 Back
BackChemical Kinetics: Rates of Chemical Reactions
Study Guide - Smart Notes

Chemical Kinetics
Introduction to Chemical Kinetics
Chemical kinetics is the branch of chemistry that studies the speed, or rate, at which chemical reactions occur and the factors that influence these rates. Understanding reaction rates is crucial for controlling industrial processes, biological systems, and laboratory experiments.
Reaction Rate: The speed at which reactants are converted to products in a chemical reaction.
Mechanism: The step-by-step molecular pathway from reactants to products.
Importance: Engineers and chemists must consider both the products and the speed of reactions for practical applications.

Factors Affecting Reaction Rates
Several factors influence how quickly a chemical reaction proceeds:
Physical State of Reactants: Reactions occur faster when reactants are in the same phase (homogeneous). Heterogeneous reactions involving solids are faster with increased surface area (e.g., powders react faster than chunks).
Reactant Concentrations: Higher concentrations generally increase reaction rates due to more frequent molecular collisions.
Temperature: Raising the temperature increases molecular kinetic energy, leading to more frequent and energetic collisions, thus increasing the reaction rate.
Presence of a Catalyst: Catalysts speed up reactions by providing alternative pathways with lower activation energy, without being consumed in the process.

Effect of Concentration on Reaction Rate
Increasing the concentration of reactants typically increases the rate of reaction, as more molecules are available to collide and react.


Example: Steel wool oxidizes slowly in air (20% O2) but burns rapidly in pure O2 due to higher oxygen concentration.
Measuring Reaction Rates
Defining Reaction Rate
The rate of a chemical reaction is defined as the change in concentration of a reactant or product per unit time. It can be measured as an average rate, instantaneous rate, or initial rate.
Average Rate: Change in concentration over a time interval.
Instantaneous Rate: Rate at a specific moment, determined by the slope of the concentration vs. time curve.
Initial Rate: Instantaneous rate at the very start of the reaction (t = 0).
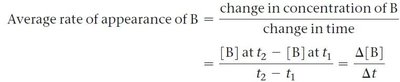
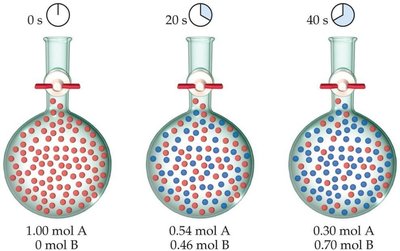

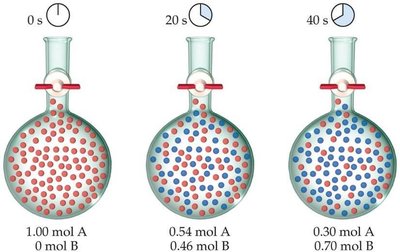
Example: Rate Data for Butyl Chloride Hydrolysis
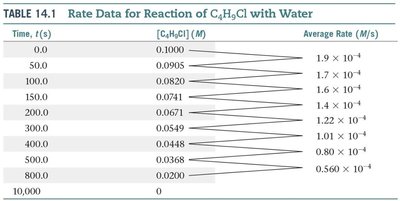
Consider the reaction: C4H9Cl(aq) + H2O(l) → C4H9OH(aq) + HCl(aq). The rate can be measured by monitoring the concentration of C4H9Cl over time.
Time, t (s) | [C4H9Cl] (M) | Average Rate (M/s) |
|---|---|---|
0.0 | 0.1000 | 1.9 × 10–4 |
50.0 | 0.0905 | 1.7 × 10–4 |
100.0 | 0.0820 | 1.6 × 10–4 |
150.0 | 0.0741 | 1.4 × 10–4 |
200.0 | 0.0671 | 1.2 × 10–4 |
300.0 | 0.0549 | 1.01 × 10–4 |
400.0 | 0.0448 | 0.80 × 10–4 |
800.0 | 0.0220 | 0.560 × 10–4 |
10,000 | 0 |
Calculating Instantaneous Rate
The instantaneous rate at a given time is the slope of the tangent to the concentration vs. time curve at that point.
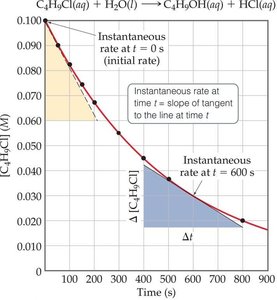
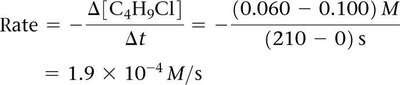
Example Calculation:
Stoichiometry and Relative Rates
Stoichiometric Relationships in Rate Expressions
For a general reaction: , the rate is related to the stoichiometric coefficients:
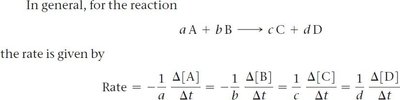
Determining the Effect of Concentration on Rate
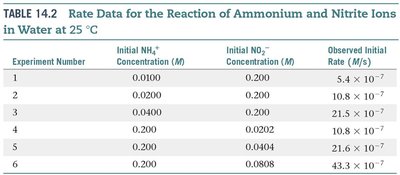
Experimental Determination of Rate Laws
To determine how each reactant affects the rate, experiments are performed where the concentration of one reactant is varied while others are held constant. The observed changes in rate reveal the reaction order with respect to each reactant.
Experiment Number | Initial NH4+ (M) | Initial NO2– (M) | Observed Initial Rate (M/s) |
|---|---|---|---|
1 | 0.0100 | 0.200 | 5.4 × 10–7 |
2 | 0.0200 | 0.200 | 10.8 × 10–7 |
3 | 0.0400 | 0.200 | 21.5 × 10–7 |
4 | 0.200 | 0.0202 | 10.8 × 10–7 |
5 | 0.200 | 0.0404 | 21.6 × 10–7 |
6 | 0.200 | 0.0808 | 43.3 × 10–7 |
Rate Law and Reaction Order
The rate law expresses the relationship between the rate and the concentrations of reactants, with exponents indicating the order with respect to each reactant:
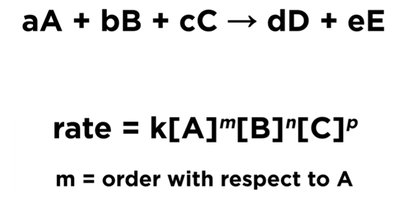
Key Points:
The order of reaction is determined experimentally and is not necessarily related to the stoichiometric coefficients.
The sum of the exponents gives the overall reaction order.
Types of Reaction Orders
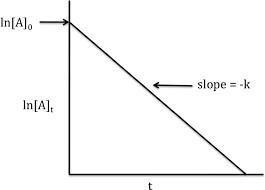
First-Order Reactions
For first-order reactions, the rate depends linearly on the concentration of one reactant:
The integrated rate law is:
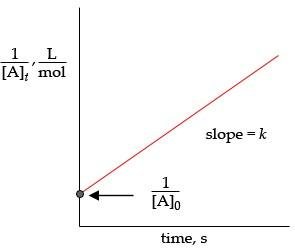
Second-Order Reactions
For second-order reactions (with respect to one reactant):
The integrated rate law is:
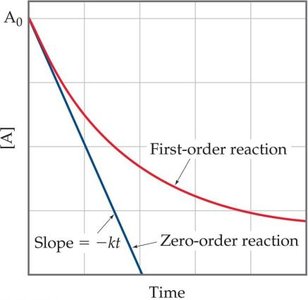
Zero-Order Reactions
For zero-order reactions, the rate is independent of the concentration of the reactant:
The integrated rate law is:
Half-Life of Reactions
First-Order Half-Life
The half-life () is the time required for the concentration of a reactant to decrease to half its initial value. For first-order reactions:
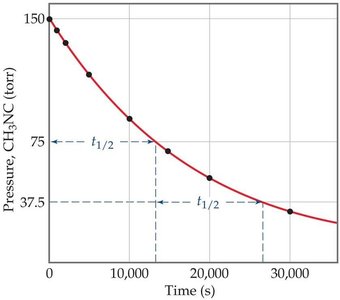
Second-Order Half-Life
For second-order reactions, the half-life depends on the initial concentration:
Summary Table: Reaction Order and Integrated Rate Laws
Order | Rate Law | Integrated Rate Law | Half-Life Expression |
|---|---|---|---|
Zero | rate = k | ||
First | rate = k[A] | ||
Second | rate = k[A]^2 |
Key Takeaways
Reaction rates are influenced by physical state, concentration, temperature, and catalysts.
Rates can be measured as average, instantaneous, or initial rates.
Rate laws must be determined experimentally and reveal the order of reaction with respect to each reactant.
Integrated rate laws allow calculation of concentrations at any time and determination of reaction order from experimental data.
Half-life expressions differ for zero-, first-, and second-order reactions.
