 Back
BackChemical Kinetics: Rates, Mechanisms, and Catalysis
Study Guide - Smart Notes

Chemical Kinetics
Introduction to Reaction Rates
Chemical kinetics is the study of the speed at which chemical reactions occur and the factors that influence these rates. Understanding kinetics is essential for controlling reactions in industrial, environmental, and biological contexts. - Reaction rates can vary from extremely fast (explosions) to extremely slow (corrosion, diamond conversion). - Kinetics provides insight into the mechanism of a reaction, revealing how reactants transform into products. 


Initiation and Timescales
Some reactions require an external trigger (such as heat or a spark) to begin. The timescale of a reaction can range from femtoseconds to billions of years, depending on the process. 

Thermodynamics vs. Kinetics
Energy Diagrams and Reaction Pathways
Chemical reactions are governed by both thermodynamics and kinetics, which are distinct concepts: - Thermodynamics determines the equilibrium position and the relative stability of reactants and products (energy difference, ΔE). - Kinetics determines how quickly a reaction proceeds (energy barrier, Ea). - The activation energy (Ea) is the minimum energy required for a reaction to occur.
Factors Affecting Reaction Rates
Physical State, Concentration, Temperature, and Catalysts
Several factors influence how fast a reaction occurs: 1. Physical state: Reactions are faster when reactants are in the same phase (homogeneous). Increased surface area speeds up heterogeneous reactions. 2. Concentration: Higher reactant concentrations lead to more collisions and faster reactions. 3. Temperature: Higher temperatures increase kinetic energy and collision frequency, accelerating reactions. 4. Catalysts: Catalysts lower the activation energy and alter the reaction pathway, increasing the rate without being consumed.
Measuring Reaction Rates
Types of Reaction Rates
The rate of a reaction is defined as the change in concentration of a reactant or product over time: - Average rate: Change over a finite time interval. - Instantaneous rate: Rate at a specific moment, given by the slope of the concentration vs. time curve. - Initial rate: Rate at the very start of the reaction (t = 0), important for determining rate laws.
Normalized and Relative Rates
Stoichiometry and Rate Expressions
The rate can be expressed for different reactants and products, normalized by their stoichiometric coefficients. For example, in the Haber-Bosch reaction:
The Rate Law
General Form and Reaction Order
The rate law relates the reaction rate to the concentrations of reactants: - k: Rate constant (units depend on reaction order) - n, m: Reaction orders (experimentally determined) - Overall order: Sum of exponents (n + m)
Determining Rate Laws
Method of Initial Rates
Rate laws are determined experimentally by measuring how the initial rate changes with varying reactant concentrations. - Take ratios of rates and concentrations to solve for reaction orders. - Use logarithms to isolate exponents:
Integrated Rate Laws
Predicting Concentrations Over Time
Integrated rate laws allow calculation of reactant or product concentrations at any time. First-order reaction:  Half-life for first-order:
Half-life for first-order:  Second-order reaction:
Second-order reaction:  Zeroth-order reaction:
Zeroth-order reaction: 
Linearization of Integrated Rate Laws
Graphical Methods
Each integrated rate law can be linearized for easier analysis: - First-order: Plot vs. (slope = -k) - Second-order: Plot vs. (slope = k) - Zeroth-order: Plot vs. (slope = -k) 


Collision Theory and Arrhenius Equation
Successful Collisions and Activation Energy
- Reactions occur when molecules collide with proper orientation and sufficient energy. - Activation energy (Ea) is the minimum energy required for a productive collision. - The Arrhenius equation relates the rate constant to temperature and activation energy:
Reaction Mechanisms
Elementary Steps and Rate-Determining Step
- Elementary reactions are single-step processes; their rate laws can be written directly from stoichiometry. - Most reactions are multi-step, with the rate-determining step (slowest step) controlling the overall rate. 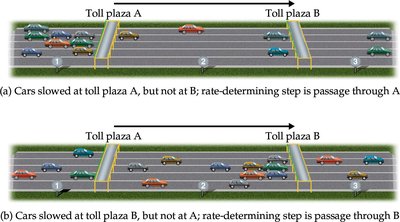
Intermediates and Approximations
- Intermediates are species formed and consumed during the reaction. - Pre-equilibrium approximation: Assumes fast equilibrium in an early step. - Steady-state approximation: Assumes intermediate concentration remains constant.
Catalysis
Homogeneous and Heterogeneous Catalysts
- Homogeneous catalysts: Same phase as reactants. - Heterogeneous catalysts: Different phase, often solid surfaces. - Catalysts lower activation energy, increase rate, but do not affect equilibrium.
Enzyme Kinetics
Mechanism of Enzyme Activity
Enzymes are biological catalysts that accelerate reactions by forming an enzyme-substrate complex. - Steady-state approximation is used to derive the rate law for enzyme-catalyzed reactions. 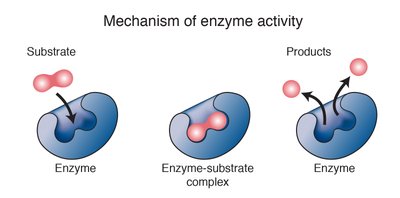
Summary Table: Integrated Rate Laws and Half-Lives
Order | Integrated Rate Law | Half-Life Expression |
|---|---|---|
First | ||
Second | ||
Zeroth |
Key Terms and Concepts
- Rate law: Mathematical relationship between rate and reactant concentrations. - Activation energy (Ea): Minimum energy for reaction. - Rate-determining step: Slowest step in a mechanism. - Catalyst: Substance that increases reaction rate without being consumed. - Enzyme: Biological catalyst. Additional info: This guide expands on brief points with academic context, definitions, and examples to ensure completeness and clarity for exam preparation.
