 Back
BackChemical Equilibrium: Concepts, Calculations, and Applications
Study Guide - Smart Notes

Chemical Equilibrium
The Concept of Equilibrium
Chemical equilibrium is the state in which the concentrations of all reactants and products in a closed system remain constant over time, as the forward and reverse reactions occur at equal rates. This dynamic balance means that, although reactions continue to occur, there is no net change in the amounts of substances present.
Dynamic equilibrium: Both forward and reverse reactions continue, but their rates are equal.
Constant concentrations: The amounts of reactants and products do not change once equilibrium is reached.
Double arrow notation: Equilibrium reactions are represented with a double arrow () to indicate reversibility.

Approaching Equilibrium
As a reaction mixture approaches equilibrium, the concentrations of reactants and products change until the rates of the forward and reverse reactions become equal. The path to equilibrium can be monitored experimentally by measuring concentration changes over time.
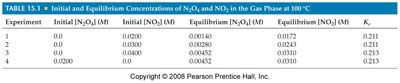
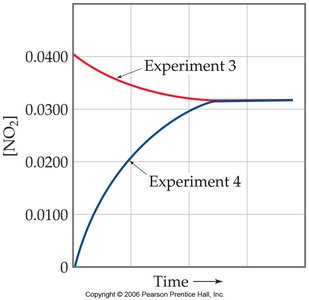
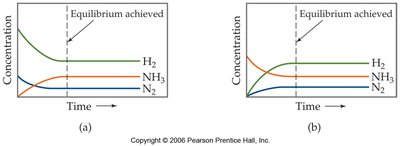
Equilibrium from Either Direction
Equilibrium can be established whether the reaction starts with only reactants, only products, or a mixture of both. Regardless of the starting point, the system will reach the same equilibrium concentrations under identical conditions.
The Equilibrium Constant
Defining the Equilibrium Constant
The equilibrium constant () quantifies the ratio of product concentrations to reactant concentrations at equilibrium, each raised to the power of their coefficients in the balanced equation. For a general reaction:
The equilibrium constant expression is:
: Uses concentrations in molarity (M).
: Used for gases, based on partial pressures.
For reactions involving gases, is related to by:
where is the change in moles of gas (), is the gas constant, and is temperature in Kelvin.
Interpreting the Value of K
If : The reaction is product-favored; products predominate at equilibrium.
If : The reaction is reactant-favored; reactants predominate at equilibrium.
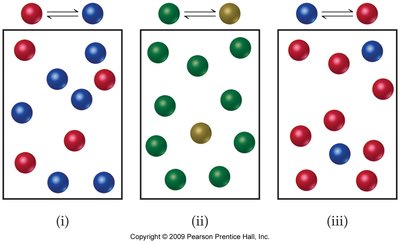
Visualizing Equilibrium Systems
Different systems at equilibrium can be compared by examining the relative numbers of reactant and product particles.
Manipulating Equilibrium Constants
Rules for Manipulating K
Reversing a reaction: The new is the reciprocal of the original.
Multiplying a reaction by a factor : The new is the original raised to the th power.
Adding reactions: The overall is the product of the individual values.
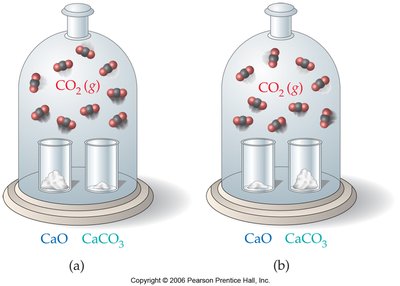
Heterogeneous Equilibria
Solids and Liquids in Equilibrium Expressions
The concentrations of pure solids and pure liquids are constant and do not appear in equilibrium constant expressions. Only the concentrations of gases and aqueous species are included.
Example: For ,

Calculating Equilibrium Concentrations
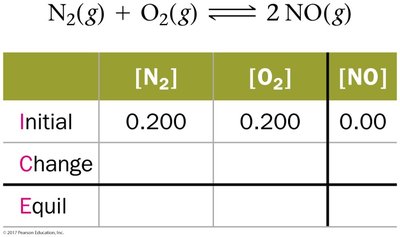
ICE Tables
ICE tables (Initial, Change, Equilibrium) are used to organize data and solve for unknown concentrations at equilibrium.
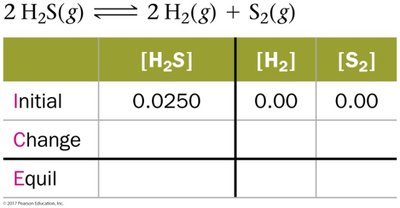
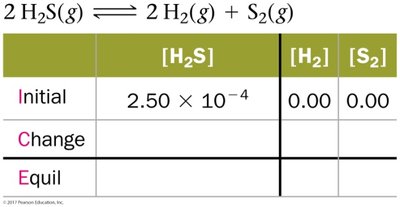
![Calculation of [H2S] from moles and volume](https://static.studychannel.pearsonprd.tech/study_guide_files/general-chemistry/sub_images/8dff37e2_image_12.png)
Quadratic Equation in Equilibrium Calculations
When solving for equilibrium concentrations, you may need to solve a quadratic equation:
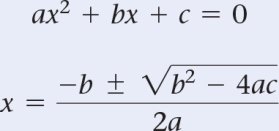
The Reaction Quotient (Q) and Predicting Direction
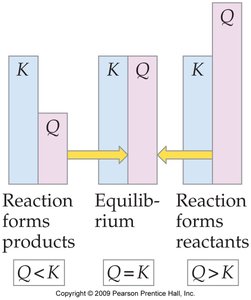
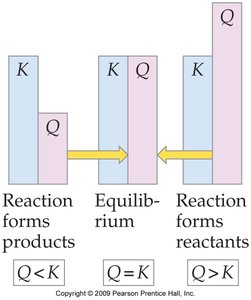
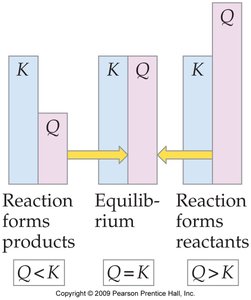
Definition and Use of Q
The reaction quotient () is calculated using the same expression as , but with initial (not necessarily equilibrium) concentrations. Comparing to predicts the direction the reaction will proceed:
If : The system is at equilibrium.
If : The reaction will proceed forward (toward products).
If : The reaction will proceed in reverse (toward reactants).
Le Châtelier’s Principle
Response to Disturbances
Le Châtelier’s Principle states that if a system at equilibrium is disturbed by a change in concentration, temperature, or pressure, the system will shift to counteract the disturbance and re-establish equilibrium.
Adding reactant: Shifts equilibrium toward products.
Removing product: Shifts equilibrium toward products.
Changing pressure (gases): Increasing pressure favors the side with fewer moles of gas.
Changing temperature: For endothermic reactions, increasing temperature favors products; for exothermic, it favors reactants.
The Haber Process Example
The synthesis of ammonia from nitrogen and hydrogen is an industrially important equilibrium process. Manipulating conditions such as concentration, pressure, and temperature can shift the equilibrium to increase ammonia yield.
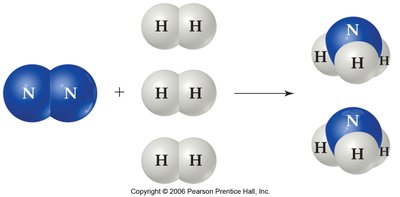
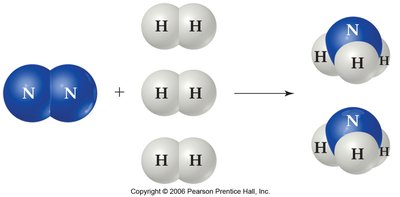
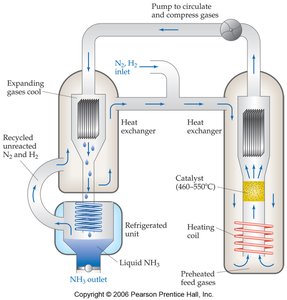
Catalysts and Equilibrium
Effect of Catalysts
Catalysts increase the rate at which equilibrium is achieved by lowering the activation energy for both the forward and reverse reactions. However, they do not affect the equilibrium position or the value of the equilibrium constant.
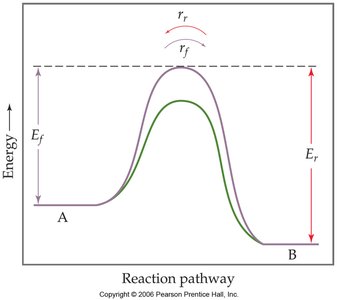
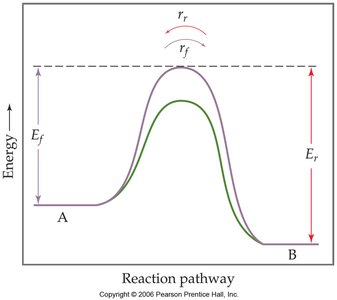
Practice and Application
Writing Equilibrium Constant Expressions
For the reaction: , the correct equilibrium constant expression is:
Solids and liquids are omitted from the expression.

Summary Table: Key Relationships in Equilibrium
Change | Effect on Equilibrium |
|---|---|
Add reactant | Shifts toward products |
Remove product | Shifts toward products |
Increase pressure (gases) | Shifts toward fewer moles of gas |
Increase temperature (endothermic) | Shifts toward products |
Add catalyst | Equilibrium reached faster; position unchanged |
Additional info: For more complex equilibrium calculations, especially when is very small or very large, approximations or the quadratic formula may be used to solve for unknown concentrations.
