 Back
BackChapter 9: Periodic Properties of the Elements – Study Notes
Study Guide - Smart Notes

Periodic Properties of the Elements
Introduction to the Periodic Table
The periodic table organizes elements based on recurring chemical and physical properties. This arrangement allows chemists to predict element behavior and understand trends in atomic structure and reactivity.
Mendeleev's Periodic Law: When elements are arranged by increasing atomic mass, certain properties recur periodically.
Modern Periodic Law: Properties of elements are a periodic function of their atomic numbers.
Elements with similar properties are grouped in columns (families or groups).
Mendeleev used periodic trends to predict properties of undiscovered elements.
Quantum-Mechanical Model and Electron Configuration
The quantum-mechanical model explains the arrangement of electrons in atoms and the origin of periodic trends. Electrons occupy orbitals defined by quantum numbers, which determine their energy, shape, and orientation.
Principal Quantum Number (n): Indicates the main energy level (shell) of an electron; n = 1, 2, 3, ...
Angular Momentum Quantum Number (l): Defines the shape of the orbital; l = 0 (s), 1 (p), 2 (d), 3 (f).
Magnetic Quantum Number (ml): Specifies the orientation of the orbital; values from –l to +l.
Spin Quantum Number (ms): Describes electron spin; values are +1/2 or –1/2.
Each electron in an atom has a unique set of these four quantum numbers.
Electron Spin and the Pauli Exclusion Principle
Electron spin is a fundamental property, and the Pauli Exclusion Principle states that no two electrons in an atom can have the same set of four quantum numbers. Thus, each orbital can hold a maximum of two electrons with opposite spins.
Orbital Diagrams: Visual representations using boxes (orbitals) and arrows (electrons with spin direction).
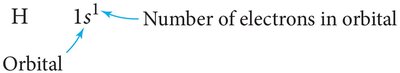
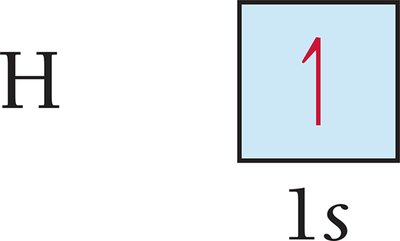

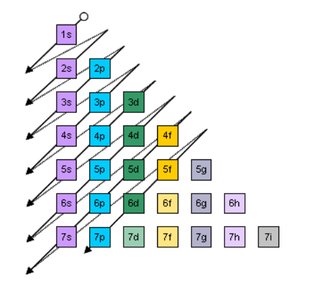
Filling Order of Orbitals and the Aufbau Principle
Electrons fill orbitals in order of increasing energy, following the Aufbau principle. The general order is:
1s → 2s → 2p → 3s → 3p → 4s → 3d → 4p → 5s → 4d → 5p → 6s → 4f → 5d → 6p → 7s → 5f → 6d → 7p
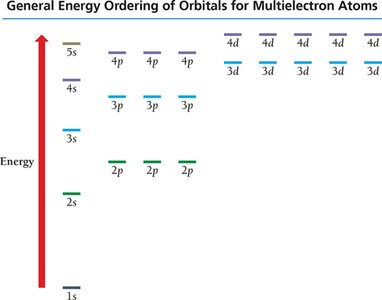
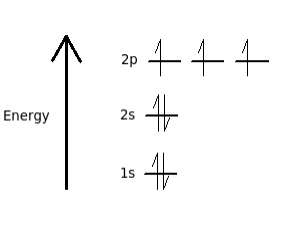
Hund's Rule: When filling orbitals of equal energy, electrons fill singly first, with parallel spins, before pairing up.
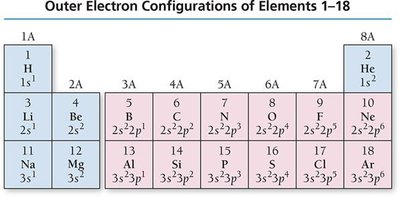
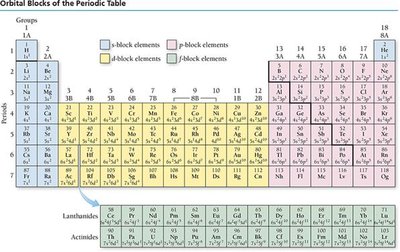
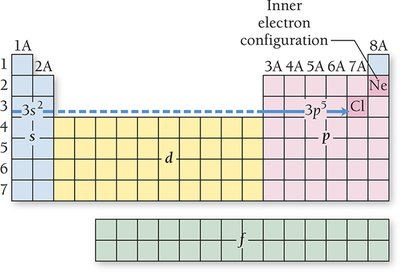
Electron Configurations and the Periodic Table
Electron configurations describe the arrangement of electrons in an atom. The periodic table reflects these configurations, with blocks corresponding to s, p, d, and f sublevels.
Valence Electrons: Electrons in the outermost shell (highest n); determine chemical properties and reactivity.
Core Electrons: Electrons in inner shells; do not participate in bonding.
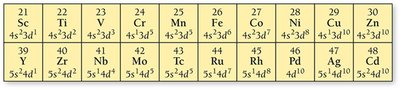
Irregular Electron Configurations
Some transition metals have irregular electron configurations due to stability associated with half-filled or fully filled d subshells. These must be determined experimentally.
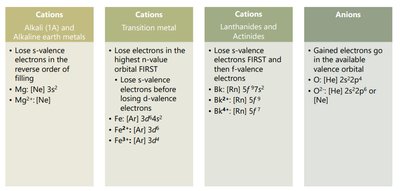
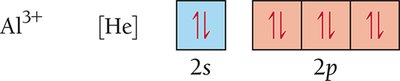
Electron Configurations of Ions
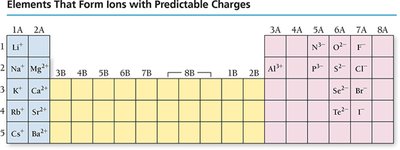
When atoms form ions, electrons are added or removed from the valence shell. Cations lose electrons, while anions gain electrons. The resulting electron configuration often matches that of a noble gas.
Periodic Trends
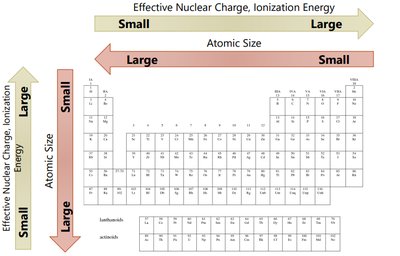
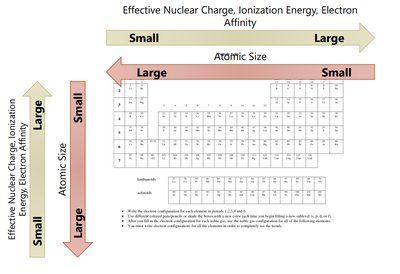
Atomic Radius
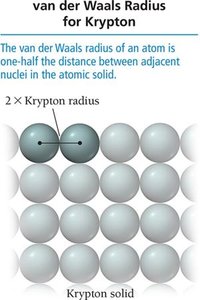
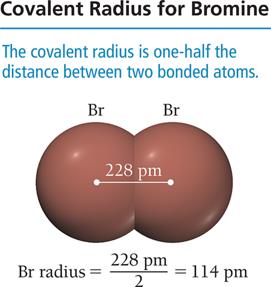
The atomic radius is a measure of the size of an atom. It can be defined in several ways, including van der Waals radius (nonbonding), covalent radius (bonding), and metallic radius.
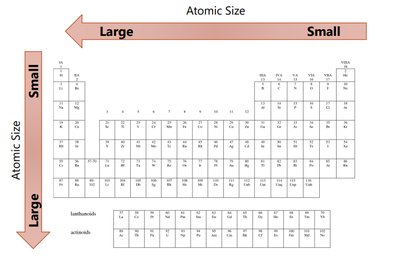
Trend Down a Group: Atomic radius increases due to addition of energy levels.
Trend Across a Period: Atomic radius decreases due to increasing effective nuclear charge pulling electrons closer.

Effective Nuclear Charge (Zeff)
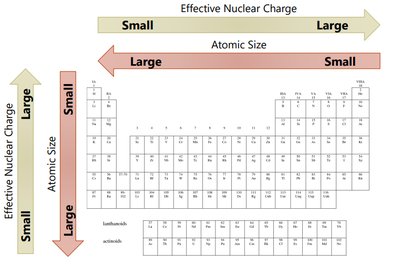
The effective nuclear charge is the net positive charge experienced by valence electrons. It is calculated as:
where Z is the atomic number and S is the number of core electrons. Zeff increases across a period and slightly increases down a group.
Ionic Radius
Cations are smaller than their parent atoms due to loss of electrons and increased attraction between remaining electrons and the nucleus. Anions are larger due to added electron-electron repulsion and decreased effective nuclear charge per electron.

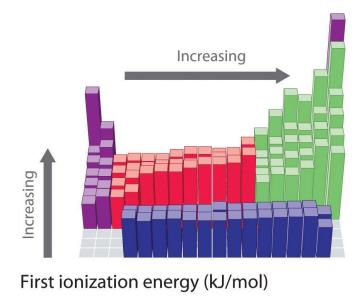
Ionization Energy (IE)
Ionization energy is the energy required to remove an electron from a gaseous atom or ion. It is always positive (endothermic).
Trend Down a Group: IE decreases as electrons are farther from the nucleus.
Trend Across a Period: IE increases due to higher effective nuclear charge.
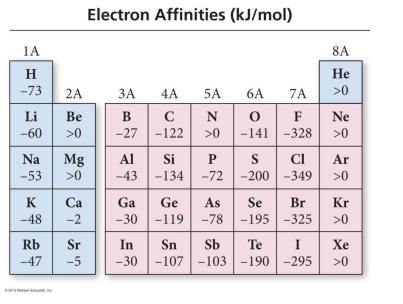
Electron Affinity (EA)
Electron affinity is the energy change when an atom gains an electron. A more negative value indicates a greater tendency to accept an electron.
Trend Down a Group: EA becomes less negative (less exothermic).
Trend Across a Period: EA becomes more negative (more exothermic) from left to right, with exceptions.
Metallic Character
Metallic character refers to how easily an element loses electrons (is oxidized). It increases down a group and decreases across a period.
Metals: Malleable, ductile, good conductors, form cations.
Nonmetals: Brittle, poor conductors, form anions.
Summary Table of Periodic Trends
Property | Trend Down a Column | Reason Down a Column | Trend Across a Row | Reason Across a Row |
|---|---|---|---|---|
Atomic Radius | Increasing | Size of outermost occupied orbital increases | Decreasing | Effective nuclear charge increases |
First Ionization Energy | Decreasing | Outermost electrons further from nucleus | Increasing | Effective nuclear charge increases |
Electron Affinity | No definite trend | Decreasing (more negative) | Effective nuclear charge increases | |
Metallic Character | Increasing | Ionization energy decreases | Decreasing | Ionization energy increases |
