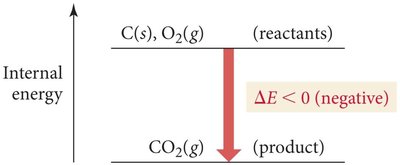
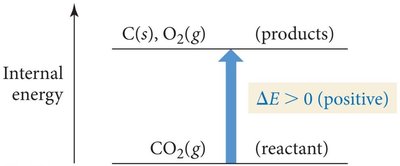
Energy Flow in Chemical Reactions
If energy flows out of the system: (negative), (positive).
If energy flows into the system: (positive), (negative).

Heat, Work, and Internal Energy
Heat (q) and Work (w)
Energy is exchanged as heat (q) or work (w):
q and w are not state functions; their values depend on the process.

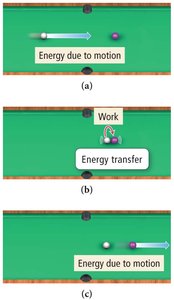
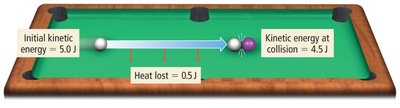
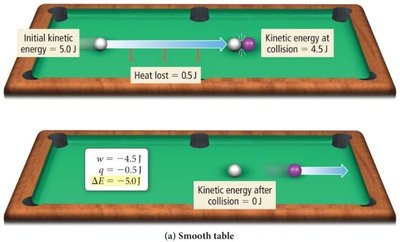
Examples: Energy Transfer in Collisions
On a smooth table, most kinetic energy is transferred as work; little is lost as heat.
On a rough table, more energy is lost as heat due to friction.
Heat Capacity and Specific Heat
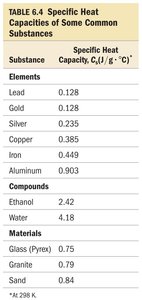
Heat Capacity (C) and Specific Heat Capacity (Cs)
Heat capacity (C): Amount of heat required to raise an object's temperature by 1°C (units: J/°C).
Specific heat capacity (Cs): Heat required to raise 1 g of a substance by 1°C (units: J/g·°C).
Molar heat capacity: Heat required to raise 1 mol of a substance by 1°C.
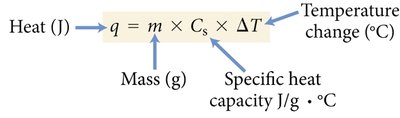
Quantifying Heat Energy
Heat absorbed or released:
Where m is mass (g), is specific heat capacity, and is temperature change (°C).
Thermal Energy Transfer and Calorimetry
Thermal Equilibrium and Conservation of Energy
When two objects at different temperatures contact, heat flows from hot to cold until equilibrium is reached.
Law of conservation of energy:
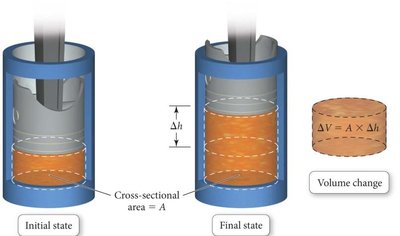
Pressure-Volume Work
Work Done by Expanding Gases
When a gas expands against constant external pressure:
1 L·atm = 101.3 J (for unit conversion)
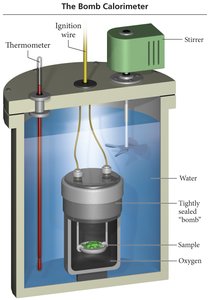
Measuring Energy Changes: Calorimetry
Constant Volume Calorimetry (Bomb Calorimeter)
Measures at constant volume (w = 0):
Heat absorbed by calorimeter:
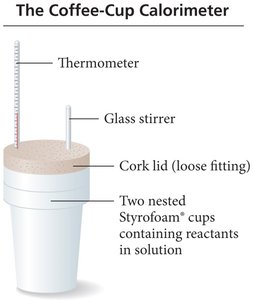
Constant Pressure Calorimetry (Coffee-Cup Calorimeter)
Measures at constant pressure (open to atmosphere).
Heat exchanged:
Enthalpy and Chemical Reactions
Enthalpy (H) and Enthalpy Change ()
Enthalpy (H):
Enthalpy change (): Heat evolved at constant pressure.
Exothermic: (heat released); Endothermic: (heat absorbed).
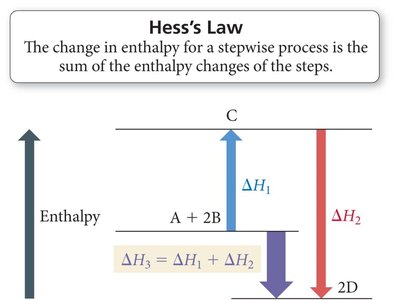
Hess's Law
If a reaction is the sum of several steps, is the sum of the values for each step.
Standard Enthalpy of Formation ()
Enthalpy change for forming 1 mol of a compound from its elements in their standard states.
for elements in their standard state is zero.
Calculating Standard Enthalpy Change for a Reaction
Summary Table: Key Equations and Conventions
Quantity | Positive (+) | Negative (−) |
|---|---|---|
q (heat) | System gains thermal energy | System loses thermal energy |
w (work) | Work done on the system | Work done by the system |
(internal energy) | Energy flows into the system | Energy flows out of the system |
Additional info: This summary covers the core concepts of thermochemistry, including energy types, conservation, heat and work, calorimetry, enthalpy, and Hess's Law, with relevant equations and diagrams for clarity.
